Quick Notes Carbonyls - Nucleophilic Addition
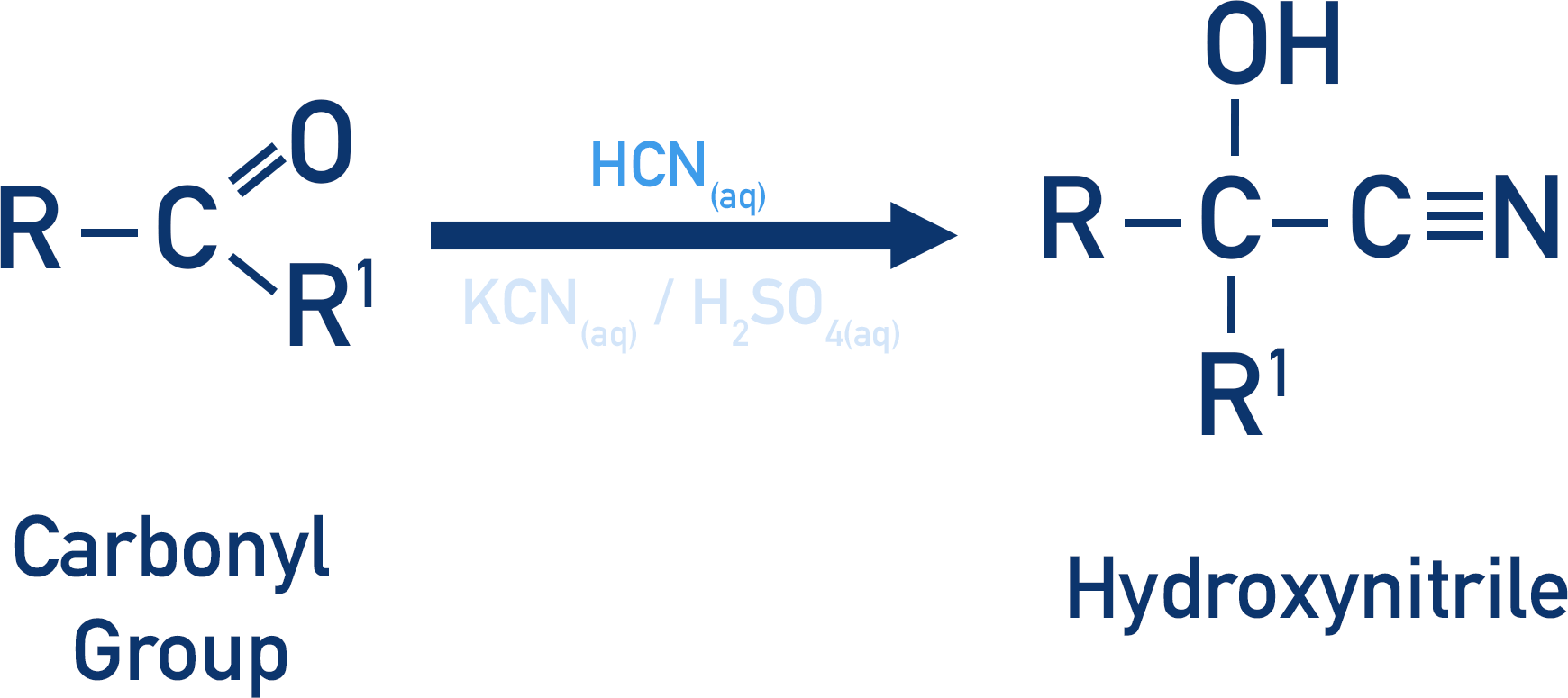
- Hydrogen cyanide reacts with carbonyl groups in nucleophilic addition reactions to form hydroxynitriles.
- The length of the carbon chain in a carbonyl molecule can be extended using this reaction.
- Hydrogen cyanide is highly reactive (and toxic); it can be prepared ‘in situ’ using a metal cyanide (KCN or NaCN) and dilute sulfuric acid (H2SO4).
