Video Tutorial Alkenes Electrophilic Addition
Quick Notes Alkenes - Electrophilic Addition Reactions
- Alkenes react by electrophilic addition reactions.
- Electron deficient species (electrophiles) are attracted to the pi-bonded electrons in a carbon-carbon double bond.
- In electrophilic addition, an electrophile causes the carbon-carbon double bond to break, and a new bond is formed between the electrophile and one of the carbon atoms.
- A carbocation (positively charged carbon atom) intermediate is formed that a negatively charged species forms a bond with.
- Primary carbocations are less stable than secondary and tertiary carbocations because they experience less of an inductive effect, meaning they aren’t as likely to form during electrophilic addition.
- Major and minor products of electrophilic addition reactions are determined by the stability of the intermediate carbocation that forms.
Full Notes Alkenes - Electrophilic Addition Reactions
The high electron density within a carbon-carbon double bond makes it vulnerable to ‘attack’ from an electron deficient species. Electron deficient species are called electrophiles; they are attracted to the electrons within the carbon-carbon double bond and can accept a pair of electrons.
The bonding pair of electrons from the double (pi, π-bond) bond in the alkene can leave the double bond and form a new covalent bond between the electrophile and one of the carbon atoms. The single (sigma, σ-bond) bond between the carbon atoms is left untouched.
A carbon atom can form four covalent bonds. The carbon atom the electrophile is bonded to now has four bonds, but the other carbon atom from the double bond only has three. This carbon atom (with three bonds) carries a positive charge because one of its electrons was taken from the pi-bond (to form a new bond between the electrophile and the other carbon atom).
A positively charged carbon atom in a molecule is called a carbocation and is highly reactive.
Because we are ‘opening’ a double bond and adding an electrophile to the original alkene, addition is taking place. The reaction type is electrophilic addition.
Addition of Bromine
When bromine comes into contact with a carbon-carbon double bond, electrophilic addition takes place. This is the basis of the ‘bromine water test’ for an alkene. If an alkene is present, bromine (Br2) is removed from the bromine water, decolourising the bromine water.
Remember, to start an addition reaction with a carbon-carbon double bond, an electrophile is required. Bromine (Br2) is a diatomic molecule and is non-polar, so it’s not an electrophile. The bromine can only act as an electrophile when it’s arranged in a certain way with the double bond.
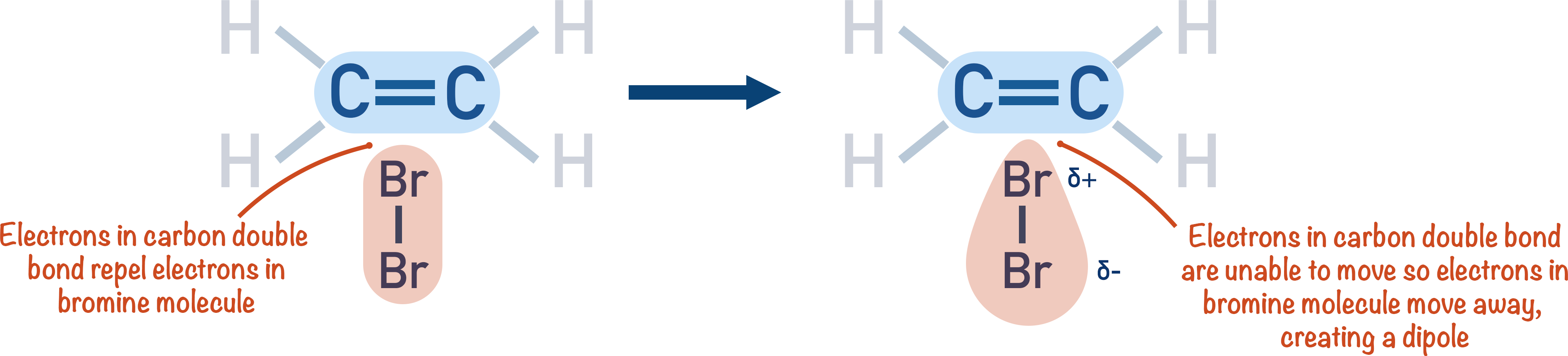
The high electron density of the carbon-carbon double bond induces a dipole in the bromine molecule. The electrons in the bromine molecule are repelled by the electrons in the double bond, resulting in the bromine atom nearest the double bond exhibiting a partially positive charge. It’s this partially positive charge that enables the bromine molecule to act as an electrophile.
Electrons from the carbon-carbon double bond move to the partially positive bromine atom and form a bond. Electrons from the Br-Br bond move to the partially negative bromine atom, leaving a bromide ion with a lone pair of electrons.
