Nuclear Magnetic Resonance (NMR)
Quick Notes
- NMR is an analytical technique used to help determine molecular structures.
- 13C NMR and 1H NMR give information about the number of unique carbon and hydrogen environments in a molecule (number of peaks in a spectra) and positions of carbon and hydrogen atoms in a molecule (chemical shift, ppm, of each peak).
- 13C NMR produces simpler spectra compared to 1H NMR.
- Chemical shift (δ) values depend on the molecular environment of carbon-13 or H-1 nuclei.
- 1H NMR spectra use integration to show the relative number of protons in each environment and the n+1 rule to show splitting patterns, giving information about the number of hydrogens bonded to adjacent, non-equivalent carbon atoms.
- Tetramethylsilane (TMS) is used as a standard because it produces a single peak (with a chemical shift set as 0 ppm).
- For H-NMR, deuterated solvents or CCl4 are used to avoid interfering signals.
Full Notes
NMR Spectroscopy has been covered in more detail here.
This page is just what you need to know for Edexcel A-level :)
Introduction to NMR Spectroscopy
NMR (Nuclear Magnetic Resonance) Spectroscopy detects nuclei in a magnetic field. 13C and 1H NMR are the most commonly used techniques.
The hydrogen-1 and carbon-13 atoms have a property called nuclear spin, which means they act like tiny magnets. When placed in a strong magnetic field, these tiny magnets can absorb energy and 'flip' their spin – this is called resonance. The amount of energy needed to do this depends on their chemical environment. Because of this, atoms in different environments absorb slightly different amounts of energy.
