Physical Chemistry
![]()

Physical chemistry is the study of the physical behaviour and interactions of matter on an atomic level. It is commonly considered as being a combination of physics and chemistry.
Physical chemistry is the most numerical (quantitative) part of chemistry and, as a result, frequently makes use of mathematical formulas.Current and exciting developments in physical chemistry centre around quantum mechanics - this is not studied in A-level Chemistry!
Atomic Structure
- Structure of an Atom
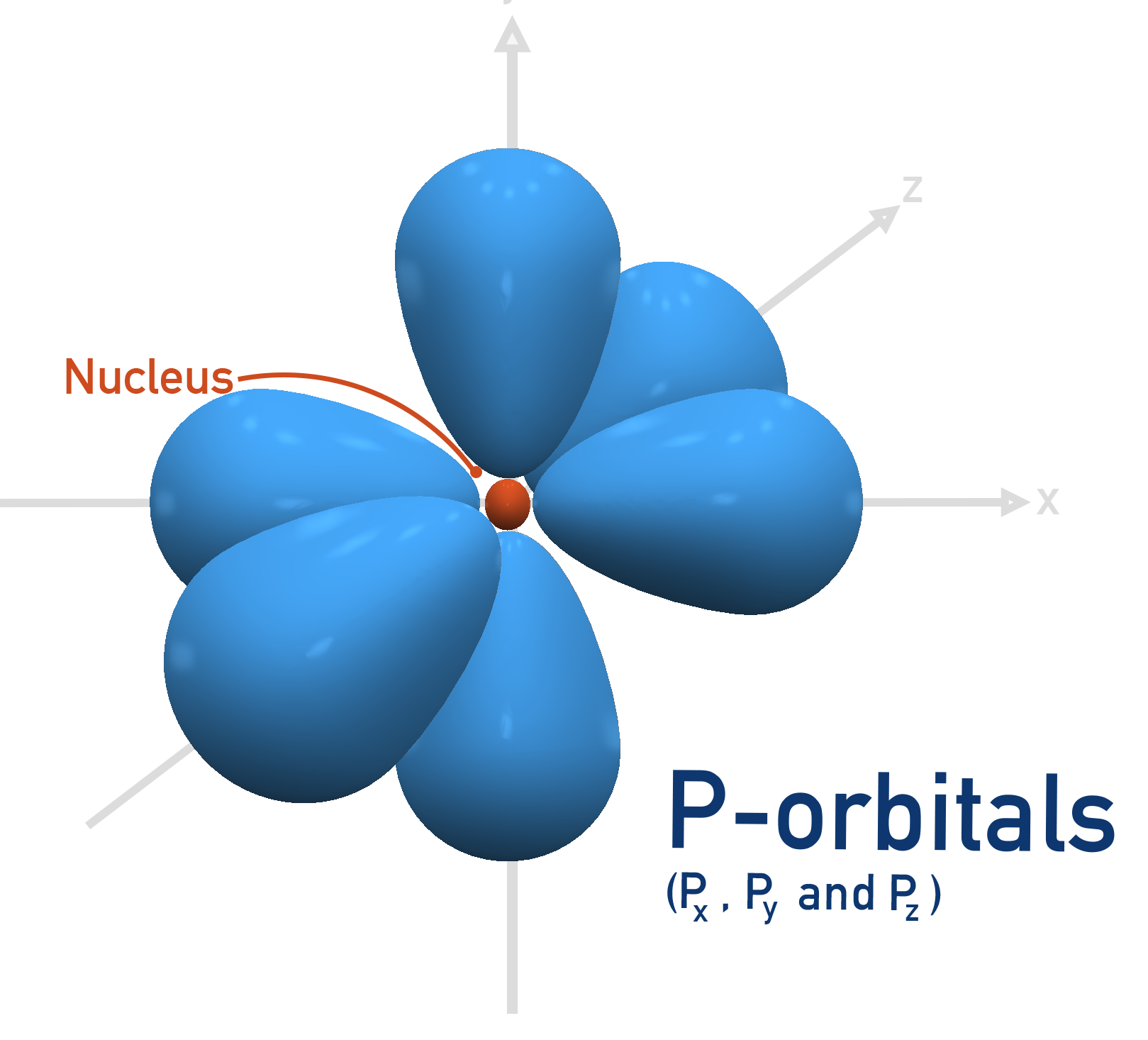
- Electron Orbitals
- Electron Configurations
- Mass Spectrometry
- Time of Flight Mass Spectrometry
Moles (Amounts)
- The Mole
- Relative Atomic Mass
- Moles - Mass
- Moles - Solutions
- Moles - Gases
- Empirical Formula
- Percentage Mass
- Atom Economy (%)
Bonding
- Bonding
- Covalent Bonding
- Co-Ordinate (dative covalent) Bonding
- Electronegativity
- Ionic Bonding
- Metallic Bonding
- Intermolecular Forces
- Permanent Dipole-Dipole
- Hydrogen Bonding
Energy and Enthalpy
Kinetics
Enthalpy and Entropy (Thermodynamics)
- Lattice Enthalpies
- Born-Haber Cycles
- Enthalpy of Solution
- Entropy
- Entropy Change
- Free Energy (Gibbs)
- Total Entropy
Rates of Reaction (Kinetics)
Equilibrium
Electrochemistry
Acids and Bases
