Quick Notes Alkanes
- Alkanes are hydrocarbons in which the carbon atoms are bonded together with single covalent bonds.
- Alkanes are non-polar and do not dissolve in water or polar solvents.
- The combustion of alkanes releases large amounts of energy, making alkanes useful as fuels.
- Complete combustion of alkanes releases carbon dioxide; incomplete combustion releases carbon monoxide.
- Short chain hydrocarbons have low melting and boiling points (due to fewer intermolecular forces holding molecules together).
- Long chain hydrocarbons have high melting and boiling points (due to greater intermolecular forces holding molecules together).
Full Notes Alkanes
If there are only carbon and hydrogen atoms in a molecule, it's called a hydrocarbon. When the carbon atoms are all bonded with single carbon-carbon bonds, it’s called an alkane.
Alkanes are not particularly reactive as they are made up of (relatively) strong carbon to carbon (C-C) and carbon to hydrogen (C-H) covalent bonds.
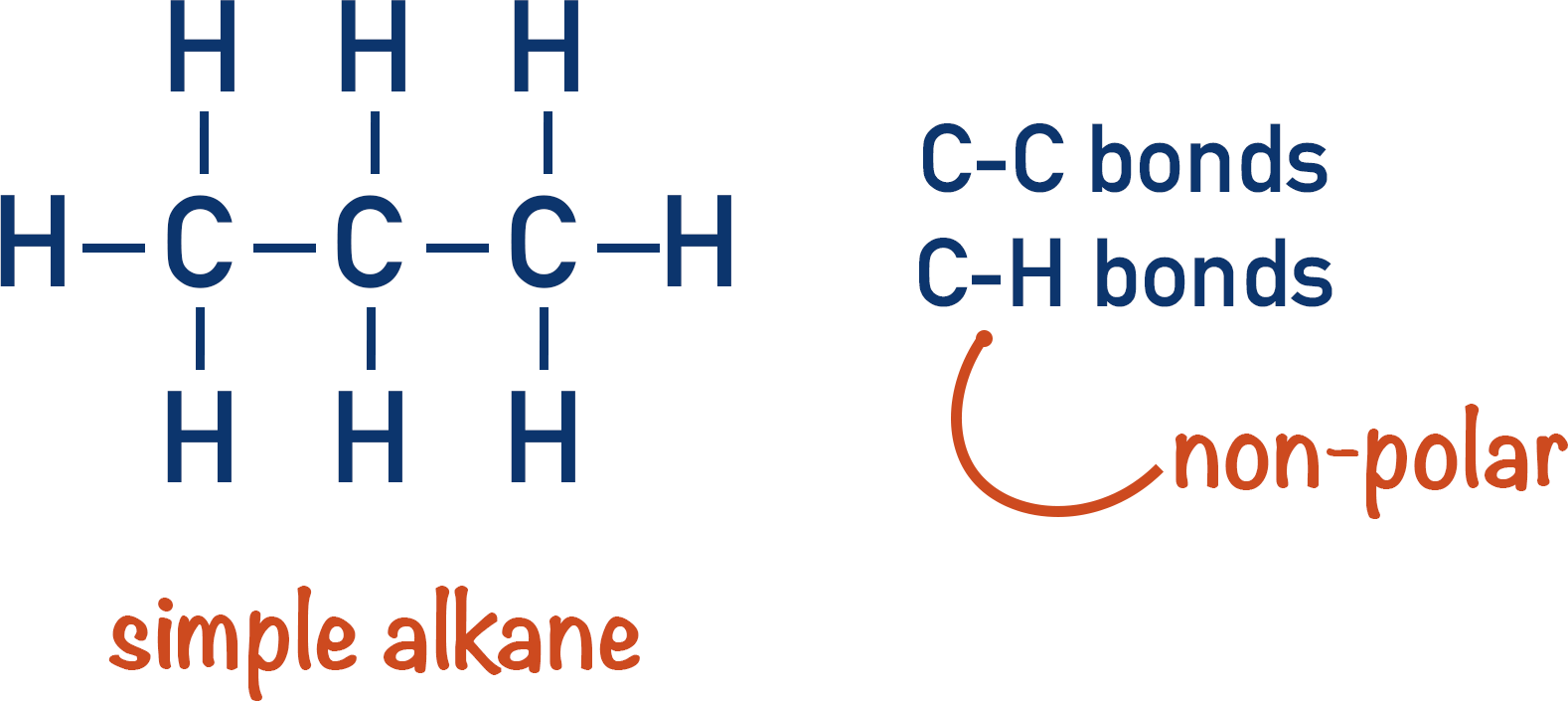
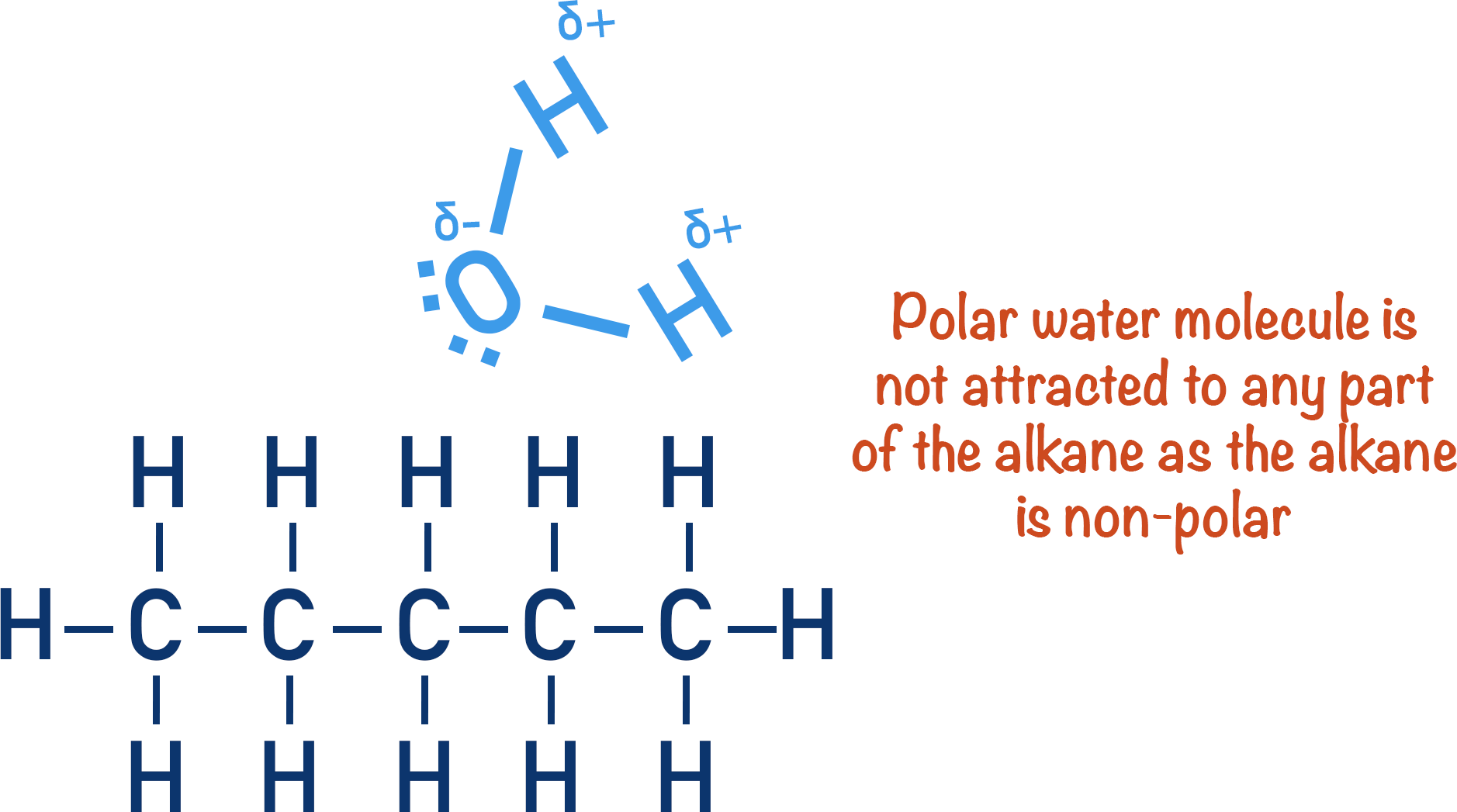
The difference in electronegativity between carbon and hydrogen is small, so carbon-hydrogen bonds are not polar. This is why hydrocarbons do not dissolve in water (liquid hydrocarbons are immiscible in water) and are commonly make up oils.
Combustion
Alkanes combust (relatively) easily. Due to the strong carbon-carbon and carbon-hydrogen bonds, they release large amounts of energy when combusted. Short chain hydrocarbons combust more easily than longer chain hydrocarbons.
When combusted, bonds in the alkane are broken. The elements in the alkane react with oxygen and their oxides are produced. If sufficient oxygen is present, complete combustion takes place, producing carbon dioxide and water.

