Quick Notes Redox
- Most atoms can become more stable by losing or gaining electrons, forming charged ions.
- If an atom loses electrons it is oxidised.
- If an atom gains electrons it is reduced.
- Reducing agents get oxidised themselves, causing another species to be reduced.
- Oxidising agents get reduced themselves, causing another species to be oxidised.
- Half-equations show how one species is being oxidised or reduced in a redox reaction.
Full Notes Redox
Atoms (apart from the noble gases) can lose and gain electrons in order to have a ‘full’ or more stable configuration of outer shell electrons. An atom that has lost or gained an electron becomes charged and is called an ion.
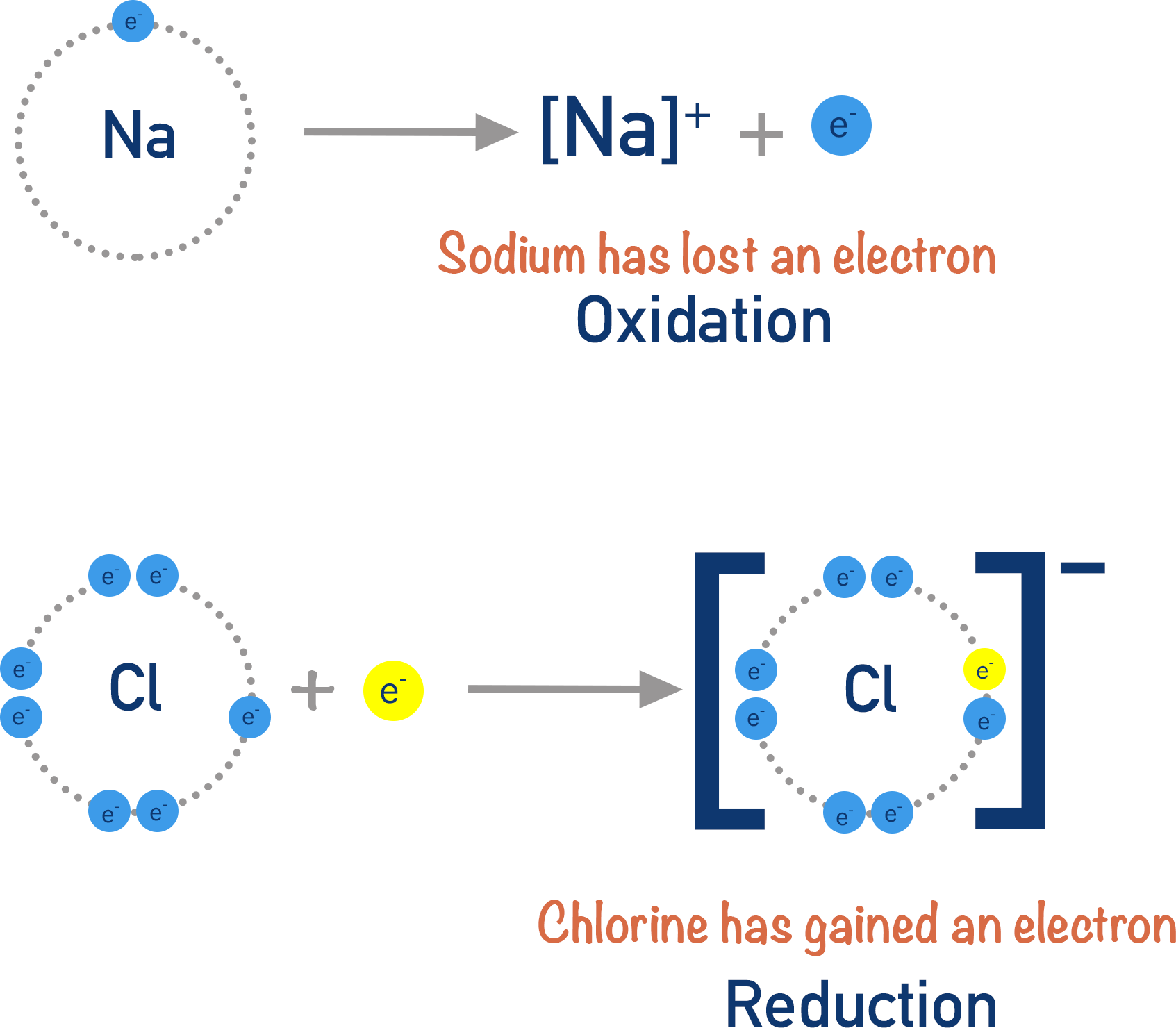
An atom that loses an electron becomes a positively charged ion and has been oxidised. When an atom or species loses electrons, we describe the reaction as ‘oxidation’.
An atom that gains an electron becomes a negatively charged ion and has been reduced. When an atom or species gains electrons, we describe the reaction as ‘reduction’.
Oil Rig is a great way of remembering these names. Oxidation is loss (of electrons) and Reduction is gain (of electrons).
A species that is easily reduced (gains electrons) can act as an oxidising agent and oxidise another species.
A species that is easily oxidised (loses electrons) can act as a reducing agent and reduce another species.
Half-equations show how an individual ion reacts or is produced in terms of electron transfer.
For example, as for the above:
Na → Na+ + e- (oxidation half-equation)
½Cl2 + e- → Cl- (reduction half-equation)
If we combine the two half-equations together, the complete reaction would be:
Na + ½Cl2 + e- → Na+ + e- + Cl-
Here, the e- on both sides cancel out, leaving
Na + ½Cl2 → Na+ + Cl-
It is important to remember that in reality these electrons are being lost or gained by another ion or atom – half-equations are only half (hence the name!) the story with redox reactions. For one species to be oxidised, another species will be reduced.