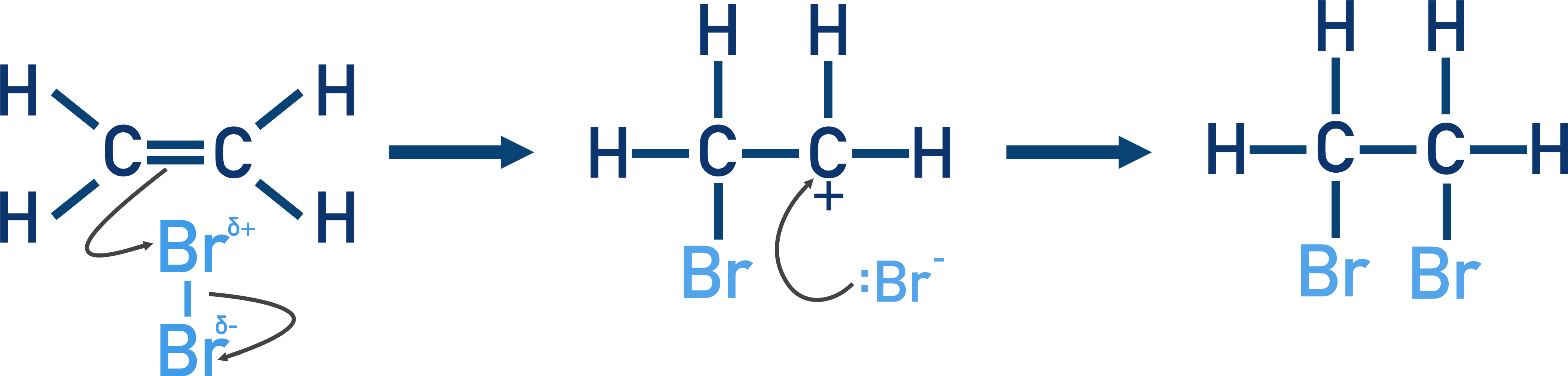
The carbon atom with three bonds now has a positive charge (carbocation). The lone pair of electrons from bromide ion we formed above then forms a bond with this carbocation.
Addition of Hydrogen Bromide
Sometimes, when addition occurs to alkenes, there are two possible products that can be formed.
For example, when hydrogen bromide (HBr) and but-1-ene undergo an addition reaction, 1-bromobutane and 2-bromobutane can be formed.
The two products are not formed in equal amounts. More 2-bromobutane is formed (making it a major product) than 1-bromobutane (minor product).
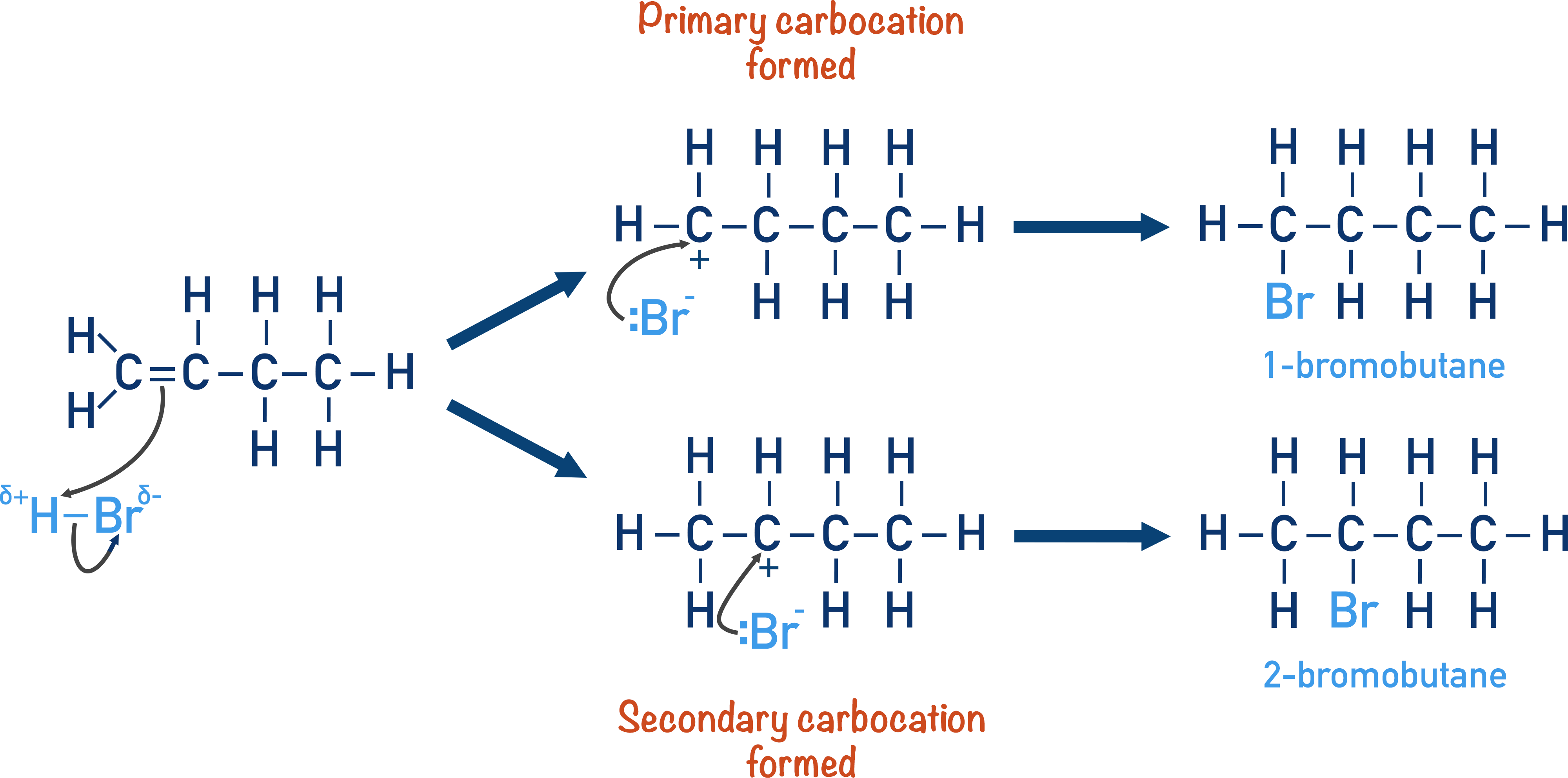
We can see the reason for this formation of a major and a minor product from the reaction mechanism. In particular, the stabilities of the carbocations formed.
