Video Tutorial Halogenoalkanes - Elimination Reactions
Quick Notes Halogenoalkanes - Elimination Reactions
- Halogenoalkanes can be converted to alkenes in an elimination reaction.
- By reacting halogenoalkanes with hydroxide ions in ethanolic conditions (anhydrous), an alkene and not an alcohol is formed.
- The reaction is carried out under reflux conditions and the hydroxide ion acts as a base (unlike in the hydrolysis of a halogenoalkane to form an alcohol).
Full Notes Halogenoalkanes - Elimination Reactions
Halogenoalkanes can be converted into alkenes by an elimination reaction.
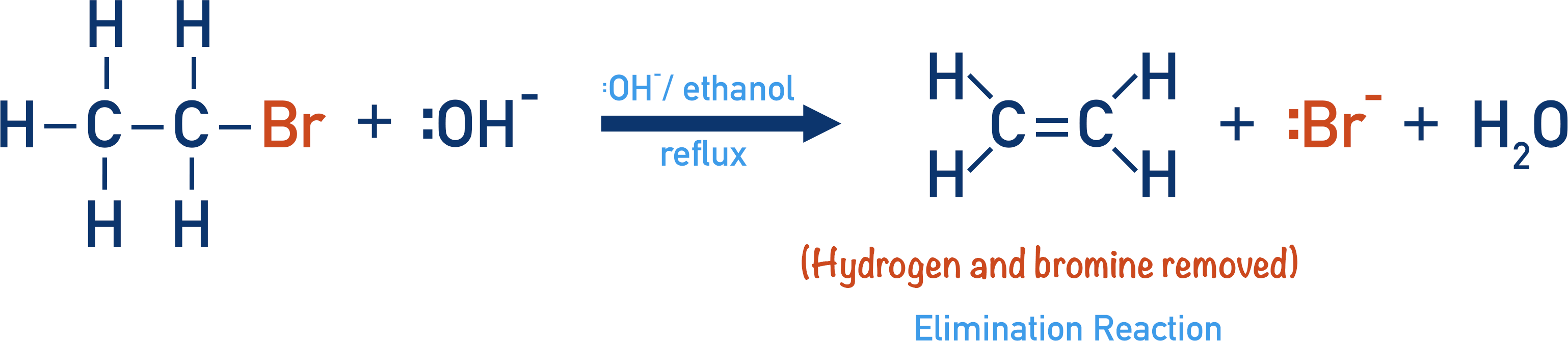
A halogenoalkane is reacted with hydroxide ions under reflux in ethanol. Ethanol is used as a solvent to ensure the conditions are anhydrous (no water!).
The hydroxide ion acts as a base, accepting a proton from a C-H bond to form H2O.The important thing to note is that the OH- ion can act as either a nucleophile or as a base, depending on the conditions it’s reacting in.

We’ve launched our new site! 🎉
Course-specific notes with built-in search!
AP • A-Level (AQA • CIE • Edexcel • OCR) • IB • NCERT 11 + 12
over 750+ new pages and 3,500 images.
Visit the new homepage
