Quick Notes Organic Dyes
- Organic dyes are compounds bonded to materials to give the material a specific colour.
- Within organic dyes, electrons are excited from low energy to high energy by absorbing energy from the visible part of the electromagnetic spectrum.
- The part of the dye where the excitation of electrons occurs is called the chromophore.
- Most organic molecules absorb higher energy waves (ultraviolet light – not visible) to excite electrons.
- Increased levels of electron delocalisation decrease the energy required to excite electrons and enable visible light to be absorbed by the molecule, forming a chromophore.
- Wavelengths of light not absorbed by the chromophore are scattered by the dye and observed as the complimentary colour to the colour of light absorbed by the dye.
Full Notes Organic Dyes
Dyes are substances with a specific colour that can be bonded with materials, to make the material the same colour as the dye.
The part of an organic dye responsible for its colour is called a chromophore.
Sometimes, in a compound, electrons can be excited from a low energy to a high energy state. If the amount of energy required to do this can be obtained from visual light, electrons can be excited by absorbing specific wavelengths of light from white light. The wavelengths not absorbed are reflected (or scattered) by the compound and combine to produce a colour - this is the complimentary colour of the wavelengths of light absorbed by the chromophore.
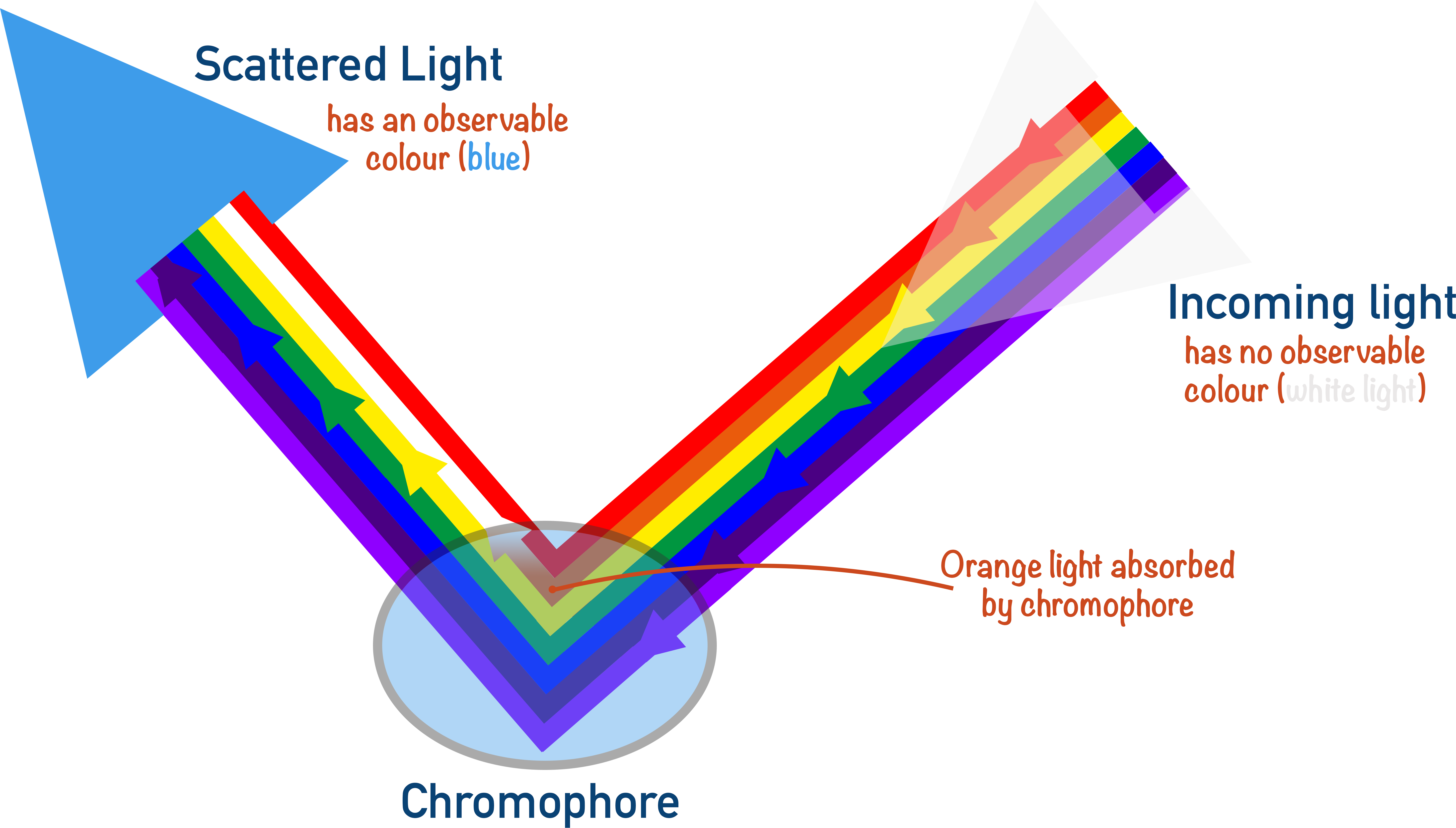
The chromophore is the part of the molecule where these electrons are excited using visible light.