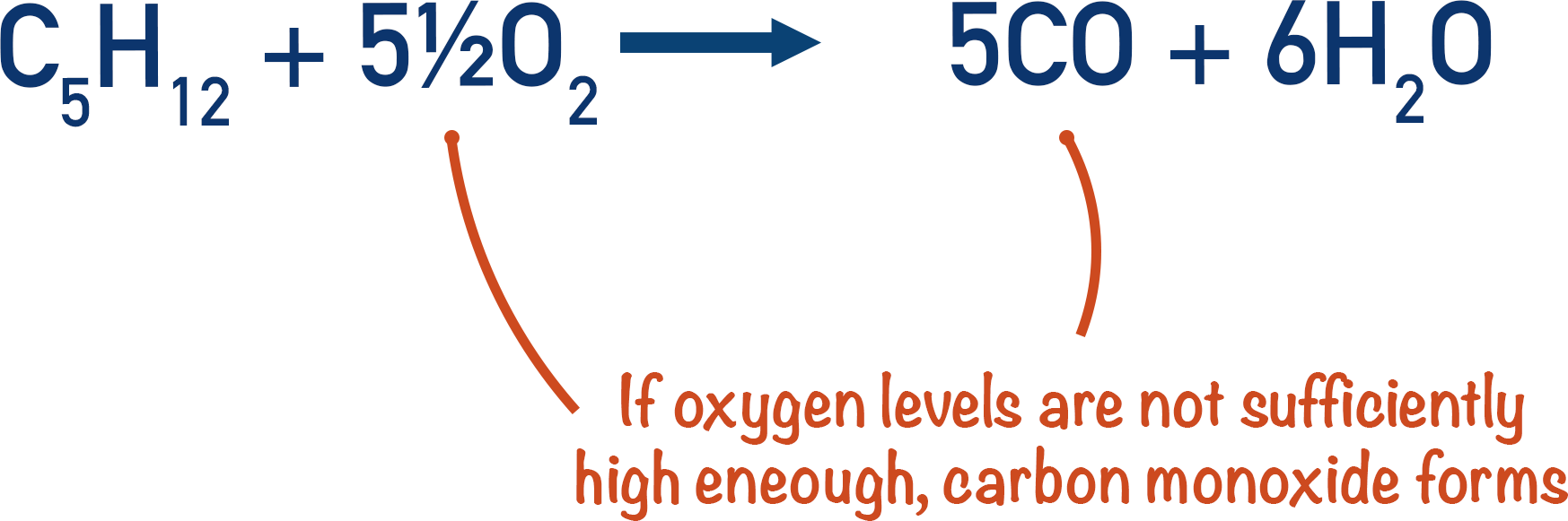
When oxygen levels are low, incomplete combustion can occur, producing carbon monoxide and water.
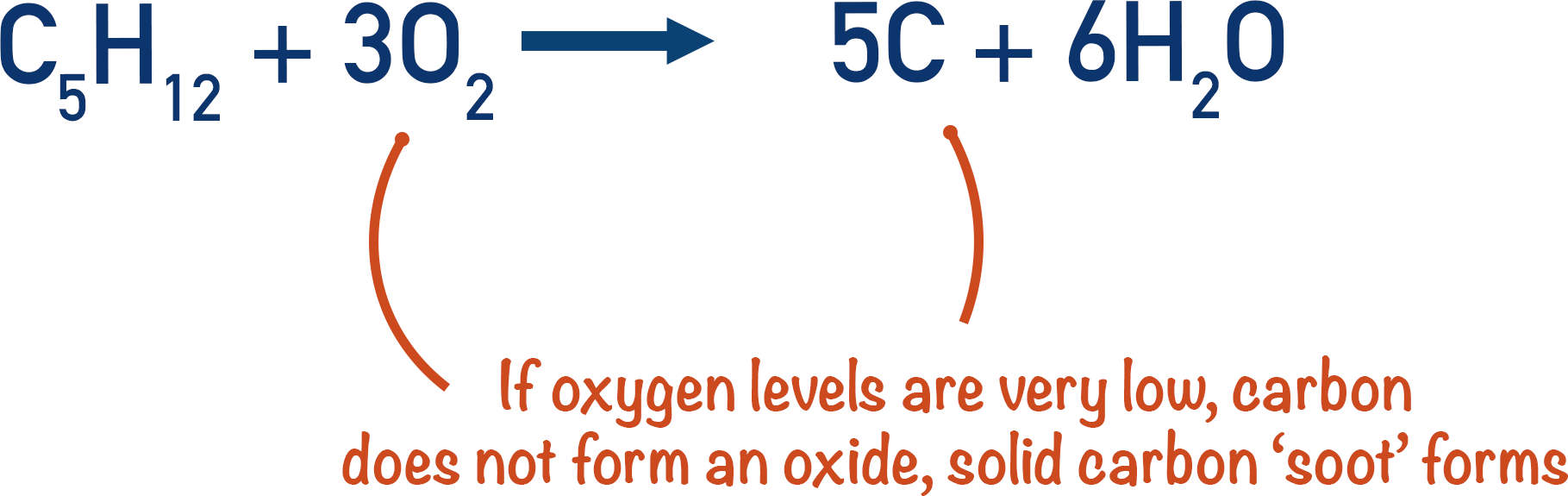
Oxygen levels that are very low can actually cause carbon (soot) and water to be produced.
