Video Tutorial Collision Theory
Quick Notes Collision Theory
- Kinetics is the study of how fast reactions occur.
- How quickly a reaction happens is called its reaction rate.
- Reactions occur when reactant particles collide together with enough energy (activation energy, Ea).
- Collisions between particles that lead to a reaction are called ‘successful collisions’.
- Increasing the frequency of successful collisions increases the rate of reaction.
- Increasing the temperature, concentration and pressure of reacting particles increases the rate of a reaction.
Full Notes Collision Theory
The study of kinetics (in chemistry) is concerned with how quickly reactions happen and how altering conditions can change the speed of reactions. The speed at which a reaction occurs is called the rate of reaction. It is usually measured by a change in concentration in a given amount of time, giving the units mol dm-3 s-1. The concentration change can be of either a reactant or product.
Collision Theory
All particles are in constant motion (even in solids, particles are constantly vibrating – albeit with limited motion), so particles are constantly ‘bumping’ into each other. This ‘bumping’ we call colliding.

If two reacting particles collide into each other hard enough (and with the right ‘orientation’), a reaction takes place – this is called a successful collision. How ‘hard’ a collision is depends upon how much energy each particle has when they collide. The minimum amount of energy that particles need for a successful collision is called the activation energy (Ea).
Right ‘orientation’ refers to the way the particles collide. For certain reactions, particles have to hit each other in a certain way to react – a bit like going through a door, you have to push the door with enough energy to make it open, but it will only open if you push in a particular direction! This isn’t very important for A-level Chemistry and is generally only mentioned when dealing with the Arrhenius Equation (second year, A2-level).
Since ‘rates of reaction’ refer to how quickly reactions happen, it’s the frequency of successful collisions that determine a reaction’s rate. Not the total number of successful collisions.
Frequency of successful collisions refers to the number of successful collisions that occurs in a given amount of time (often per second, s-1).
The majority of collisions between particles are not successful, as the particles colliding do not collide with enough energy to react.
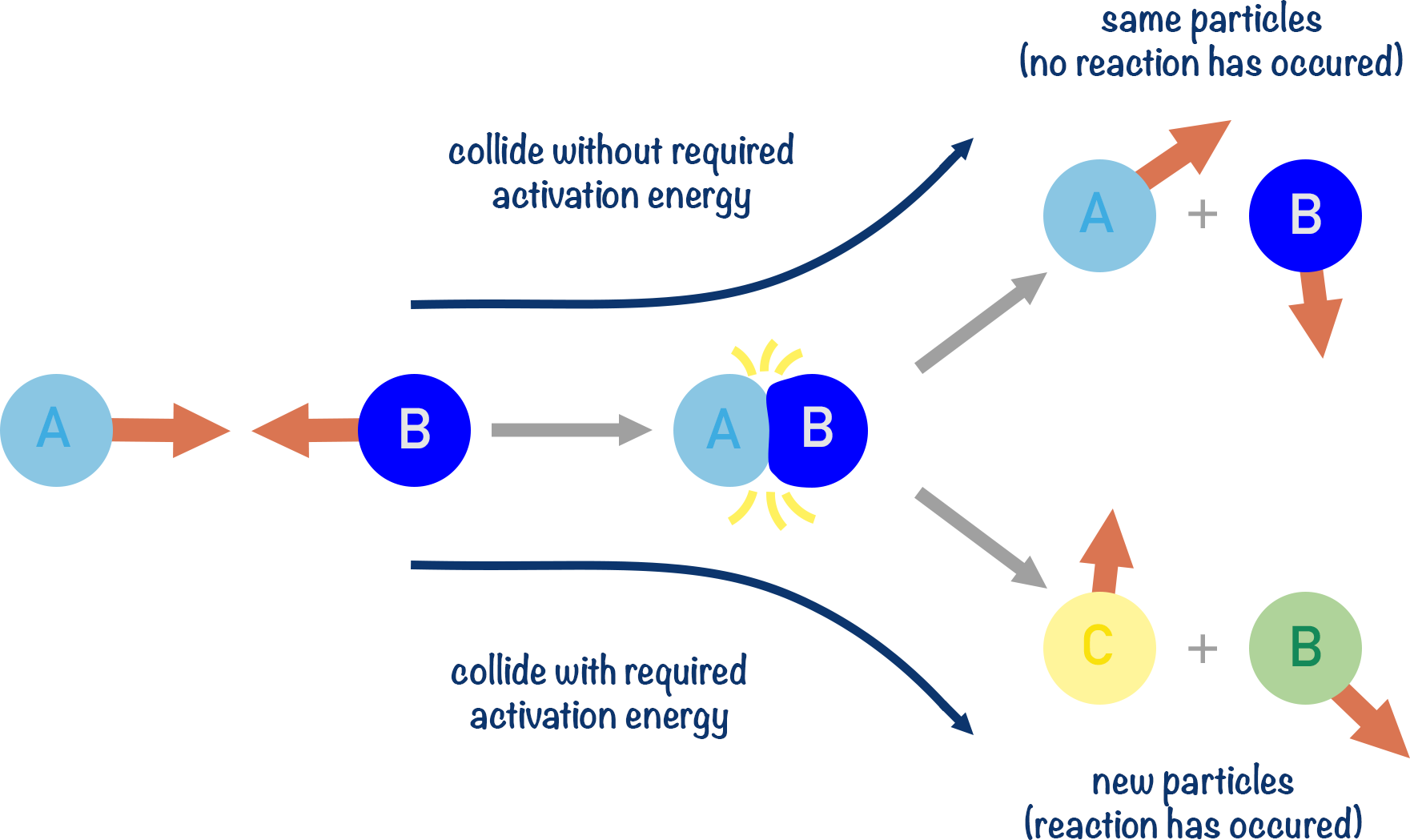
Activation energy is fixed for a particular reaction and cannot be changed (unless a catalyst is used, see below). So, when we want to increase the rate of a reaction, we have to give the reacting particles more energy or increase the number of particles colliding. This is why reactions (usually) happen faster at higher temperatures and when higher concentrations of reactants are used.
Effect of Temperature on Reaction Rate:
Imagine the reaction A + B → C. If we carry out this reaction at 25oC and at 75oC, at which temperature would we expect the reaction to happen fastest?