Chemical shift (δ) is used to describe this energy and is measured in parts per million (ppm) relative to TMS (δ = 0).

The most important thing with NMR is to recognise unique carbon and hydrogen environments in a molecule, before worrying about more advanced areas like splitting. Remember it isn’t just the immediate atoms bonded to a carbon or hydrogen you need to look at, it is also what those atoms are themselves bonded to.
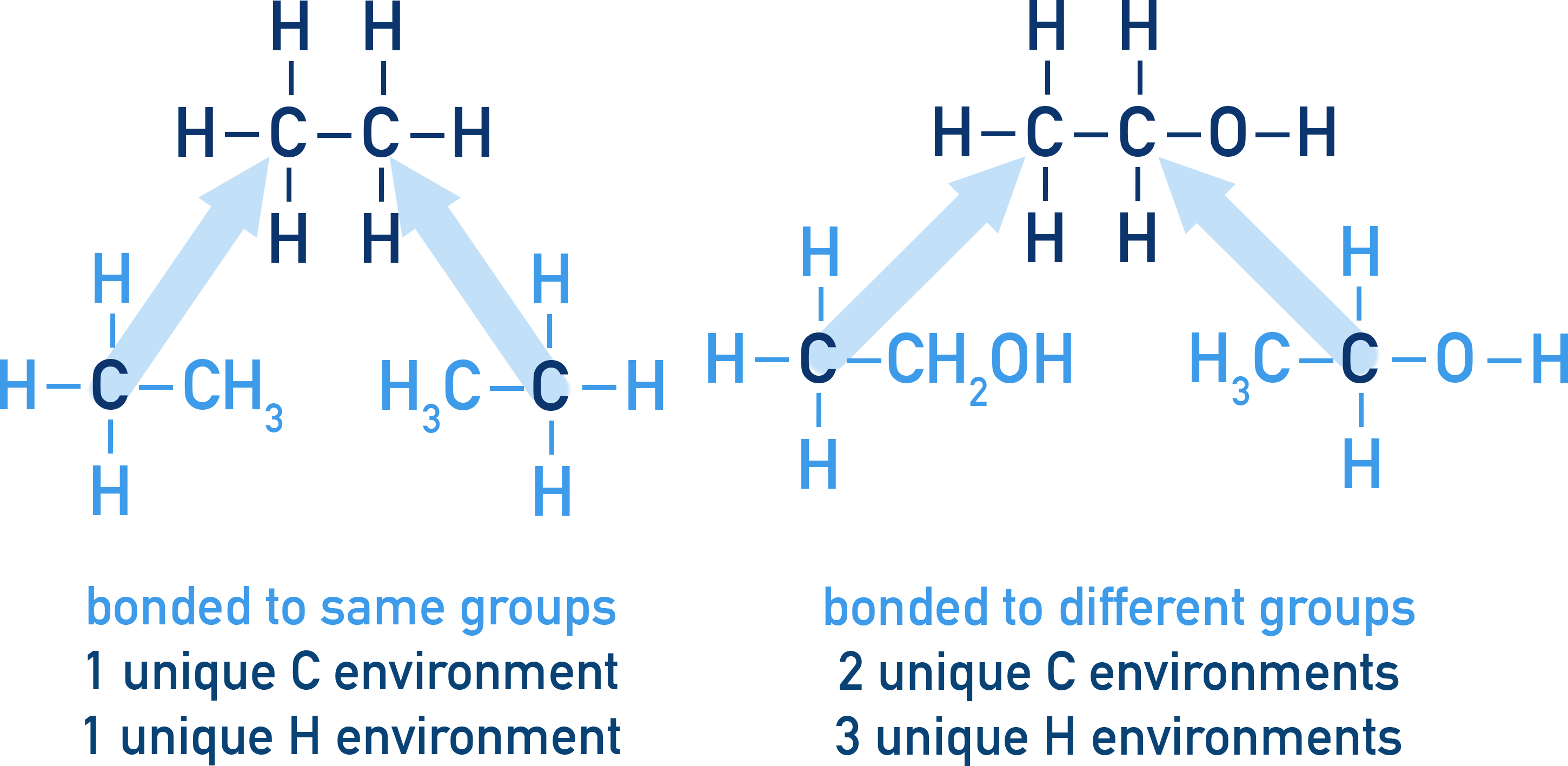
¹³C NMR vs ¹H NMR Quick Summary
| Feature | 13C NMR | 1H NMR |
|---|---|---|
| Detects | Carbon-13 nuclei | Hydrogen nuclei |
| Spectral Complexity | Simpler (1 peak per unique C environment) | More complex (splitting patterns) |
