Video Tutorial The Mole
Quick Notes The Mole
- Atoms and particles are very small, so referring to actual number of atoms in a sample isn’t very practical.
- The mole is a unit used to refer to an amount of atoms or particles.
- Just like one dozen = 12 of something, one mole = 6.022 x 1023 of something.
- The number one mole refers to is known as Avogadro’s constant.
Full Notes The Mole
In chemistry there are two real ways of measuring how much matter (‘something’) we have – mass and volume.
Atoms are very small. This means, even for a small sample, a substance will have a lot of atoms in it. So many in fact, it is not realistic to refer to the whole number of atoms or particles in the sample. Instead, we use a unit called ‘the mole’. A mole is just a way of saying how many particles of something you have.
Just like 1 dozen = 12 of something; 1 mole = 6.02 x 1023 of something.
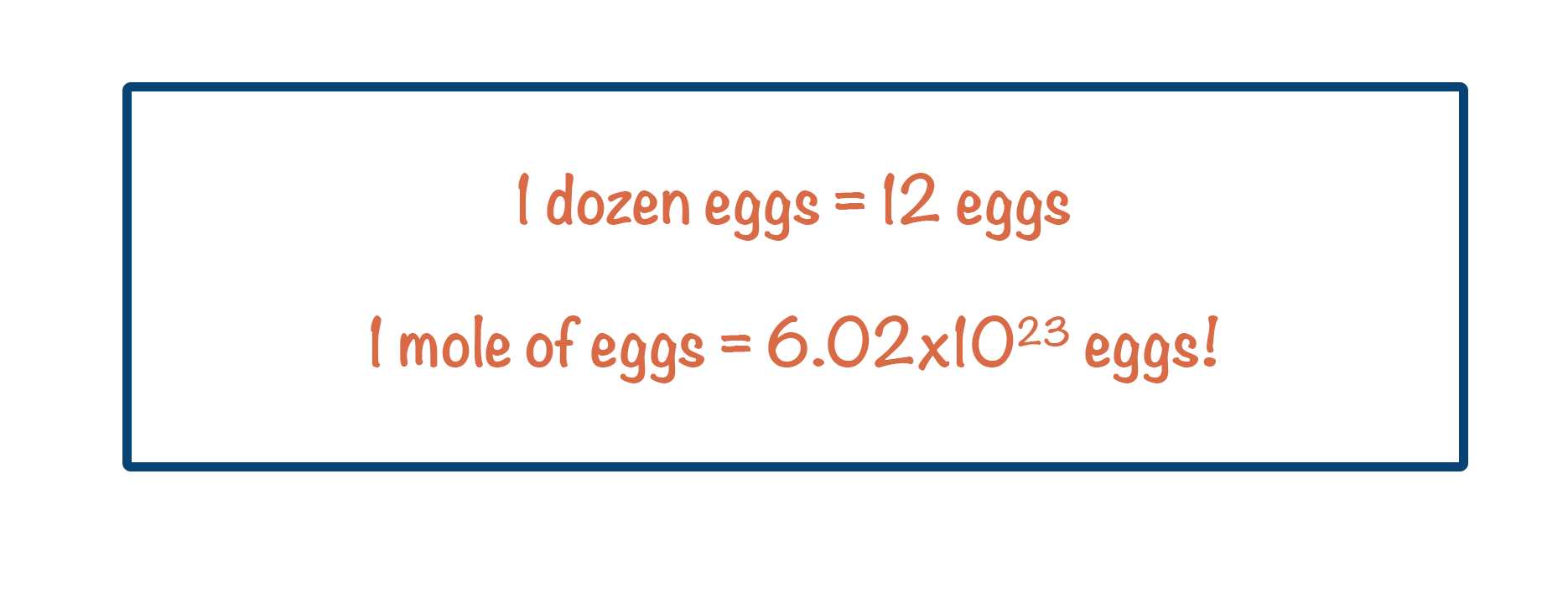
If you have two dozen atoms, you know that you have twenty-four atoms (2 x 12 = 24). If you have two moles worth of atoms in a sample, it simply means you have 1.25 x 1024 atoms of that sample (2 x 6.02 x 1023).

Again - always remember – a mole is just a simple way of saying how many particles of something you have.
A bit extra: Where does ‘the mole’ come from?
Avagadro’s constant was not actually discovered by the scientist Avagadro!
Avagadro (an Italian scientist who lived in the 1800s) proposed the idea that the volume of space occupied by a particular number of particles of a gas is the same as the volume of space occupied by the same number of particles of a different gas (at the same temperature and pressure).
It was many years later that scientists started to link the relative masses of atoms and Avagadro’s idea.
The link showed that the number of atoms in a particular sample of an element would be the same as the number of atoms in a sample of a different element – if the masses of both samples were in the same ratio as their relative masses. This is a lot simpler than it sounds!
For example, in 1g of hydrogen there would be the same number of atoms as there would be in 12g of carbon. This is because the relative mass of hydrogen is 1 compared to the relative mass of carbon (12).
When the mass of an element (in grams) is the same value as its relative mass (for example, 1 for hydrogen, 12 for carbon), scientists found that the actual number of atoms present is 6.022141 x 1023. This value was named ‘Avagadro’s constant’ in honour of Avagadro.
We’ve launched our new site! 🎉
Course-specific notes with built-in search!
AP • A-Level (AQA • CIE • Edexcel • OCR) • IB • NCERT 11 + 12
over 750+ new pages and 3,500 images.
Visit the new homepage