At 75oC! If the temperature is higher, particles A + B would have more energy. When particles collide with more energy, more collisions are successful (more collisions with the required activation energy). This means there will be more successful collisions in a given amount of time compared to at 25oC.
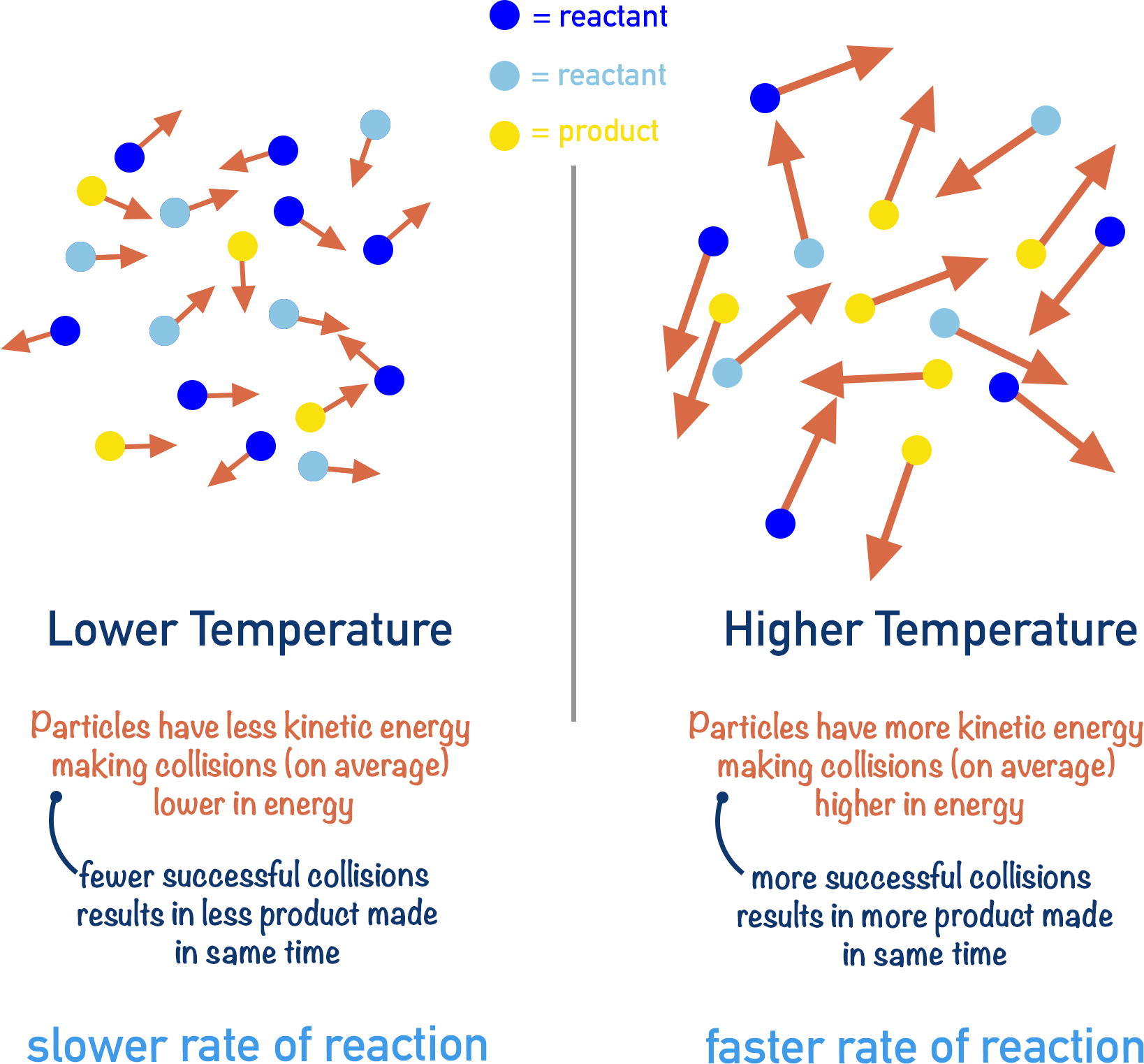
It is important to note that at both 25oC and 75oC successful collisions are occurring. It is just that at 75oC more successful collisions occur in a given amount of time, so the reaction is happening faster.
Effect of Concentration on Reaction Rate:
Increasing the reaction rate means we have to increase the frequency of successful collisions. If we increase the TOTAL number of collisions that are occurring in a given amount of time, we are also increasing the number of SUCCESSFUL collisions that are occurring.
If the concentration of colliding particles is increased, there are more particles in the same amount of space. This means there will be more collisions – if you increase the number of people moving around in a room, there will be more collisions because there are more people to collide with.
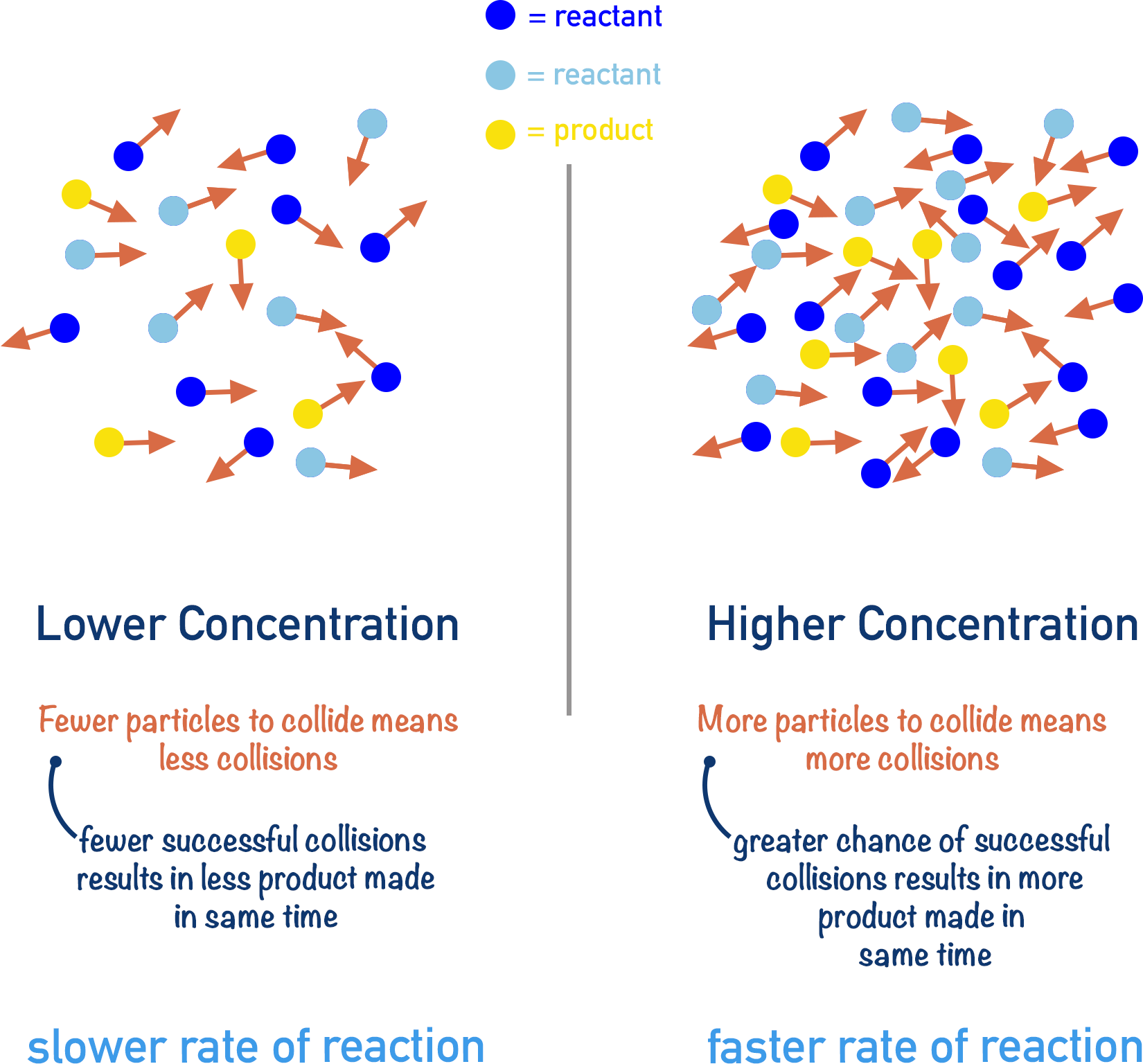
By increasing the concentration of reacting particles, the rate of a reaction is increased. The reacting particles are not given more energy, but there is simply an increase in the chances of a successful collision.
It is important to note that the proportion of successful collisions compared to non-successful collisions will be the same for both higher and lower concentrations. For example, if the concentration of reactants in a reaction is 1 mol dm-3 and 25% of collisions are successful in the first minute, with a 2 mol dm-3 concentration it will be still be 25% of collisions that are successful in the first minute. The only difference is that there are more collisions overall.