| Chemical Shift Range | 0 – 200 ppm | 0 – 12 ppm |
| Peak Integration | Not relevant | Shows relative proton numbers |
| Spin-Spin Splitting | Not observed | n+1 rule |
Chemical Shift (δ) and Molecular Environment
Chemical shift (δ) depends on electron density around a nucleus. More electronegative groups shift peaks downfield (higher δ values).
Data book values are used to compare peaks and identify possible bonding groups within a sample.
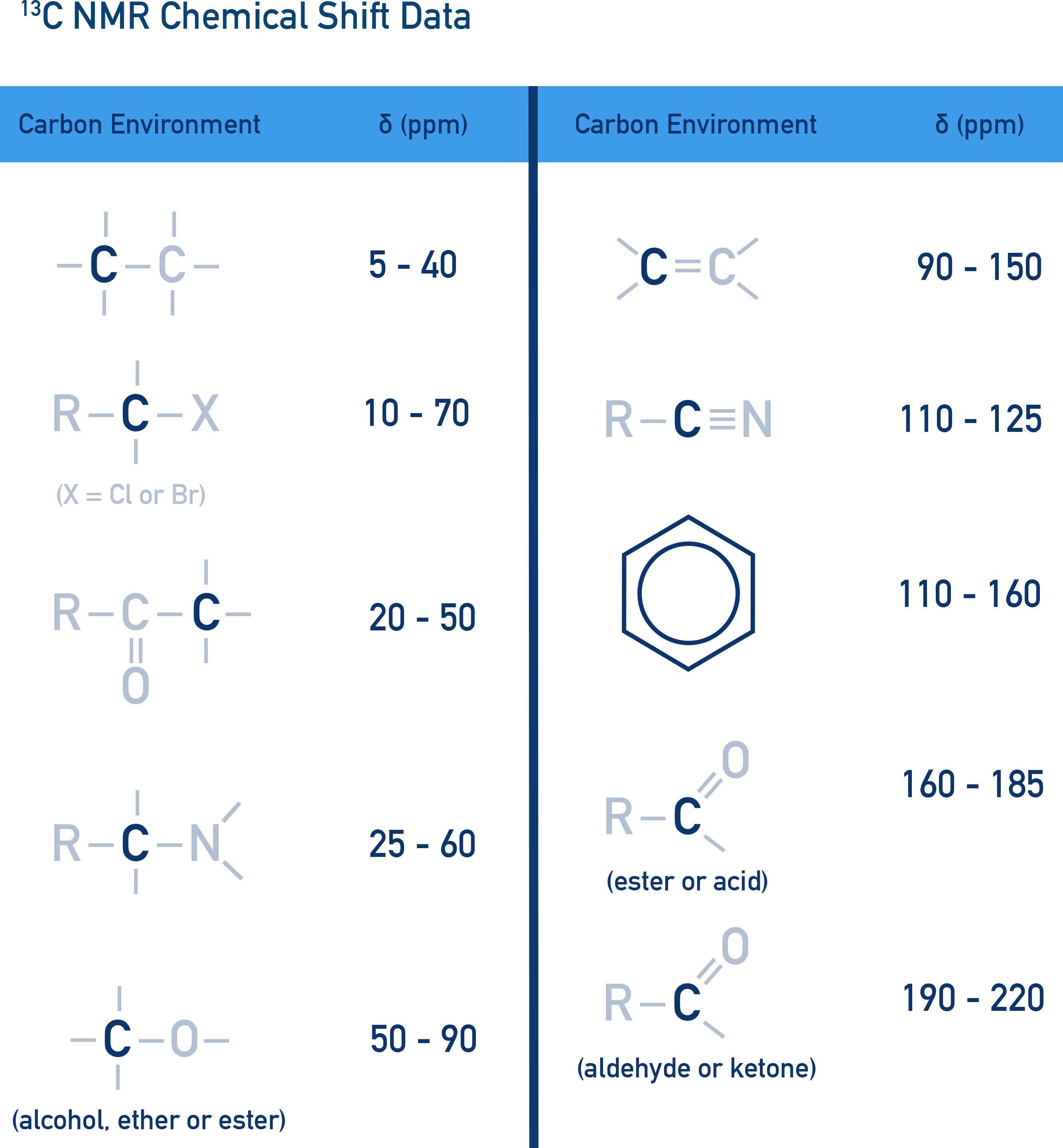
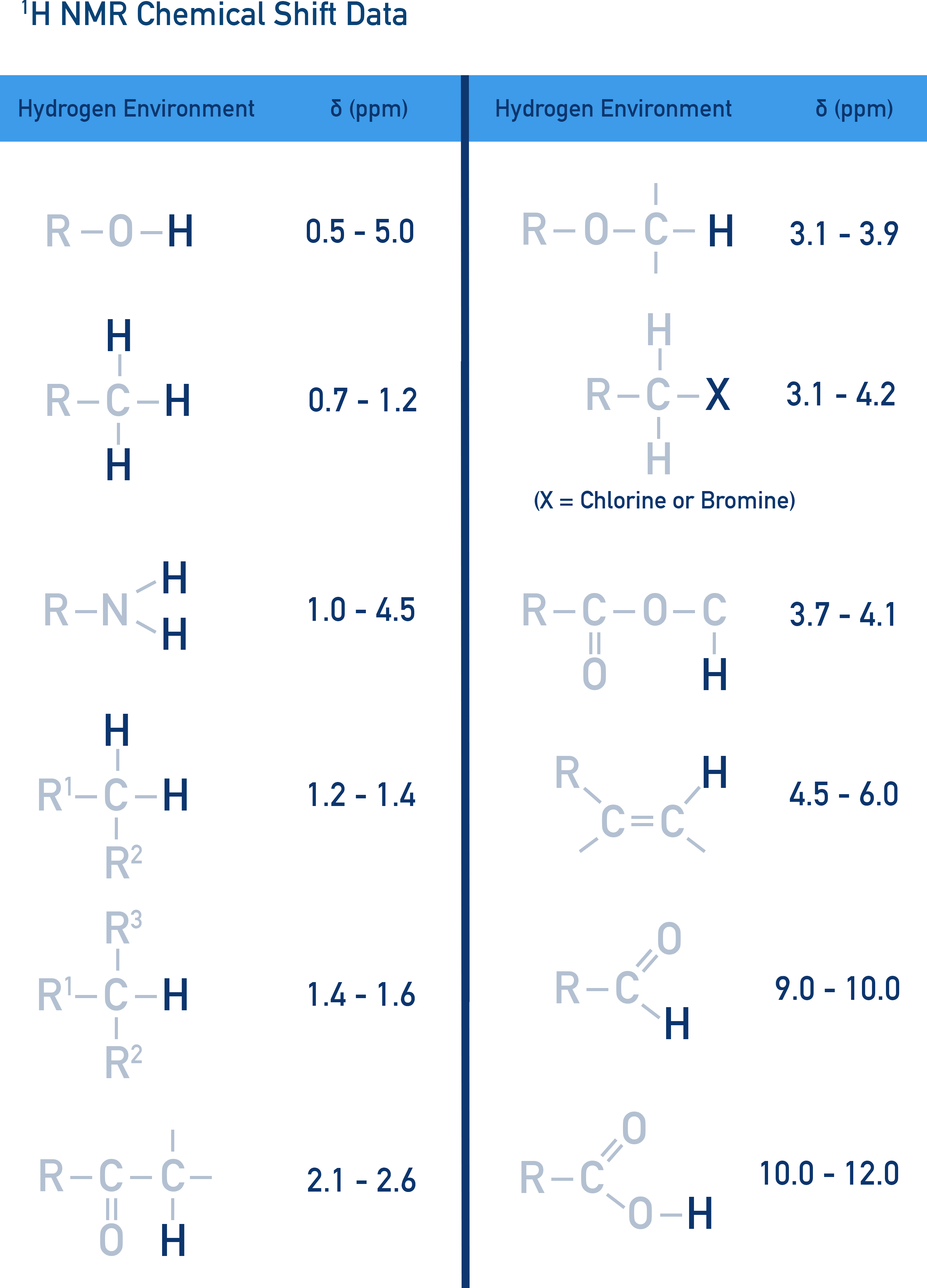
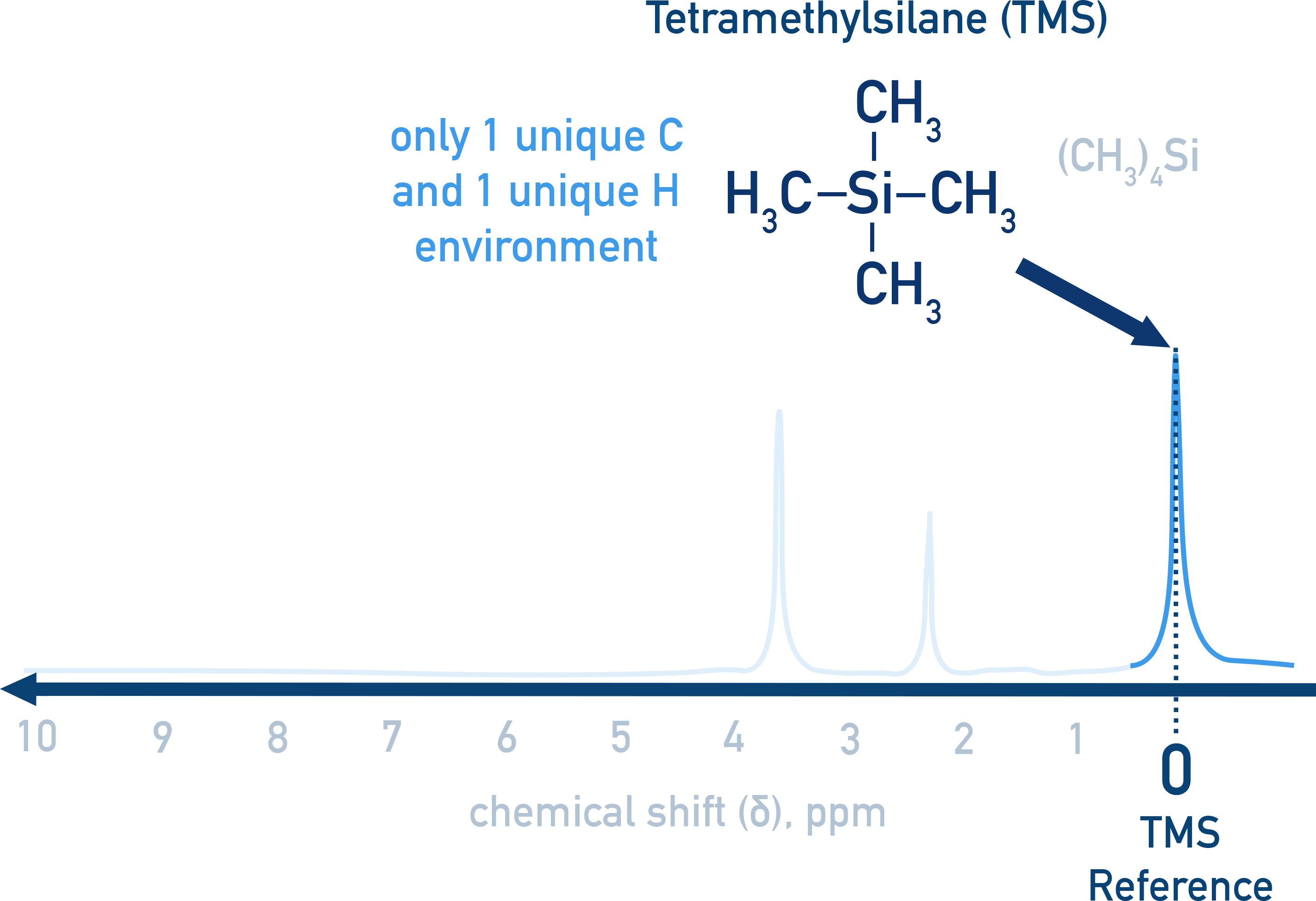
Tetramethylsilane (TMS) as a Standard
TMS ((CH3)4Si) is used as an internal standard (δ = 0 ppm). This enables comparison of absorbances in NMR spectra with data book values.
