Quick Notes Alkenes
- Alkenes are hydrocarbons in which two or more of the carbon atoms are bonded together with a double bond.
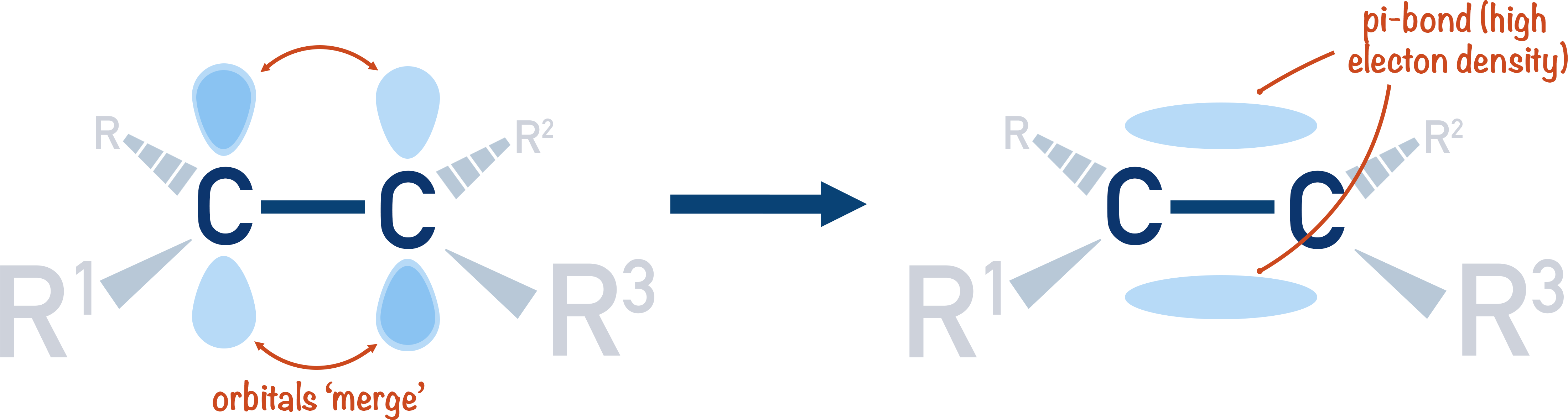
- A carbon double bond is made by the merging of a 2p-orbital from each carbon atom, creating an area of high electron density between the two atoms, called a ‘pi-bond’.
- Electron deficient species (electrophiles) are attracted to the electrons in the double bond, which makes alkenes more reactive than simple alkanes.
Full Notes Alkenes
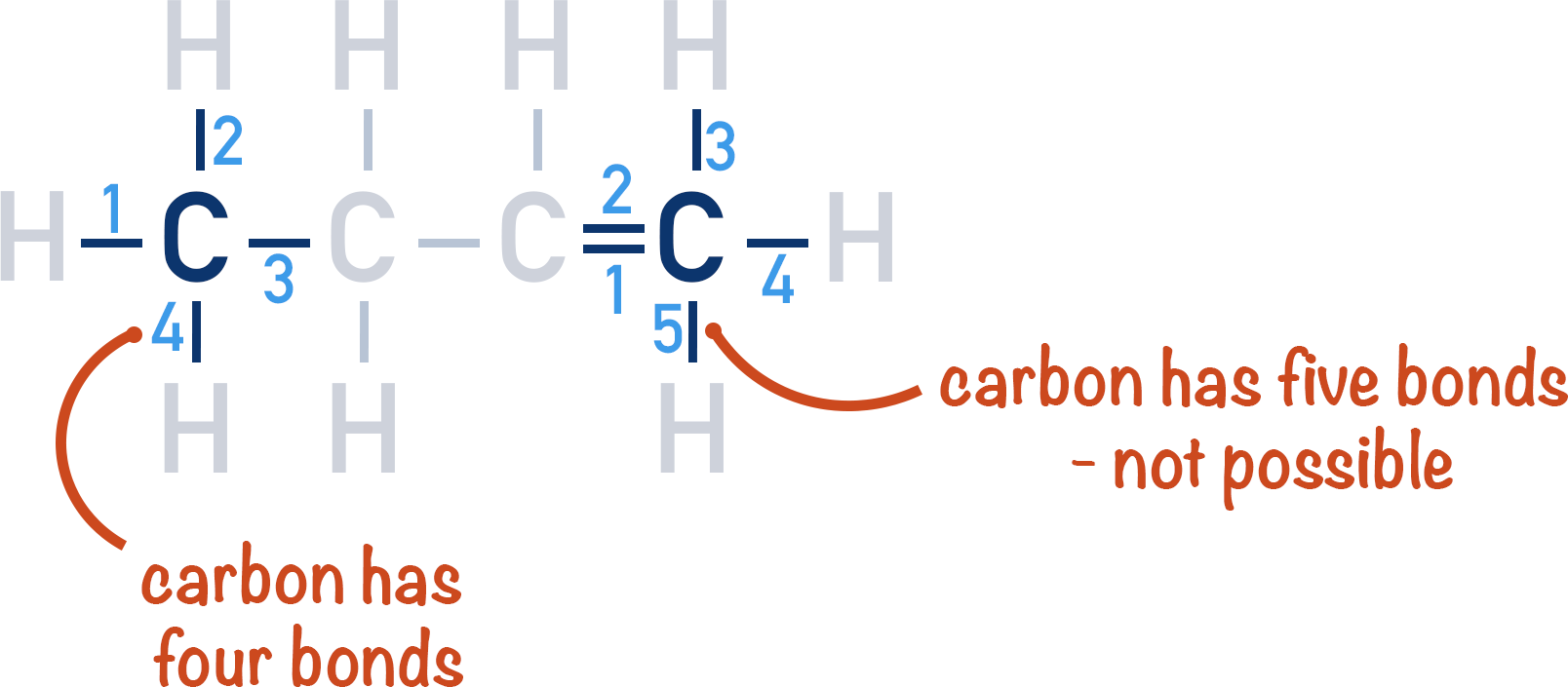
Carbon can form double bonds between atoms of itself. A carbon chain can still exist, as carbon atoms are still bonded together, but the number of hydrogen atoms bonded to each carbon will change. A carbon atom can only form four covalent bonds. A double bond counts as two of those four, so only two bonds are left for each carbon to make with other atoms.