The H-Br bond is polar, so the partially positive hydrogen atom acts as an electrophile – with a bonding pair of electrons from the carbon-carbon double bond moving to form a new bond with the hydrogen atom. The hydrogen atom can bond to either one of the carbon atoms in the carbon-carbon double bond.
This now leaves the other carbon with a positive charge (carbocation). A carbocation is highly reactive and unstable. In the above, there are two possible carbocations that can be formed, and they both have different stabilities.
Carbocation Stability
Alkyl groups have a slightly electron donating effect, in this case that means they can ‘feed’ electron density towards the carbocation, effectively diluting its positive charge and making it more stable. This is called a positive inductive effect and can be used to predict the stability of a particular carbocation intermediate. The greater the inductive effect experienced by a carbocation, the more stable it is.
Carbocations can fall into three categories: primary, secondary and tertiary.
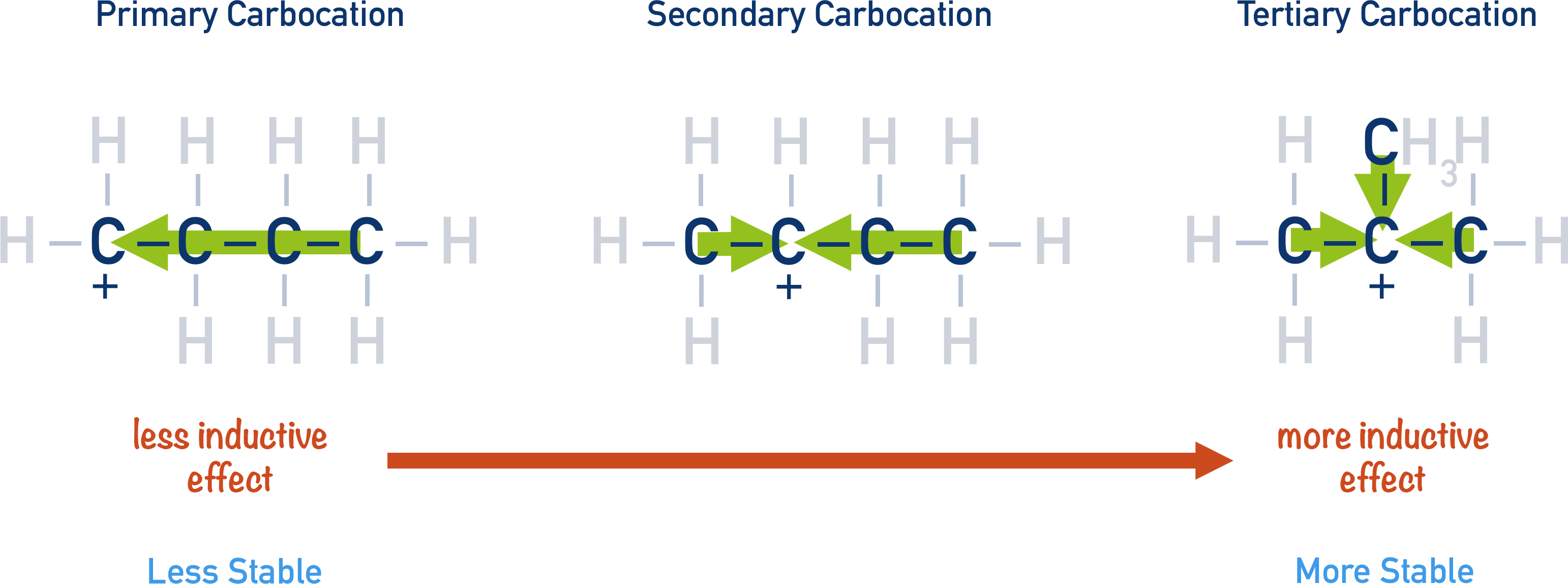
In a primary carbocation, there is only one alkyl group that can ‘feed’ electron density towards the carbocation. It is unable to stabilise the positive charge as much as a secondary carbocation, which has two alkyl groups that can ‘feed’ electron density. In nature, stability wins!
If two intermediates are possible in a reaction, the most stable one (lowest energy) will be formed more readily.
Since the bromide ion in the example reaction bonds to the carbocations in each pathway, the position of the carbocation dictates the final position of the bromine in the molecule.
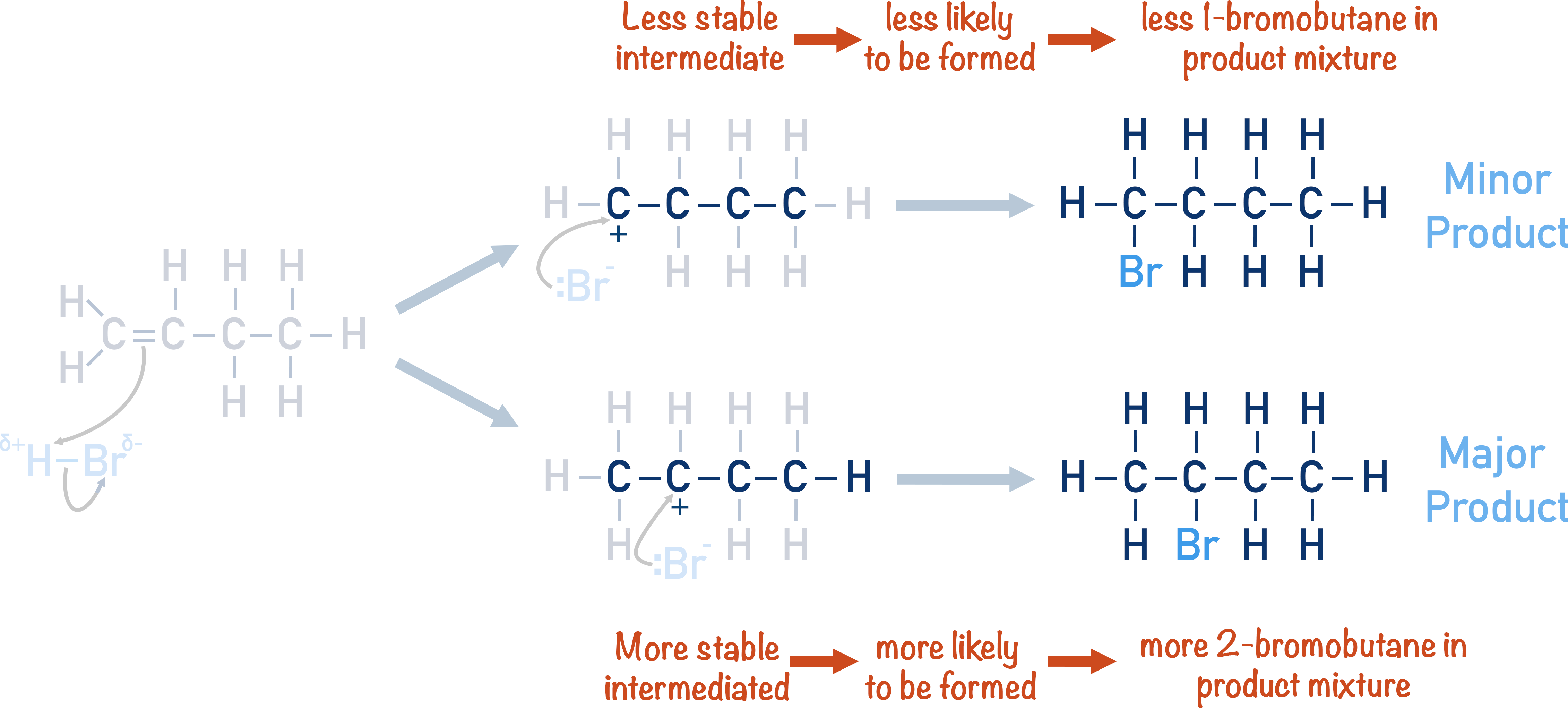
Because the secondary carbocation is more stable than the primary carbocation, it will form more readily. This means more 2-bromobutane is formed than 1-bromo-butane, making 2-bromobutane the major product.