Effect of Pressure on a Reaction Rate:
In a gaseous system, increasing the pressure acting on the reactants effectively forces them closer together. This gives the same effect on the rate of reaction as increasing the concentration of reactants.
Remember – concentration is just a measure of how much of substance you have in a given volume.
By forcing particles closer together, there will be more particles in the same volume as before the pressure was increased. The number of particles in that volume increases.
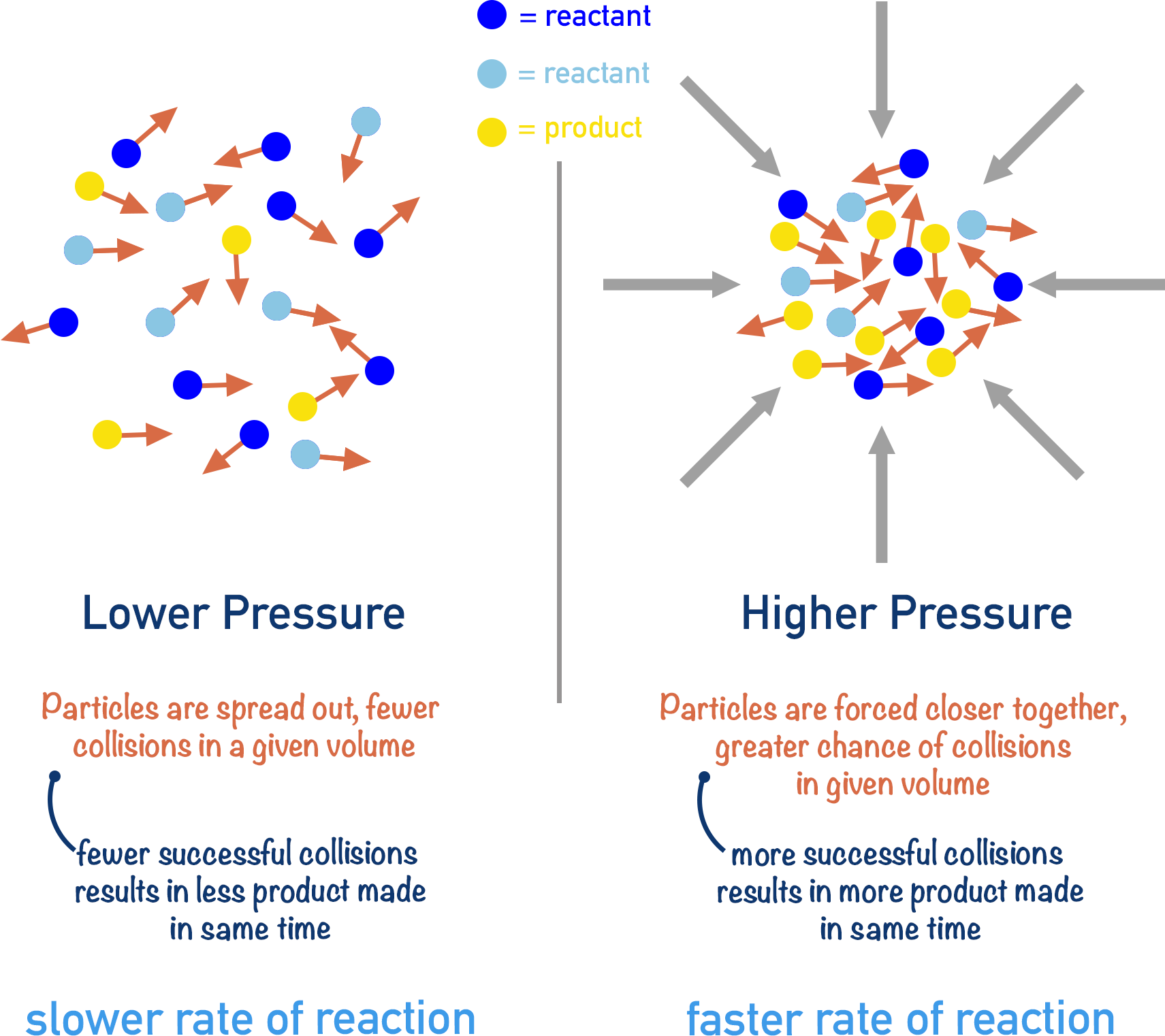
A higher number of particles colliding gives a greater chance for successful collisions. Increased frequency of successful collisions increases the rate of reaction.
Effect of a Catalyst on a Reaction Rate:
The activation energy of a particular reaction route is fixed – it doesn't change. If we want particles with lower energies to successfully collide, the only thing we can do is to find another ‘way’ for the reaction to happen. A different way for a reaction to happen is called an ‘alternative pathway’.
Rather like going for a hike – if you don’t want to climb over a mountain to get to the other side then you need to find another way, as you cannot change the mountain.
A catalyst is a species that can provide an alternative pathway for a reaction to happen.