- Produces a single peak (all atoms in same environment)
- Non-reactive and volatile
- Low chemical shift (does not interfere with peaks)
¹H NMR Integration
Integration (peak area) shows the ratio of protons in each unique environment.
Example:
Propane (C3H8) shows two peaks with a ratio of 1:3, matching the hydrogen environments.
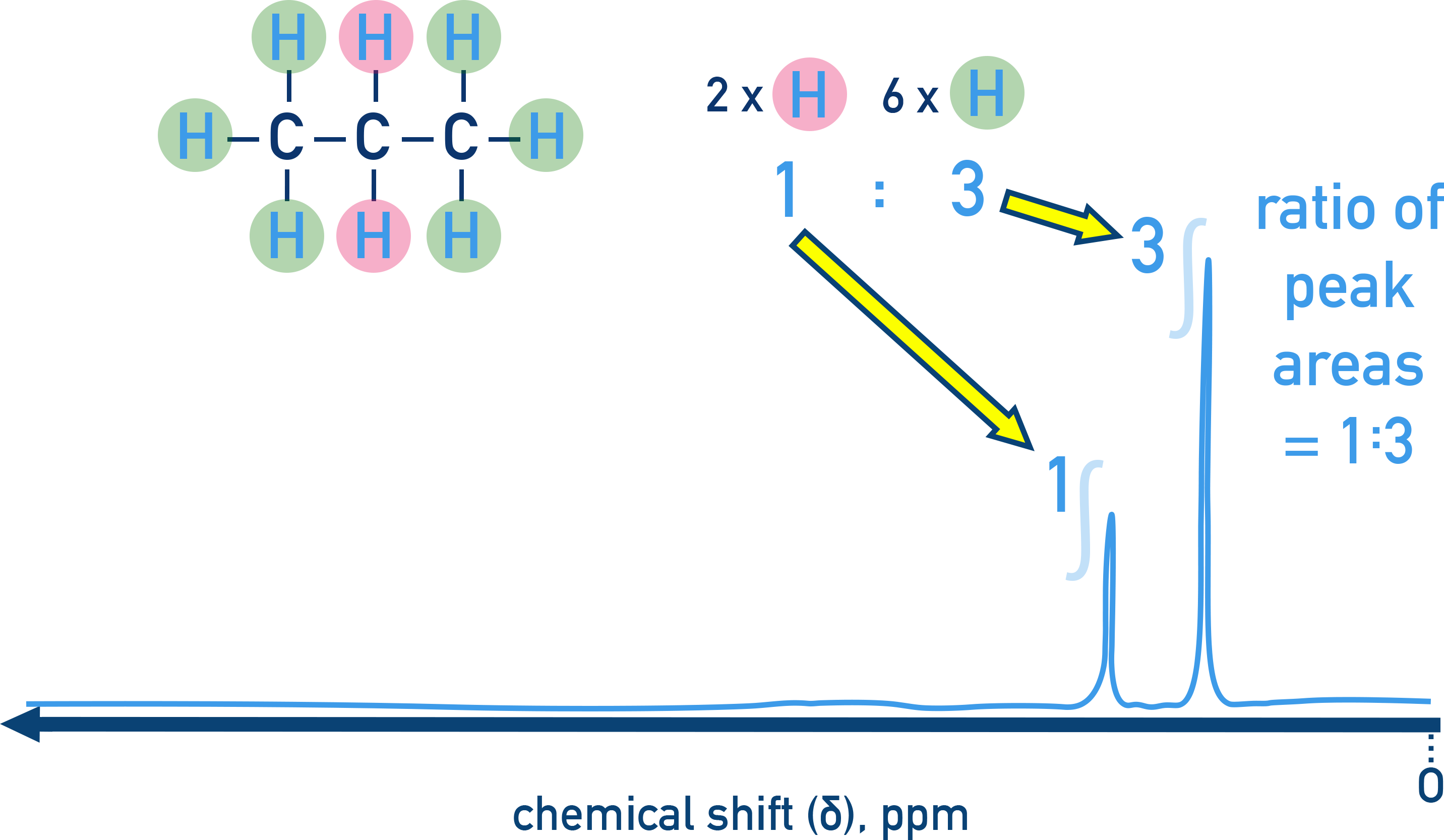

Be careful: integration ratios are just ratios, not the actual number of protons. Use the molecular formula to scale the ratios into actual numbers in each environment.