Addition Polymerisation
The carbon-carbon double bond in an alkene enables polymers to be formed. The double bond can be opened up and the carbon atoms are free to bond with other carbon atoms from other molecules – whose double bonds have also been opened up. For more detail, see Polymers .
A polymer is just a very long chain molecule, made up of the same basic unit bonded over and over again.
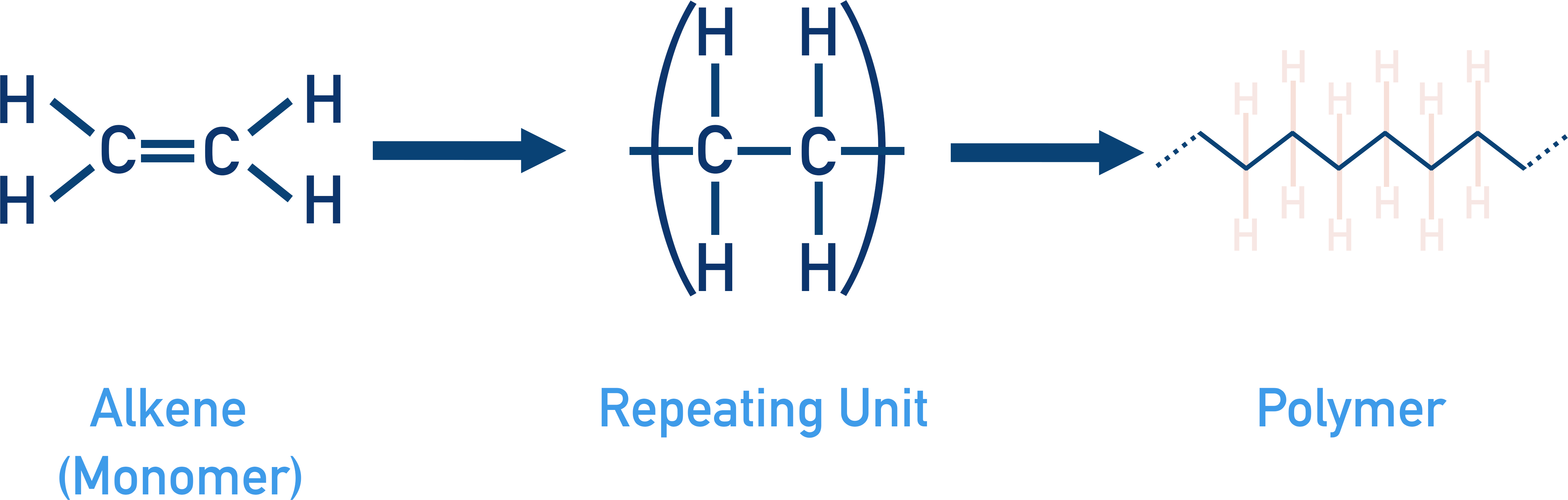
A repeating unit is the basic unit that makes up a polymer. Repeating units are made from small molecules called monomers.
Due to the large size of polymer molecules, they have a high relative molecular mass.
Their large size also means they have large amounts of intermolecular forces between molecules, often making them solids at room temperature. However, their intermolecular forces are usually very weak, so their shapes can sometimes be changed easily, making polymers very useful in manufacturing.
We’ve launched our new site! 🎉
Course-specific notes with built-in search!
AP • A-Level (AQA • CIE • Edexcel • OCR) • IB • NCERT 11 + 12
over 750+ new pages and 3,500 images.
Visit the new homepage
