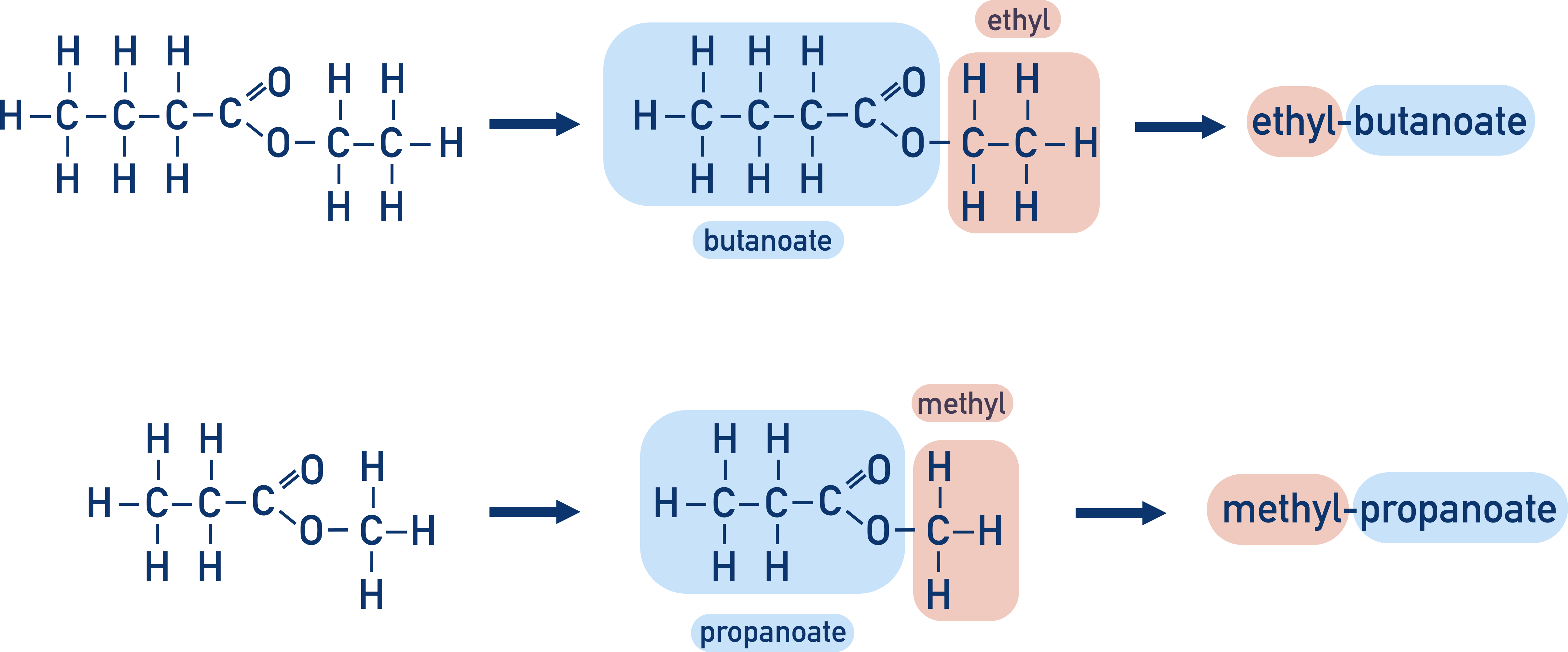
Quick Notes Naming Esters
- Esters are named based on the length of the carbon chains in both the carboxylic acid and alcohol that are used to make the ester.
- The general form is alkyl-carboxylate.

- The first part of the name comes from the carbon chain in the alcohol (-alkyl).
- The second part of the name is the same as the carboxylate ion of the carboxylic acid (-carboxylate).
Full Notes Naming Esters
The name given to an ester is based upon the carboxylic acid and alcohol that reacted together to produce the ester.
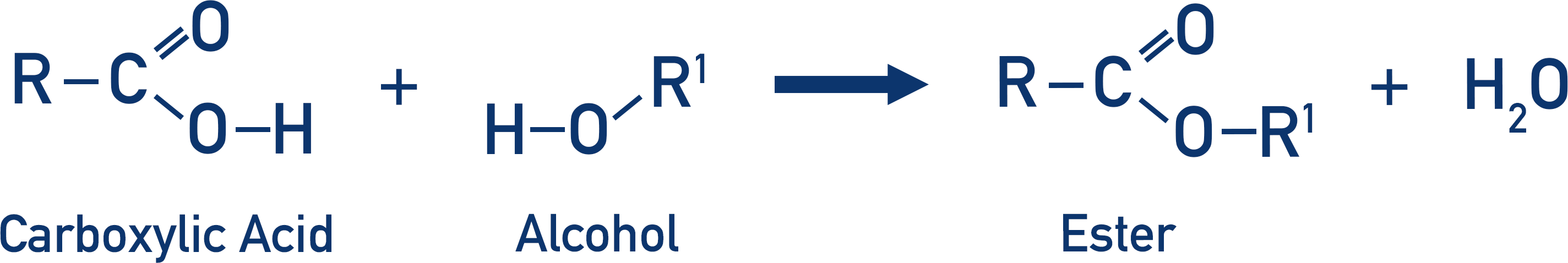
The carboxylic acid takes the name of the carboxylate (for example, ethanoic acid becomes ethanoate).