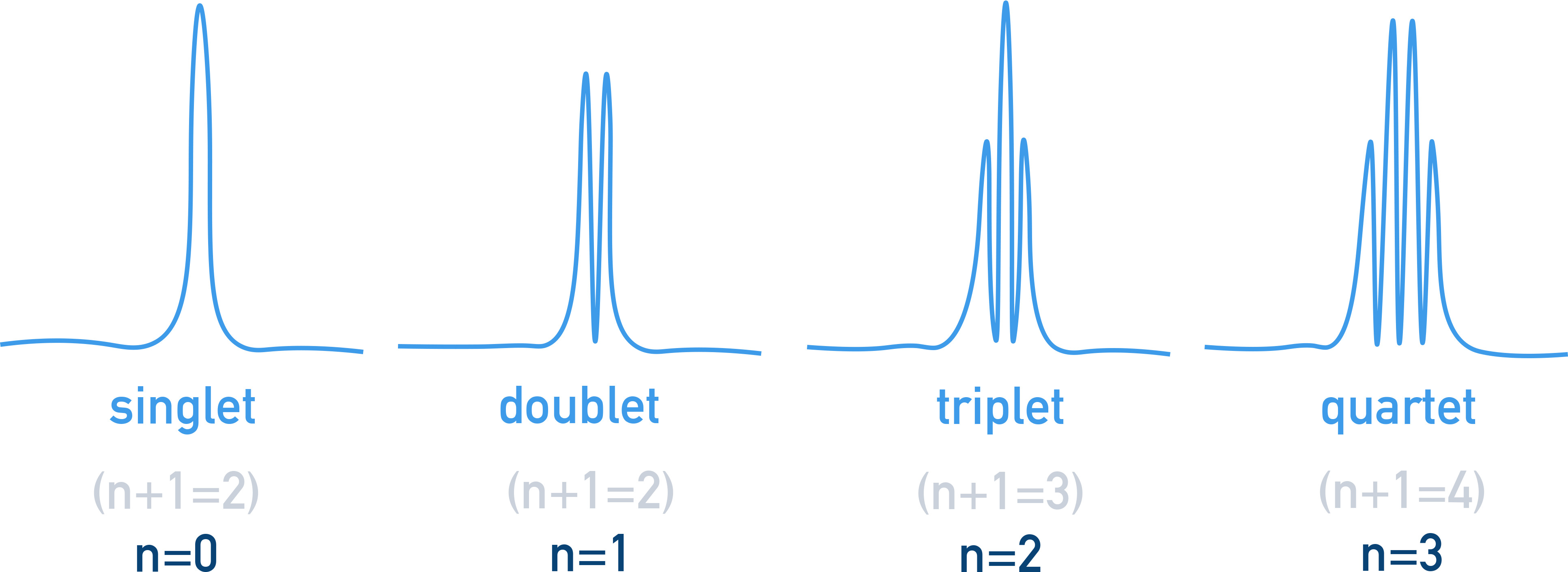
¹H NMR Spin-Spin Coupling (n+1 rule)
Hydrogens bonded to adjacent, non-equivalent carbon atoms cause peaks to split. The number of hydrogens on adjacent carbons determines how many times the peak is split, following the n+1 rule.
