Video Tutorial Halogenoalkanes - Nucleophilic Substitution Reactions
Quick Notes Halogenoalkanes - Nucleophilic Substitution Reactions
- Carbon-halogen bonds are highly polar.
- Due to the high electronegativity of halogens, the carbon becomes partially positively charged and the halogen partially negatively charged.
- Polarity of the carbon-halogen bonds decreases as you go down group 7.
- The carbon in a carbon-halogen bond is easily attacked by electron donating species (nucleophiles) that swap places with the halogen in nucleophilic substitution reactions.
- Key Reactions:
- Halogenoalkanes with sodium hydroxide in aqueous conditions form alcohols.
- Halogenoalkanes with ammonia in ethanolic conditions form amines.
- Halogenoalkanes with cyanide ions in ethanolic conditions form nitriles.
- Ethanolic conditions are needed instead of aqueous conditions, otherwise alcohols would form.
Full Notes Halogenoalkanes - Nucleophilic Substitution Reactions
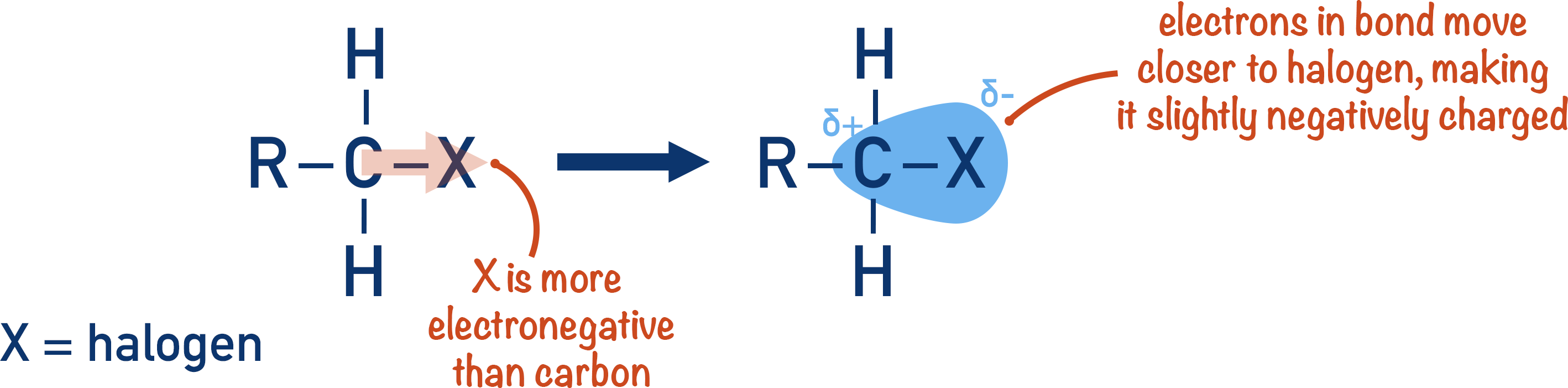
Halogens form highly polar bonds when bonded to carbon. This polarity makes halogenoalkanes reactive.
