Quick Notes Acid Anhydrides
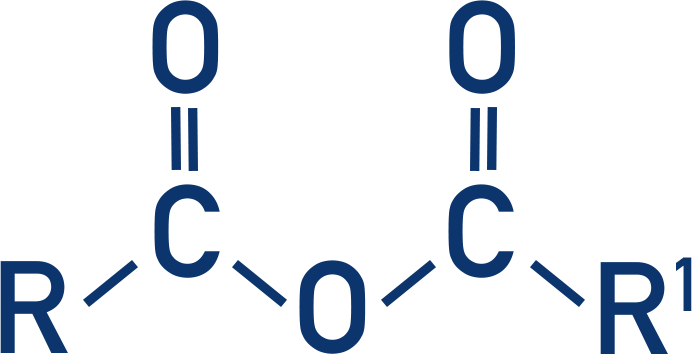
- Acid anhydrides are made from two carboxylic acids joined together in a condensation reaction.
- Acid anhydrides react in similar ways to acyl chlorides, but they are less reactive, making them safer to use.
- Acid Anhydride + Alcohol → Ester + Carboxylic Acid
Full Notes Acid Anhydrides
Acid anhydrides are made from two carboxylic acid groups joined together.

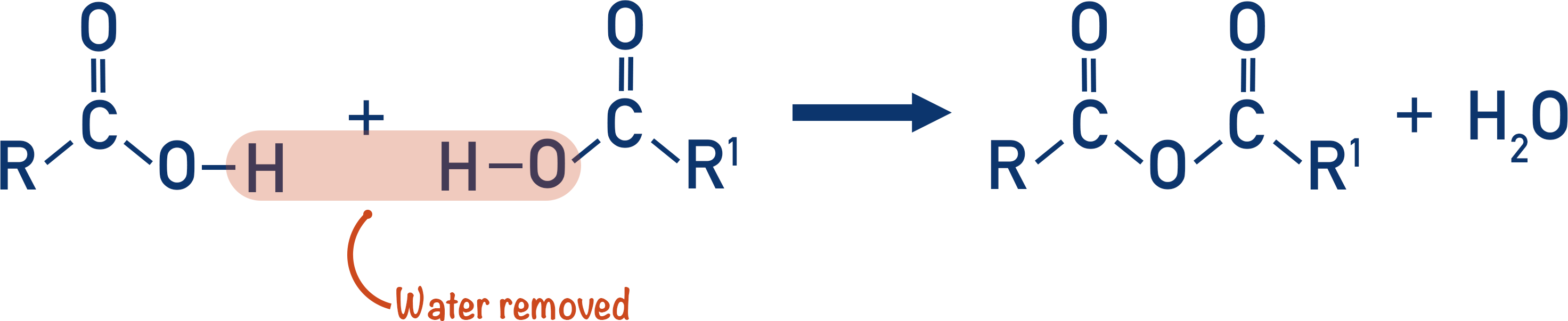
To form an acid anhydride, a water molecule is lost in a condensation reaction between two carboxylic acids. This is why acid anhydrides are called anhydride – lack of water.
Reactions of Acid Anhydrides
Acid anhydrides react in a similar way to acyl chlorides, but they are less reactive. Being less reactive is actually very useful because they are far safer (and cheaper) to use in reactions.

Reacting an acid anhydride with an alcohol produces an ester and a carboxylic acid. This is essentially the same reaction as with an acyl chloride (see Acyl Chlorides), but rather than releasing HCl, a carboxylic acid is formed.