Catalyst – species that increases the rate of a reaction by providing an alternative reaction pathway.
To use the above example, imagine the catalyst as being a buggy that can drive you around the mountain – getting you to the same destination but saving you a lot of energy. Just like the buggy can be used again to help more people around the mountain, a catalyst remains unchanged at the end of a reaction.
Note, catalysts can actually change during the reaction but they reform at the end, so they are not considered a reactant. They are not ‘used-up’ in the reaction.
By providing a lower energy alternative pathway, the catalyst lowers the activation energy required for a reaction to occur. This means particles’ collisions that happen at a lower energy now have sufficient energy to react - there are more successful collisions.
A bit like people who are less fit are still able to get around the mountain if they use a buggy.
A catalyst does not alter the energy of the particles colliding, it simply enables collisions that happen at lower energy to become successful. These collisions were still occurring before a catalyst was used, but where before they were not successful, now they are!
The rate of reaction is how many successful collisions are happening in a given amount of time. If we lower the activation energy, we increase the number of successful collisions occurring, so a catalyst increases the rate of a reaction.
We’ve launched our new site! 🎉
Course-specific notes with built-in search!
AP • A-Level (AQA • CIE • Edexcel • OCR) • IB • NCERT 11 + 12
over 750+ new pages and 3,500 images.
Visit the new homepage