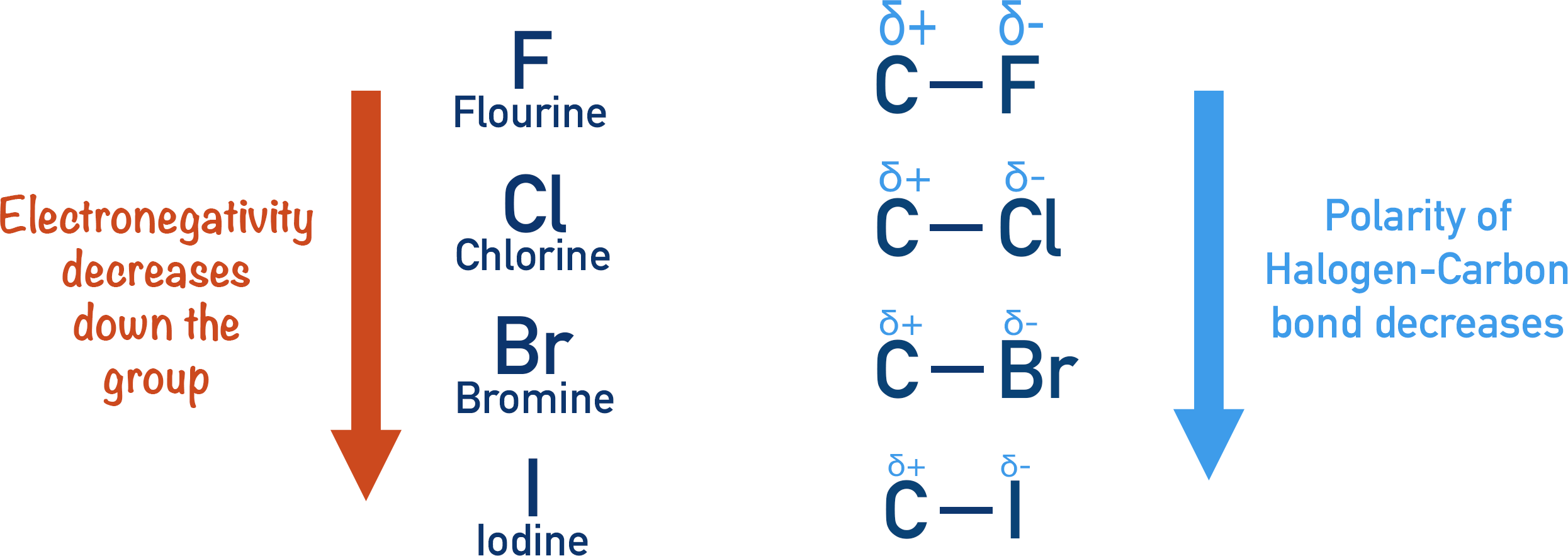
Electronegativity increases as you go across a period in the periodic table, but decreases as you go down a group. This means fluorine is the most electronegative halogen, and iodine is the least electronegative halogen. Carbon-fluorine bonds are the most polar, and carbon-iodine are the least polar.
The halogen in a carbon-halogen bond has a partial negative charge, and the carbon atom has a partial positive charge. This partially positive carbon atom attracts electron donating species (nucleophiles). Nucleophiles can donate their electrons to the carbon, forming a bond. The electrons in the carbon-halogen bond are forced to go to the halogen, which becomes a negatively charged ion with a lone pair of electrons and leaves the molecule.