For most organic molecules, visible light does not provide enough energy to excite electrons, so no visible light is absorbed. As a result, no visible colour is observed. Ultraviolet light contains more energy per wave than visible light and is often absorbed by organic molecules to excite electrons. Ultraviolet is not visible to human eyes, so the effect is not visibly observed.
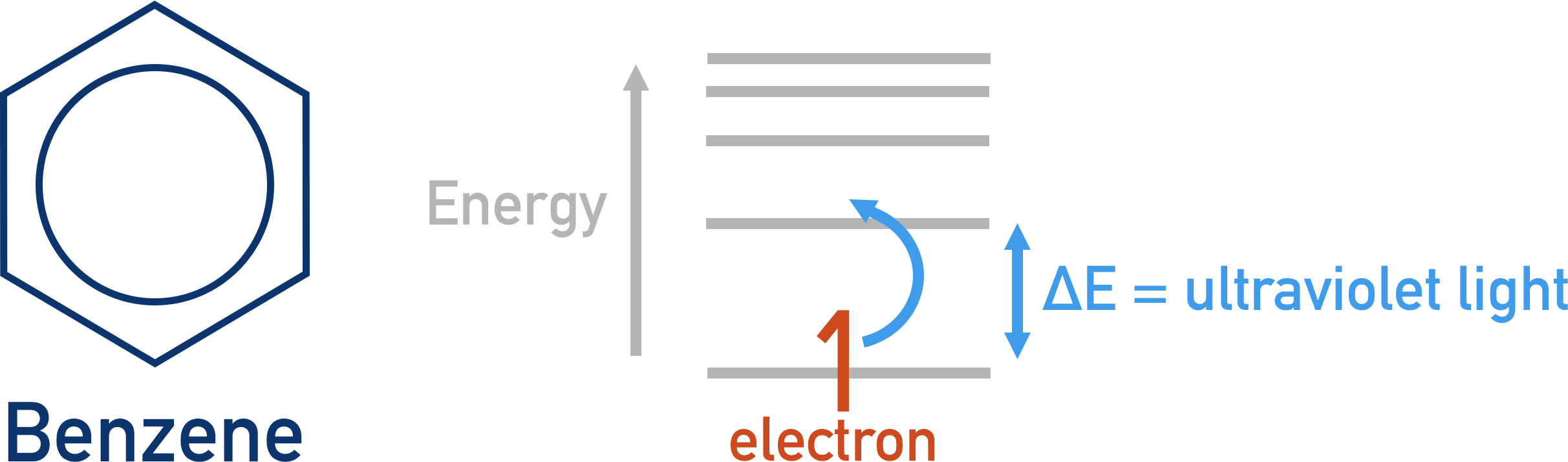
In benzene there is delocalisation of electrons and a pi-bond system, but the level of delocalisation is not enough to allow visible light to excite an electron.