Full Notes Carbonyls - Nucleophilic Addition
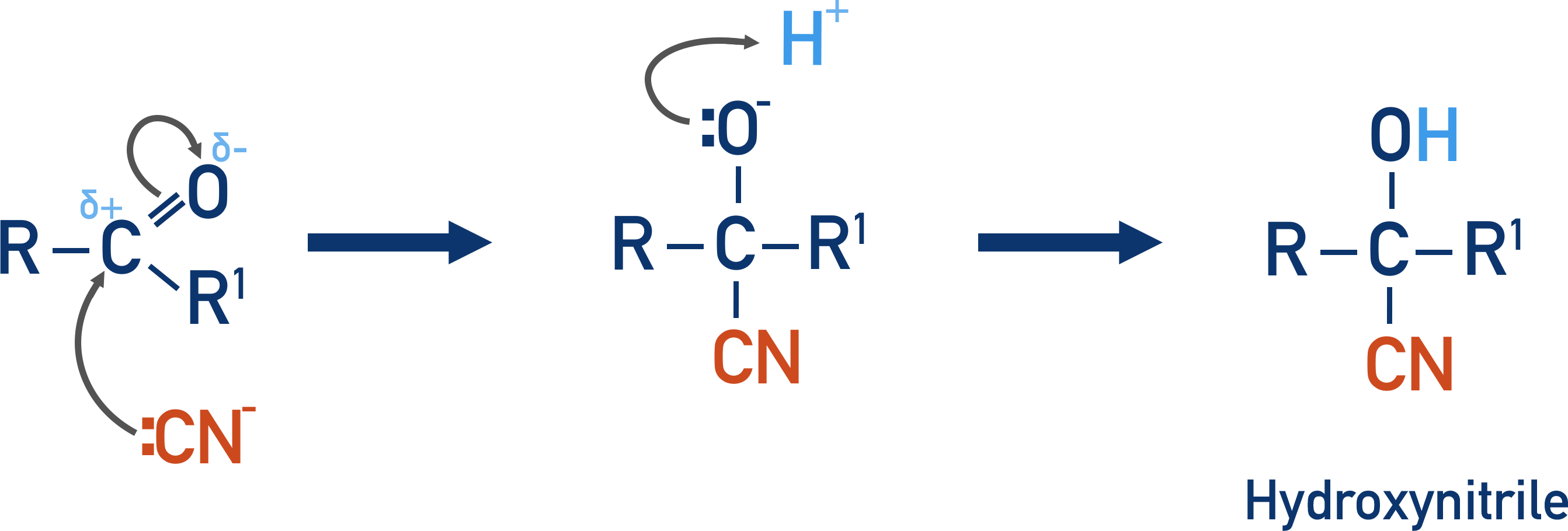
When hydrogen cyanide is added to a carbonyl group, nucleophilic addition occurs.
Hydrogen cyanide is a highly toxic, dangerous substance. So, it’s often produced in solution with the carbonyl compound that it’s going to react with. A metal cyanide ion (usually NaCN or KCN) is added with dilute sulfuric acid. The metal cyanide dissociates in solution releasing CN- ions; the dilute sulfuric acid releases H+ ions.
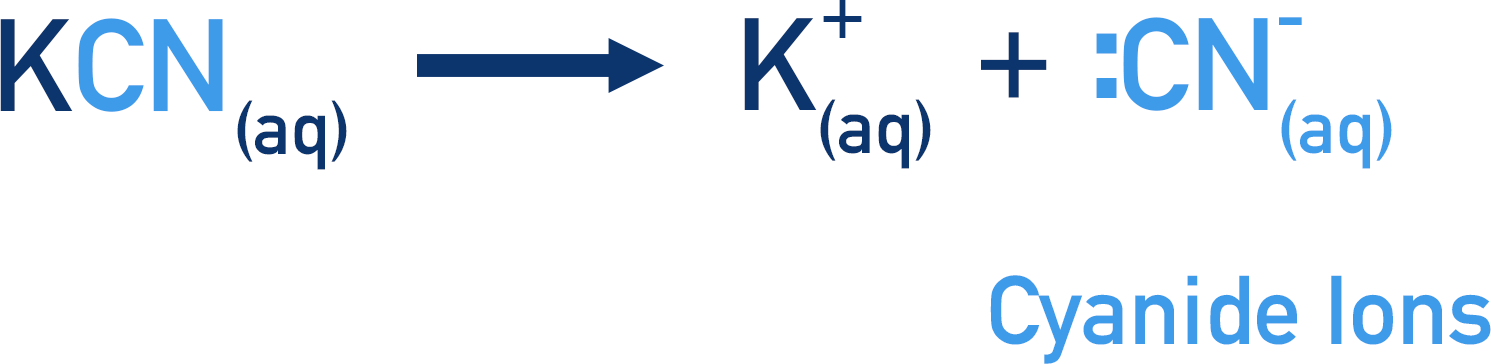
Cyanide ions (CN-) are strong nucleophiles that are attracted to the carbon with a partial positive charge in a carbonyl.