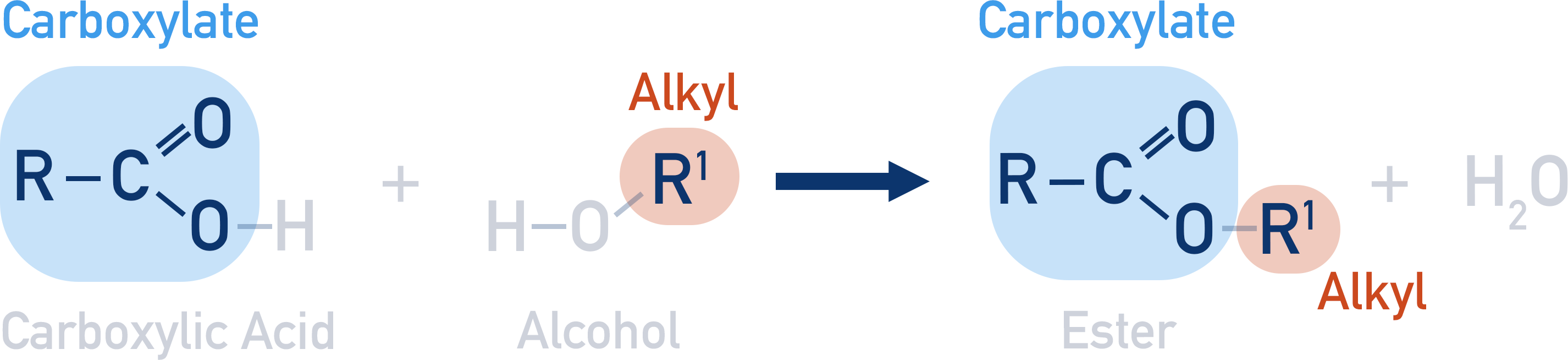
Please note: in the actual mechanism for esterification, the oxygen from the alcohol does not actually get lost to form water, it is the oxygen from the hydroxyl group in the carboxylic acid that forms the water lost. For A-level students, I show it this way to help with naming esters.

The alcohol group becomes an alkyl group (for example, methanol becomes methyl).
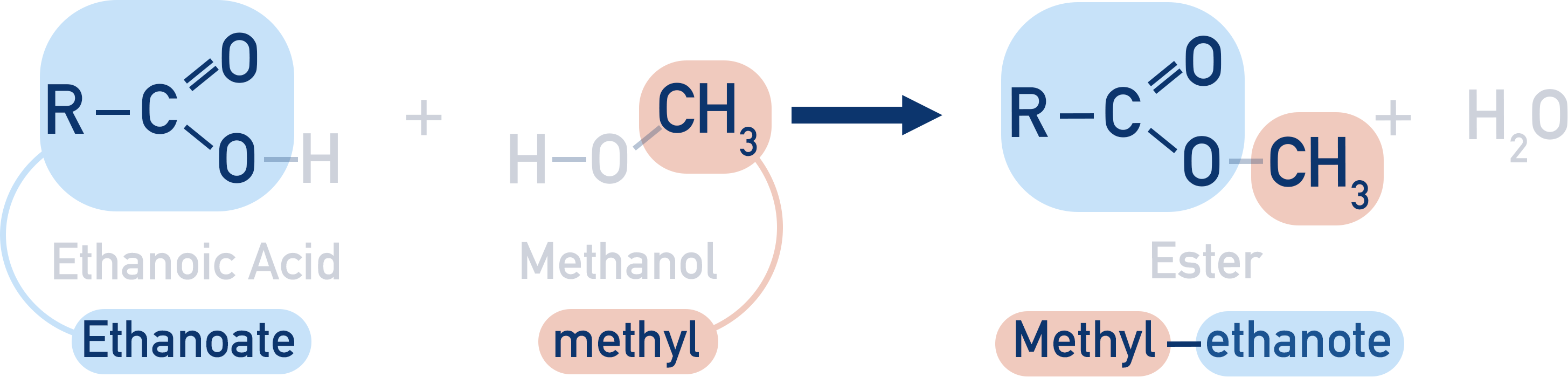
In simple terms: the alcohol part goes first as an alkyl, the carboxylic acid part goes second as an –oate.