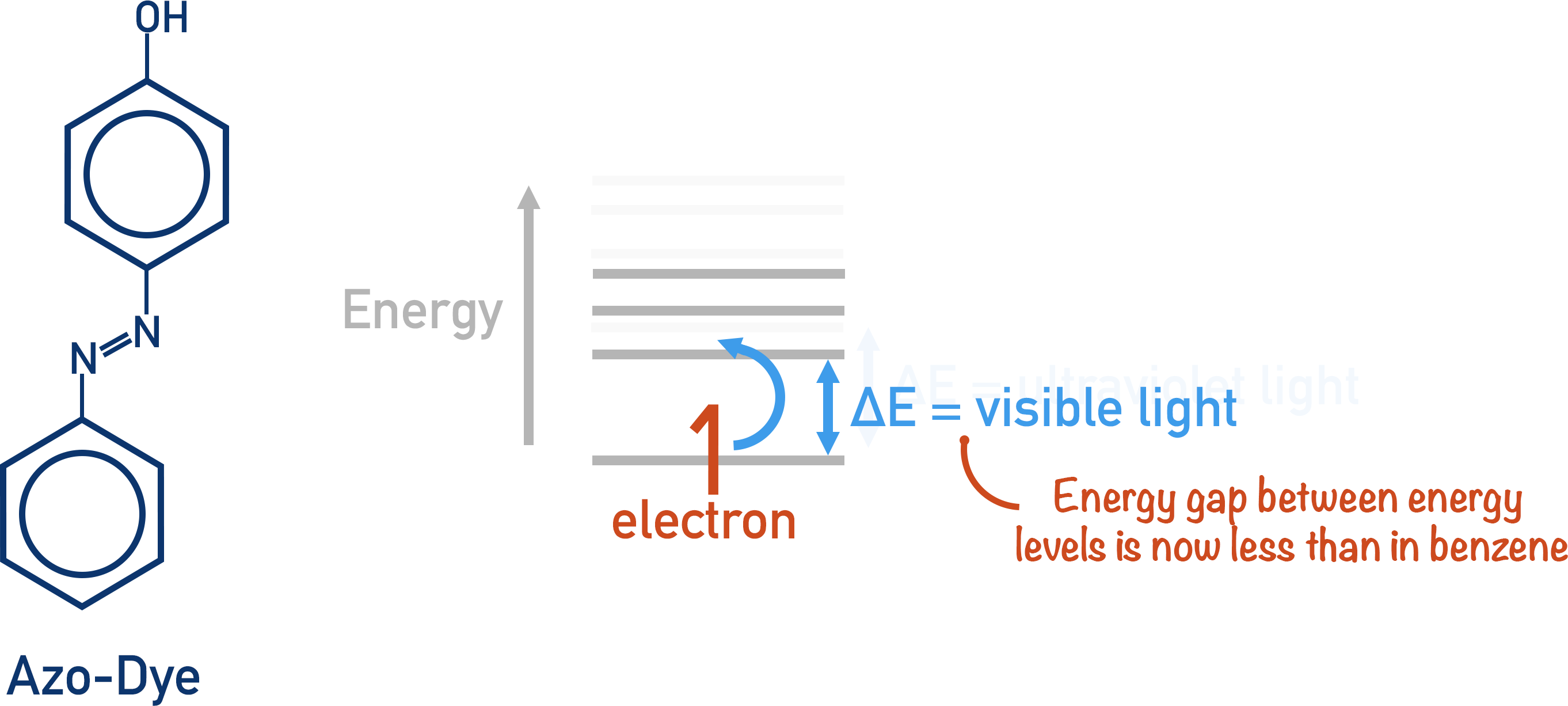
If the energy gap between a low energy state and a high energy state is made smaller, then the energy required to excite an electron is available in visible light.
The greater the degree of delocalisation of electrons within an organic molecule, the smaller the energy gap. This is why most organic dyes have high degrees of electron delocalisation (conjugated pi-bond systems).
Azo dyes have a very high level of delocalisation, and the energy gap is small enough for visible light to provide enough energy for an electron to be excited.