Depending on the purity of the alkane fuel being combusted, other oxides can be produced. Some of these oxides can be very bad for the environment and toxic for animals; they are called pollutants. Nitrogen oxides, sulphur dioxide and carbon monoxide gases are present in car emissions. The appearance of black smoke from a car exhaust indicates a poor air to fuel mixture in the engine, resulting in insufficient oxygen for complete combustion to occur.
Melting and Boiling Points
Although alkanes themselves don’t tend to react very often, changing the size of their carbon chains can alter their behaviour and properties.
As alkane molecules are not polar, the only intermolecular forces that can exist between them are temporary dipole-dipole interactions. Temporary dipole-dipole interactions are weak and require little energy to break.
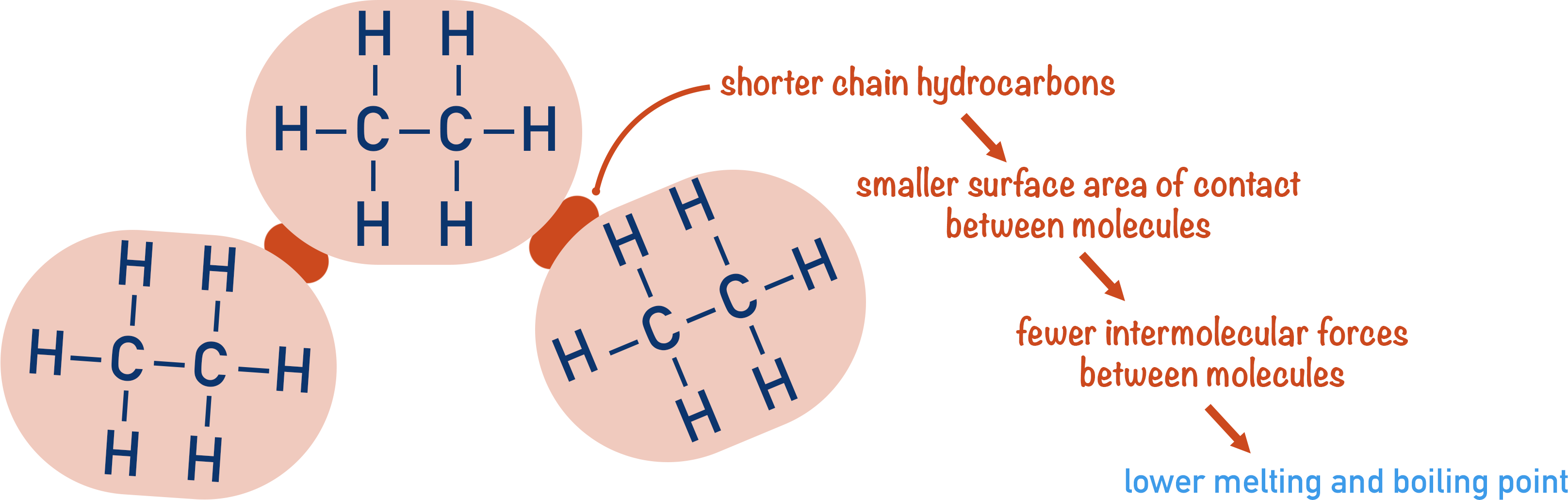
Short chain alkanes are small molecules, so they cannot form many temporary dipole-dipole interactions between themselves. Few interactions means very little energy is needed to overcome them, giving short chain alkanes very low melting and boiling points.
As you increase the carbon chain length, the alkane molecules become longer, so more temporary dipole-dipole interactions can form between them as there is a greater area of contact between molecules. More interactions means more energy is needed to overcome them, giving longer chain alkanes higher melting and boiling points.