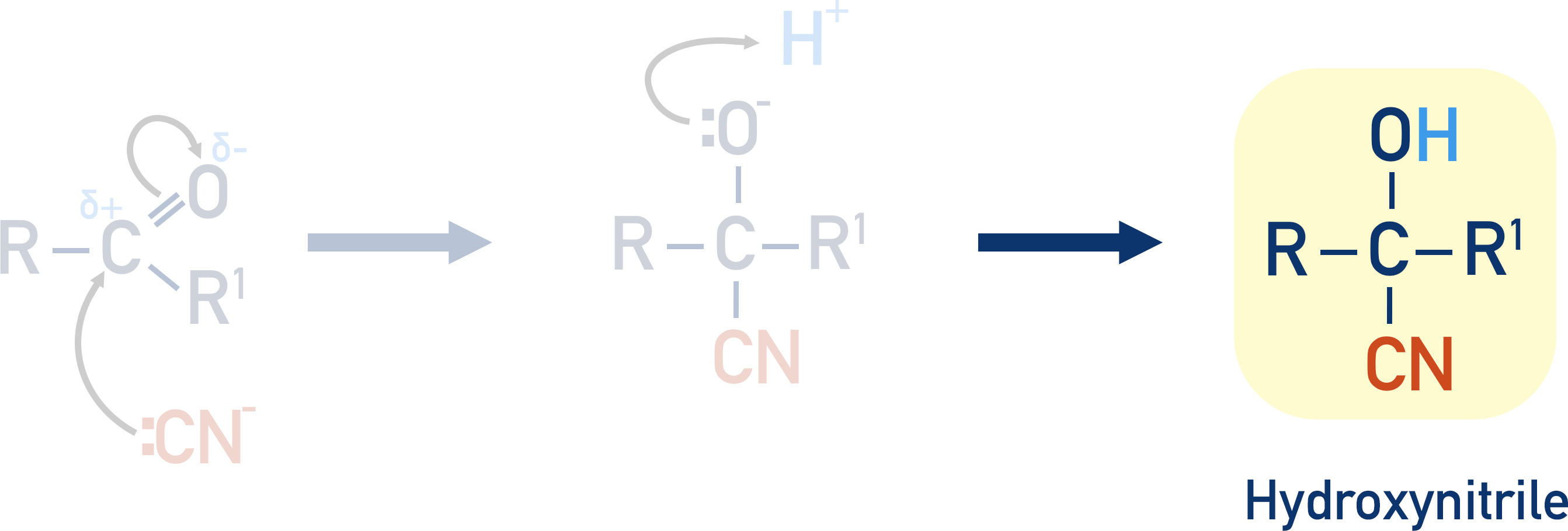
Mechanism
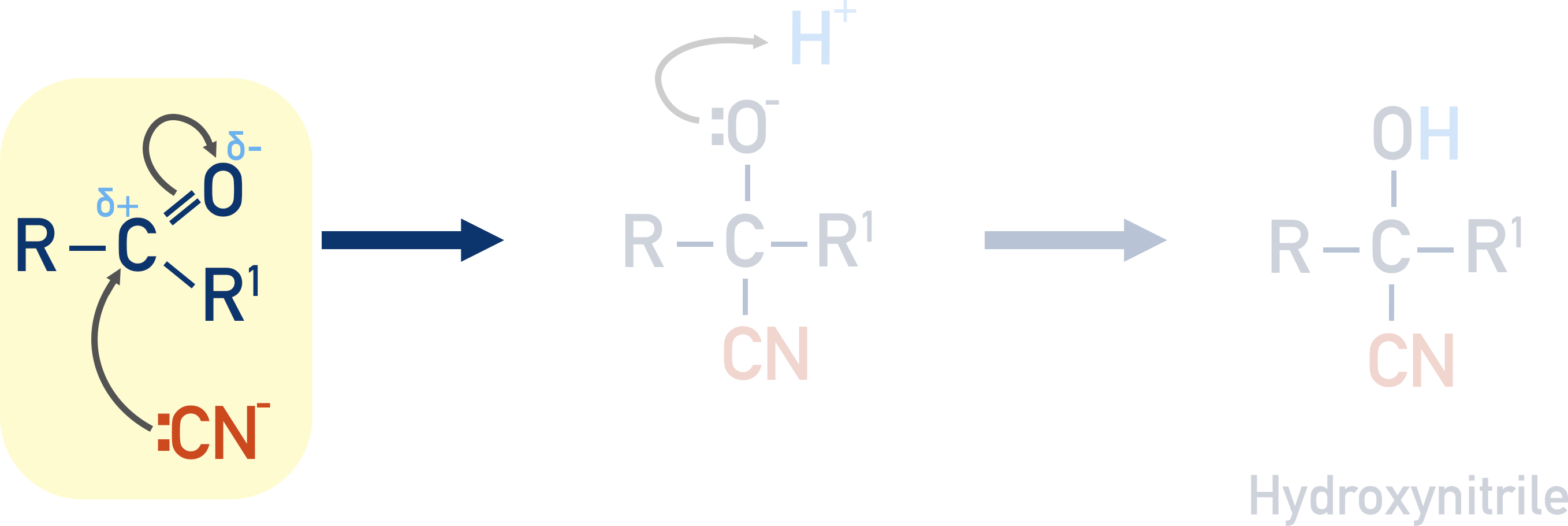
The lone pair of electrons on the carbon in the nitrile move to the partially positive carbon in the carbonyl. The C=O bond breaks, with