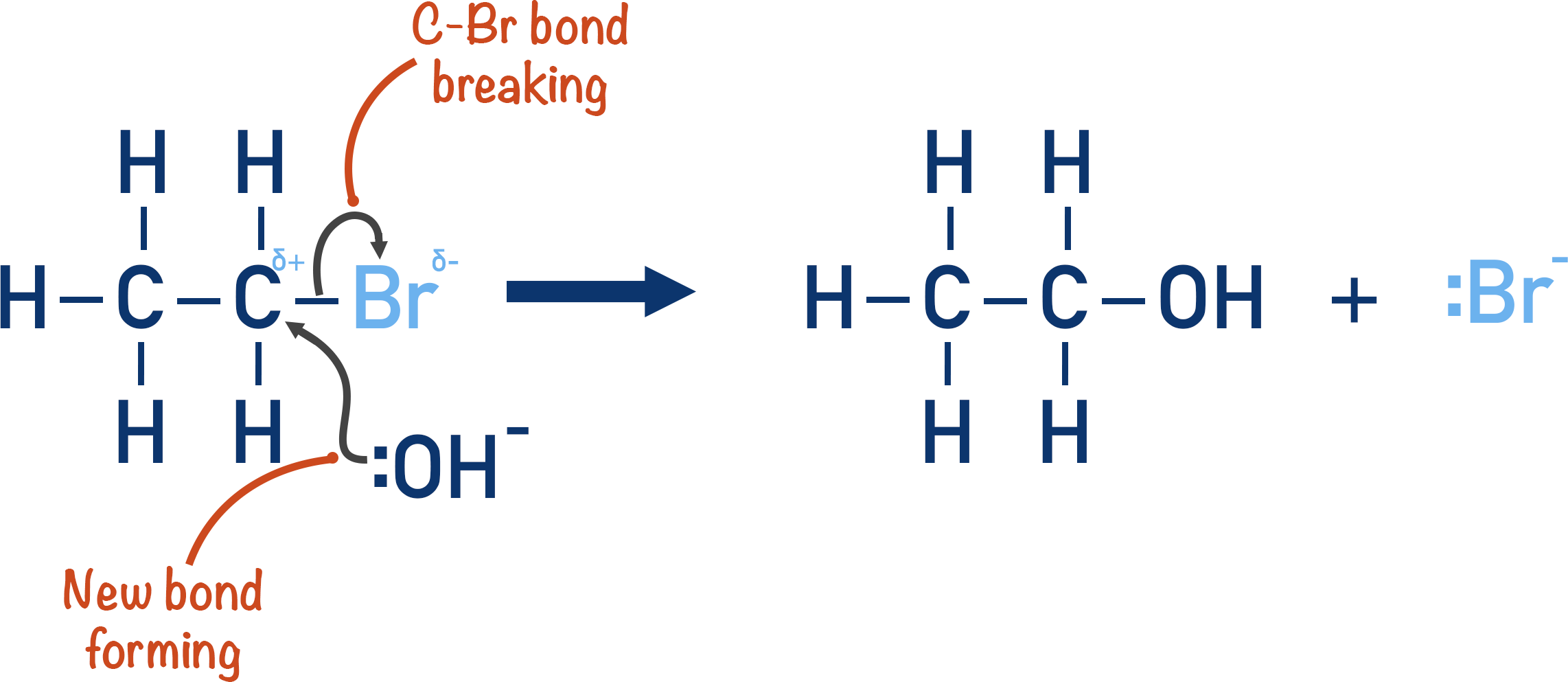
As the nucleophile is effectively ‘swapping’ places with the halogen, the reaction is a nucleophilic substation reaction. The mechanism shown is a common example: the hydrolysis reaction between a halogenoalkane and sodium hydroxide in the presence of water (aqueous conditions), to produce an alcohol.
Halogenoalkane with Ammonia
A type of functional group called amines can be produced when halogenoalkanes are reacted with ammonia (NH3) and ethanol under reflux conditions. The ethanol acts as a solvent. Water cannot be used as the solvent otherwise hydrolysis (see above) would take place.
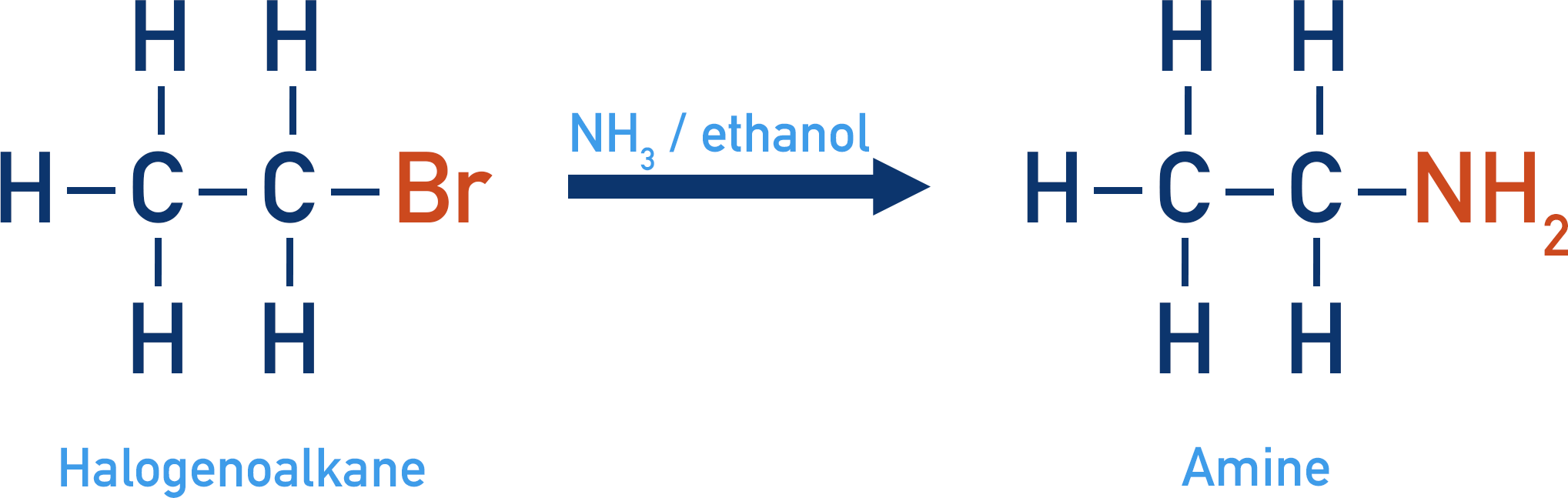
The ammonia acts as a nucleophile and is substituted with the halogen, the reaction is a nucleophilic substitution.