This gives alkenes with only one double bond the general formula CnH2n (where n is the number of carbon atoms in the molecule).
Hydrocarbons that have one or more double bond(s) within their carbon chain are called alkenes. Alkenes are more reactive than alkanes, so they can be very useful when making new molecules in organic chemistry.
The Double Bond
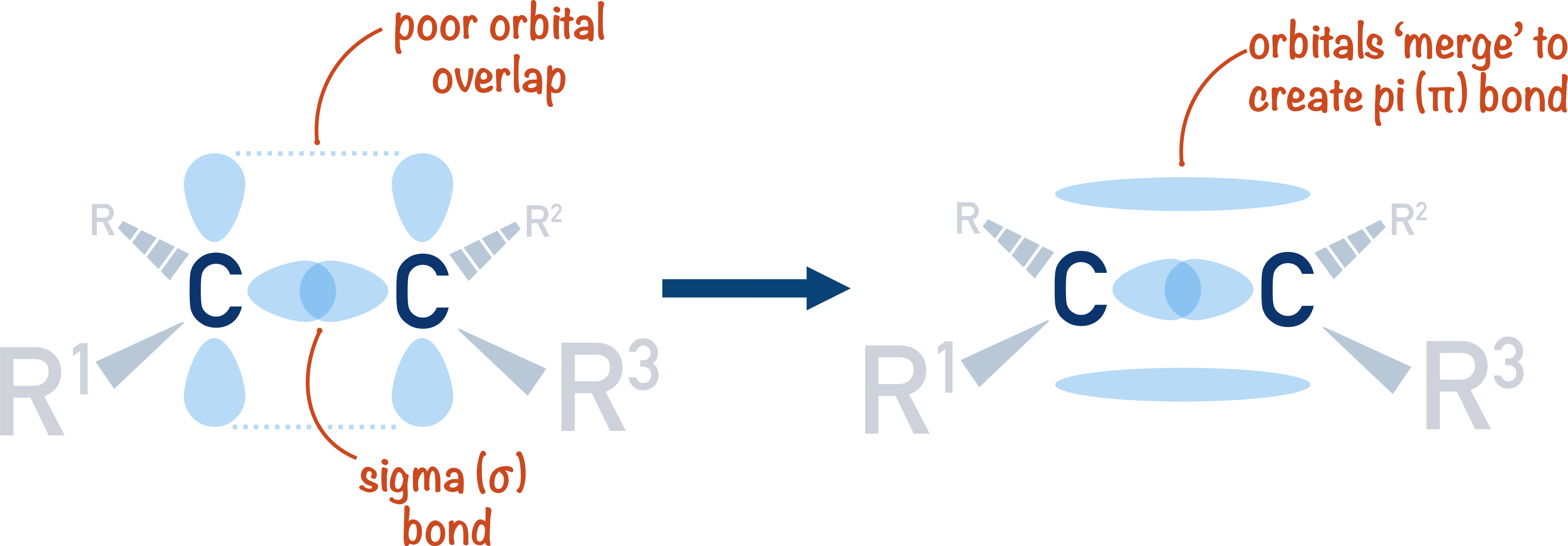
A covalent bond is the sharing of a pair of electrons between two atoms. In order for a bond to form, electron orbitals have to overlap. As there are different shapes of electron orbitals, there are sometimes different ways orbitals can overlap between atoms.
How ‘well’ two orbitals overlap can indicate how strong a particular bond is likely to be. Poor overlap results in a weak bond, good overlapping results in a stronger bond. ‘Perfect’ overlapping creates a single bond called a sigma bond. Sigma bonds are represented with the symbol 'σ'.
When bonding orbitals are unable to overlap fully, they can sometimes 'adust' themselves to try and get as close to each other as possible and 'merge'. When this happens, a pi-bond (double bond!) is formed. Pi bonds are represented by the symbol 'π'.
As orbital overlap is poor in a pi-bond, the bond is much weaker than a sigma bond and is easier to break.
Carbon single bonds are sigma bonds due to good orbital overlap between electron orbitals from the bonding carbon atoms – this is what makes them so strong.
In a carbon-carbon double bond, a 2p-orbital from each carbon atom cannot fully overlap, so they have to ‘merge’. This causes two regions of electron density to form – above and below the sigma single bond between the carbon atoms.
Electrons are negatively charged particles, so wherever electrons exist there is a region of negative charge. In a carbon-carbon double bond the merged 2p-orbitals contain electrons, making them highly vulnerable to ‘attack’ from positive ions and electron deficient species (electrophiles). This is why carbon-carbon double bonds are, relatively, easily broken.