Video Tutorial Iodine Clock Reaction
Quick Notes Clock Reactions
- Clock reactions are used to find the initial rate of a reaction.
- The length of time taken to form a small amount of product is measured.
- A colour change or observation is used to show when this small amount of product has formed.
- Measuring the initial rate of a reaction is difficult because the concentrations of the reactants are constantly changing, meaning the rate of reaction is also changing.
- The iodine clock experiment is a common example of a clock reaction at A-level Chemistry.
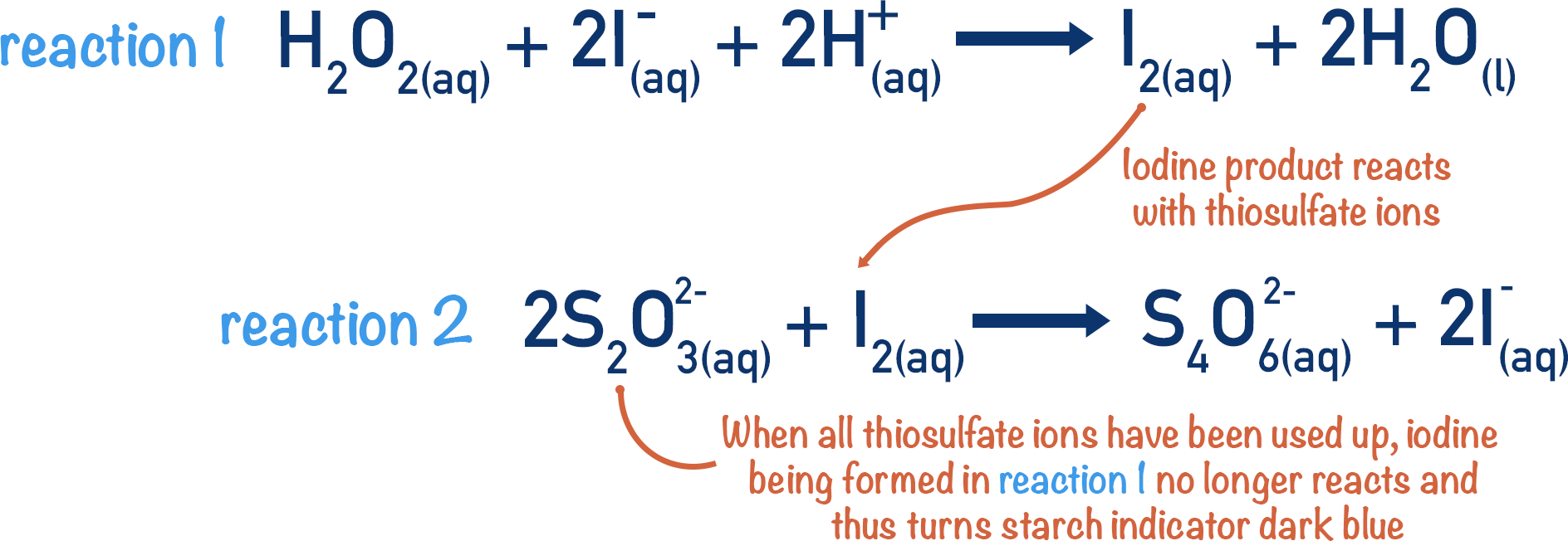
- In the main reaction, hydrogen peroxide reacts with iodide ions to produce iodine molecules.
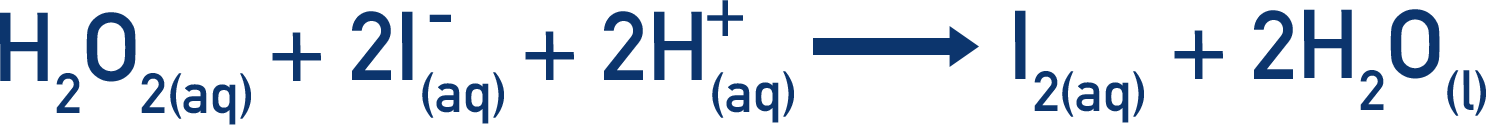
- In a second reaction, thiosulfate ions react with the iodine produced in the main reaction to form iodide ions again.

- Starch indicator is added to the reaction mixture, which turns dark blue in the presence of iodine. As long as there are thiosulfate ions in the mixture, the starch will not cause a colour change.
- The thiosulfate ions are instantly converting any iodine molecules formed into iodide ions.
- When all the thiosulfate ions are used up the starch will cause a color change.
- Iodine molecules will no longer be reacted back to iodide ions by the thiosulfate ions and they can react with the starch (causing colour change).
- The amount of thiosulfate ions in the mixture is known and the length of time taken for the mixture to change color is recorded.
- This is used to find how long it took to form a specific amount of iodine and give the initial rate of reaction.
- In the main reaction, hydrogen peroxide reacts with iodide ions to produce iodine molecules.
Full Notes Clock Reactions
The initial rate of a reaction refers to how fast a reaction is happening at the very start of the reaction. The rate of a reaction changes as the reaction proceeds, due to changes in the concentrations of the reactants (see Rates of Reaction).
To find a rate of a reaction, the change in concentration of a reactant (or product) needs to be measured against time. Finding changes of a concentration during the initial stages of a reaction can be very difficult because the concentration is changing rapidly as the reaction proceeds.
One effective method is to time how long it takes to produce a small amount of product compared to the starting amount of reactants. This means that, although the amount of the reactants is changing, the change is very small compared to the concentration overall – meaning the change to the rate is minimal, enabling us to study the initial rate of the reaction.