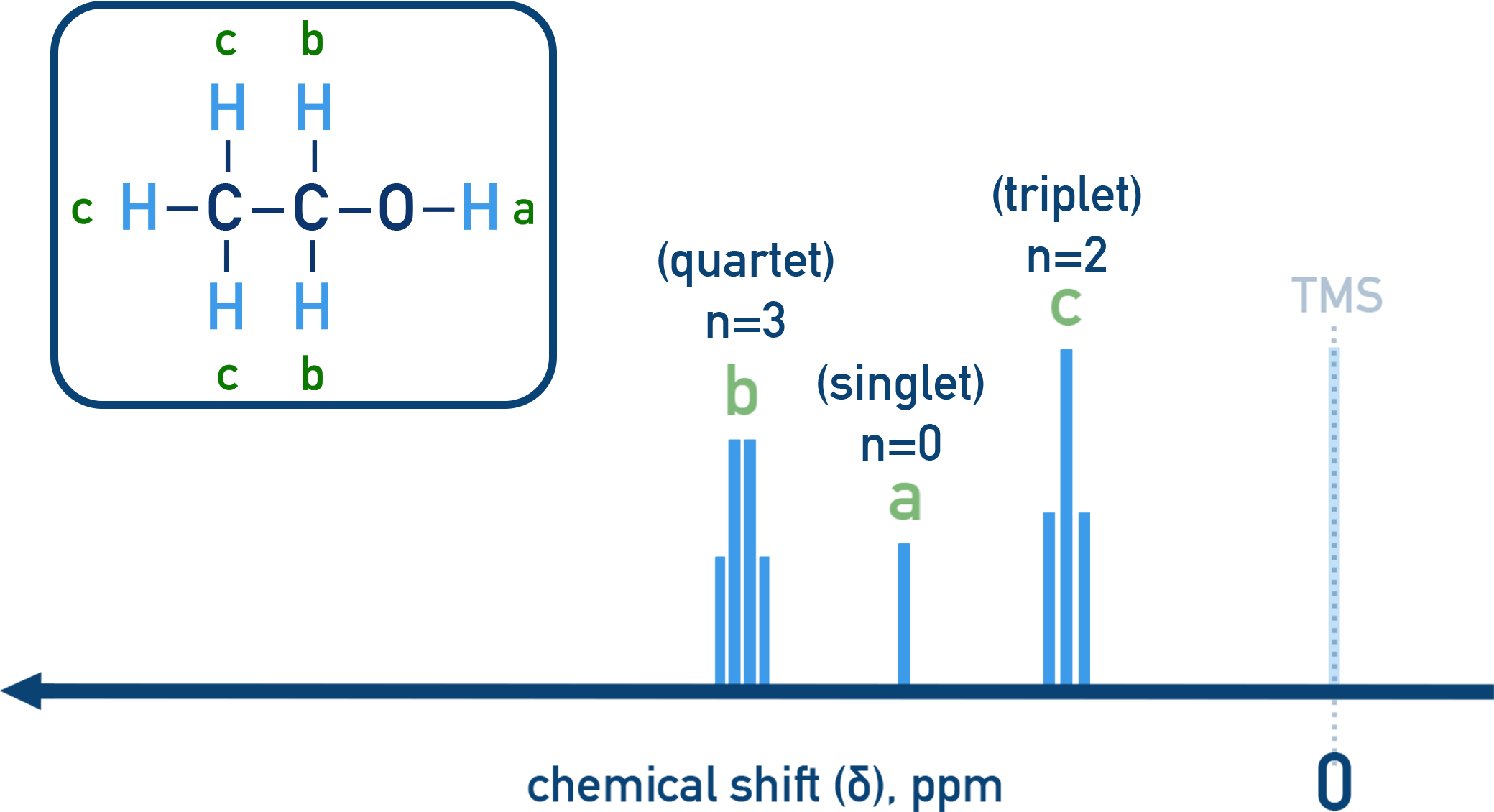
Example Ethanol in ¹H NMR
CH3 group shows a triplet (next to CH2).
CH2 group shows a quartet (next to CH3).
OH appears as a singlet (no splitting).
Summary
- NMR identifies molecular structures by detecting nuclei in magnetic fields.
- 13C NMR gives the number of carbon environments, 1H NMR gives hydrogen environments with splitting and integration.
- Chemical shift values depend on surrounding atoms and electronegativity.
- TMS is used as a standard peak at 0 ppm.
- Integration shows proton ratios, and splitting follows the n+1 rule.