Halogenoalkane with Cyanide Ions
A type of functional group called nitriles can be produced when halogenoalkanes are reacted with cyanide ions (:CN-) and ethanol under reflux conditions. As with a halogenoalkane and ammonia, ethanol acts as a solvent (instead of water) to ensure no hydrolysis takes place.
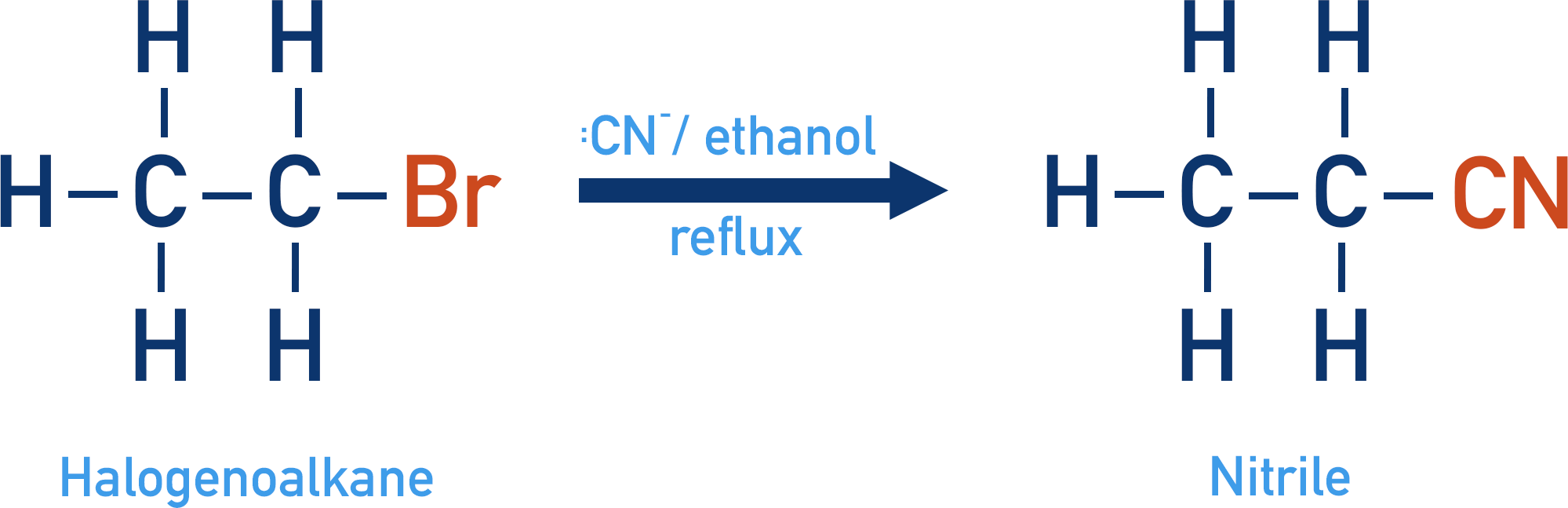
The cyanide ion acts as a nucleophile and is substituted for the halogen, the reaction is a nucleophilic substitution.
We’ve launched our new site! 🎉
Course-specific notes with built-in search!
AP • A-Level (AQA • CIE • Edexcel • OCR) • IB • NCERT 11 + 12
over 750+ new pages and 3,500 images.
Visit the new homepage