In order to determine when a small amount of product has been made, we need a visual indication of some kind – usually an indicator (although sometimes precipitate formation can be used). The problem is indicators can be very sensitive and will often change colour as soon as a product is formed – this is no good as we won’t know exactly how much product has been formed to cause the colour change. The colour change would also happen so fast, we would never be able to time it accurately.
To overcome this, another substance is added to the mixture that reacts with the product from the main reaction. This means that no colour change will occur until all of this substance is used up. By adding a known amount of this ‘new reactant’, we can determine how much product must have been formed from the first reaction by the time a colour change occurs.
This process is called a clock reaction. Clock reactions can be confusing to A-level students, but their idea is actually very simple.
Iodine Clock Reaction
A common example of a clock reaction at A-level Chemistry is the iodine clock reaction.
The basic reaction involves hydrogen peroxide and potassium iodide (in the presence of an acid catalyst).
The hydrogen peroxide oxides the iodide ions and iodine (I2) is formed.
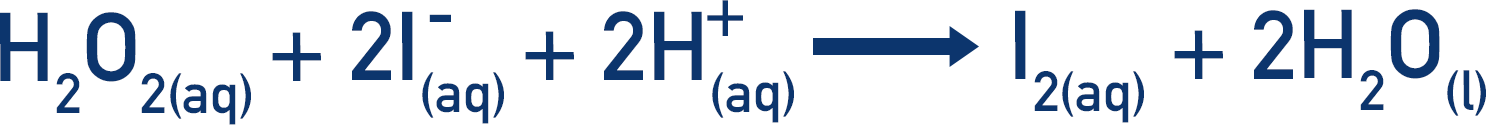
Using a simple starch indicator, we can see when iodine has been formed (starch turns dark blue (blackish) in the presence of iodine).
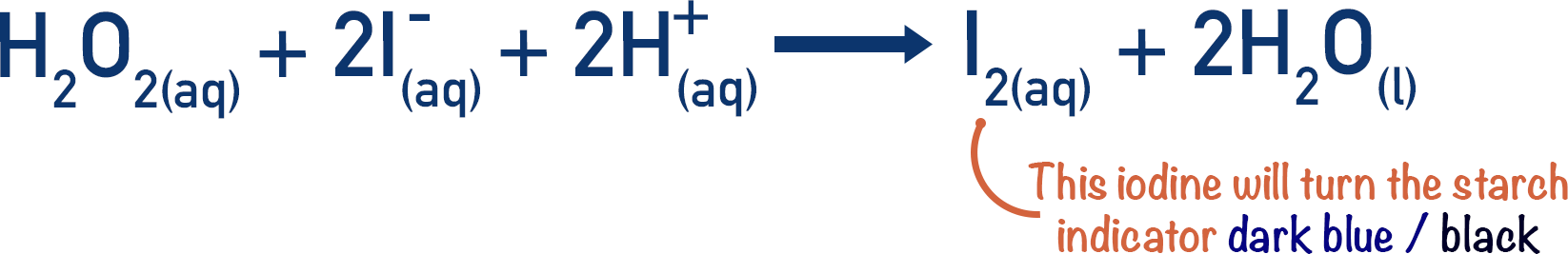
However, as we are trying to determine the initial rate of the reaction we also need to time how long it takes to form a specific amount of iodine.
To do this, a known amount of sodium thiosulfate is added. Thiosulfate ions react with iodine to form iodide ions.
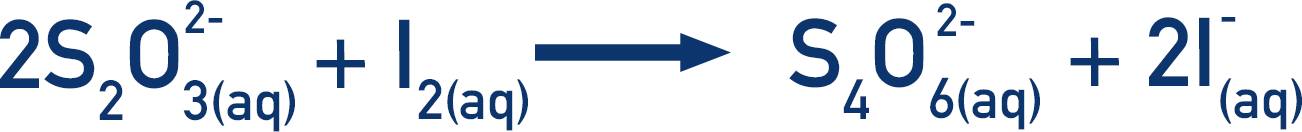
Now, every time a molecule of iodine is made in the first reaction, it is instantly converted back into iodide ions by the thiosulfate ions. As long as the thiosulfate ions are reacting with the iodine formed, there will be no colour change to the mixture (as there is no iodine to turn the starch indicator dark blue).
As soon as the thiosulfate in the mixture is used up, however, the iodine formed by reaction one stays as iodine. This means the solution turns dark blue (due to the starch indicator).
The time it takes the solution to change colour is determined by the amount of thiosulfate ions there are at the start. The higher the amount of thiosulfate ions, the longer it will take for the solution to change colour. As we are trying to find the initial rate of reaction, we want the amount of thiosulfate ions to be very small compared to the amount of hydrogen peroxide and iodide ions in the mixture (see above).
If, for example, one mole of thiosulfate ions is present in the mixture when the hydrogen peroxide and iodide ions are mixed, and it takes 30 seconds for the solution to change colour, this means it has taken 30 seconds to produce 0.5 moles of iodine (the reacting ratio of thiosulfate ions to iodine is 2:1).
We now have a ‘rate of reaction’, as we know how long it took to produce a set amount (0.5 moles) of iodine.
By changing the concentration of hydrogen peroxide or potassium iodide, but keeping the amount of thiosulfate ions the same, we can see how the rate of reaction changes as we change the concentrations of each reactant. This is now just the same as determining a rate equation from reaction data (see Rate Equation).
We’ve launched our new site! 🎉
Course-specific notes with built-in search!
AP • A-Level (AQA • CIE • Edexcel • OCR) • IB • NCERT 11 + 12
over 750+ new pages and 3,500 images.
Visit the new homepage