the oxygen atom gaining a pair of electrons and a negative charge.
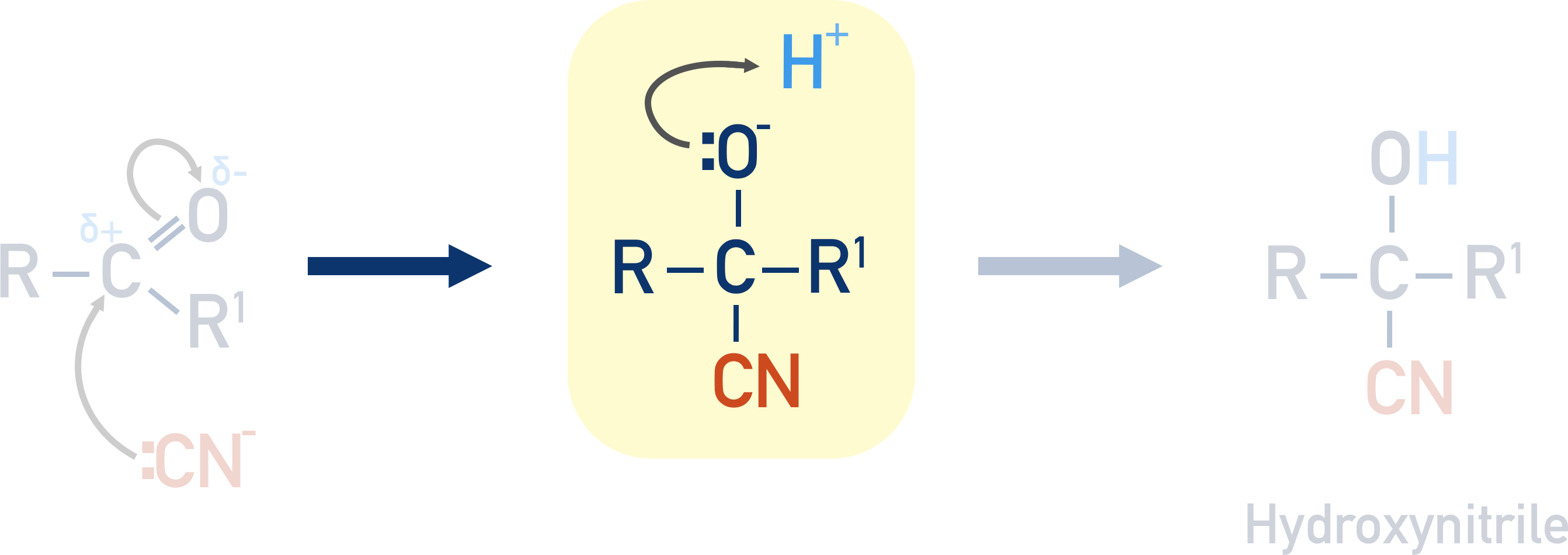
A H+ ion in solution (from the sulfuric acid) then bonds with the negatively charged oxygen atom, forming an –OH group.