Video Tutorial Free Radical Substitution
Quick Notes Alkanes - Free Radical Substitution
- Covalent bonds can break in two ways:
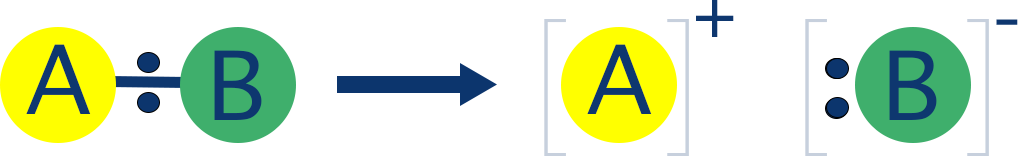
- Heterolytic fission - bond breaks unevenly and both electrons from the bond go to one atom.
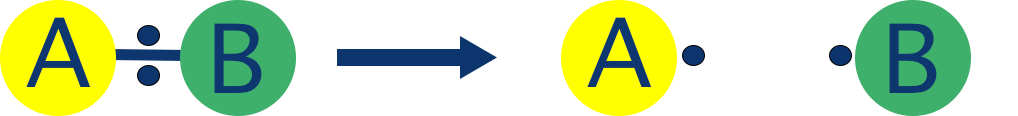
- Homolytic fission - bond breaks evenly and each bonded atom gets one electron, forming free-radicals.
- Free radicals are species that have an unpaired electron and they are highly reactive.
- Halogens can react with alkanes in free-radical substitution reactions, the mechanism occurs as a ‘chain’ reaction with three stages:
- Initiation - U.V. light is needed to start the reaction and cause homolytic fission of the halogen molecule, creating two halogen radicals.
- Propagation – radical species react with the alkane and get substituted into the molecule, creating further radicals.
- Termination – two radical species combine to create a covalent bond and terminate the chain as the product is not a free radical.
- Heterolytic fission - bond breaks unevenly and both electrons from the bond go to one atom.
Full Notes Alkanes - Free Radical Substitution
In a covalent bond, a pair of electrons is shared between two atoms. When a covalent bond breaks, it can break in two ways. The breaking of a bond is called fission.
In one scenario, both bonding electrons go to one of the atoms. This creates a negatively charged ion and a positively charged ion. This kind of bond breaking is called heterolytic fission and is the most common type in organic chemistry.
Note –negative and positive ions aren’t always formed as new bonds can form at the same time as a covalent bond breaks.
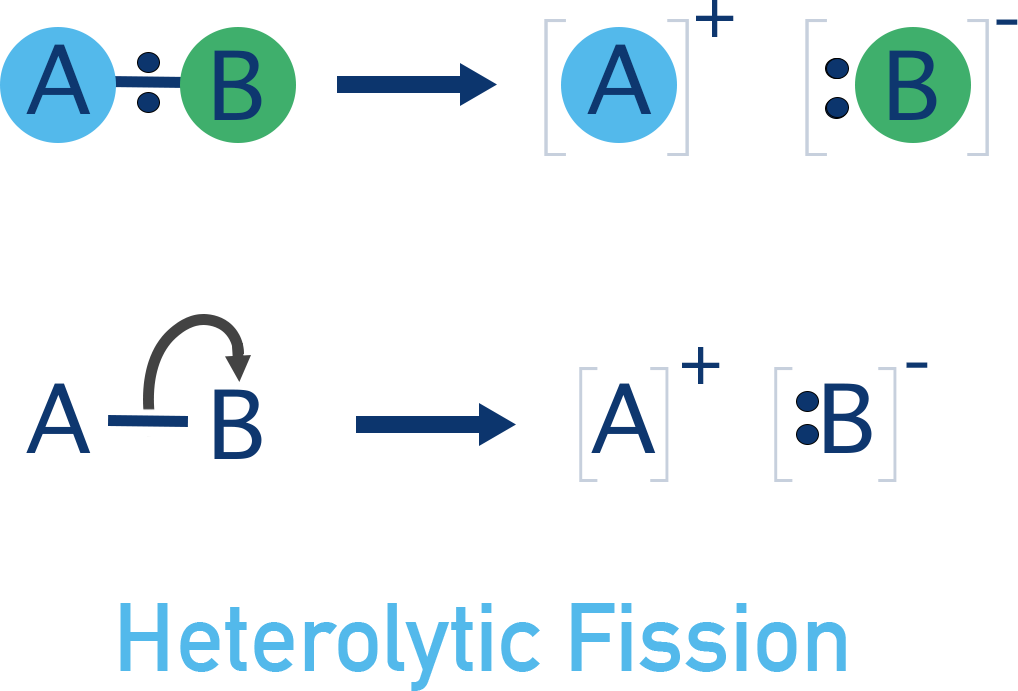
In a second scenario, the bonding electrons could be shared between the two atoms when the bond breaks. This would produce two products that have one unpaired electron each.
A species with one unpaired electron is called a radical. This kind of bond breaking is called homolytic fission. Radicals are highly unstable and very reactive.
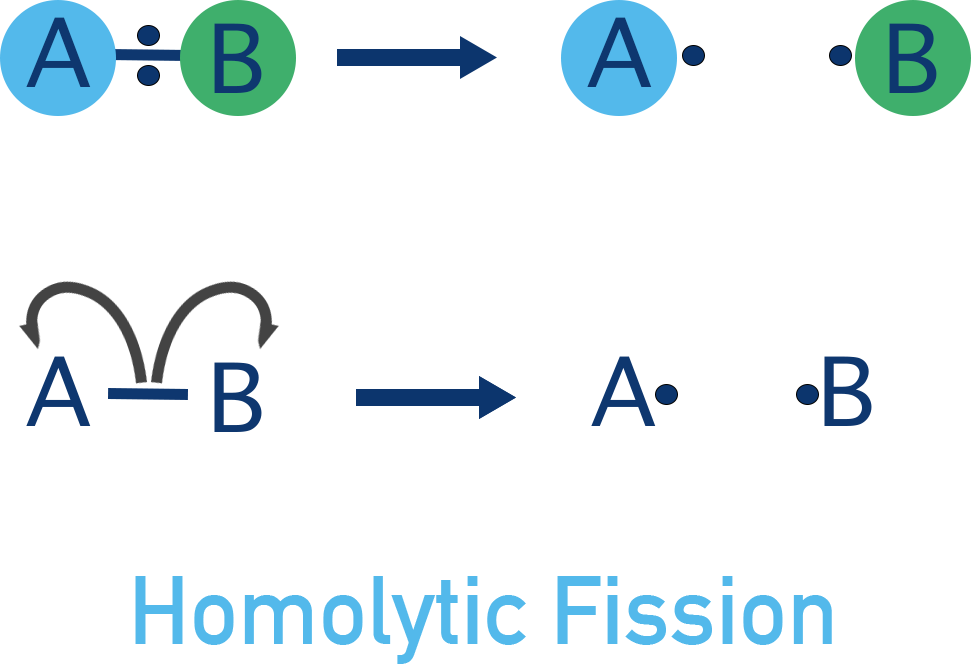
A radical is written with a • symbol to show one unpaired electron (e.g. Br•).
Free-radical Substitution of Alkanes with Halogens
In a substitution reaction, a reacting species is substituted (swapped) for a bonded species in a compound.
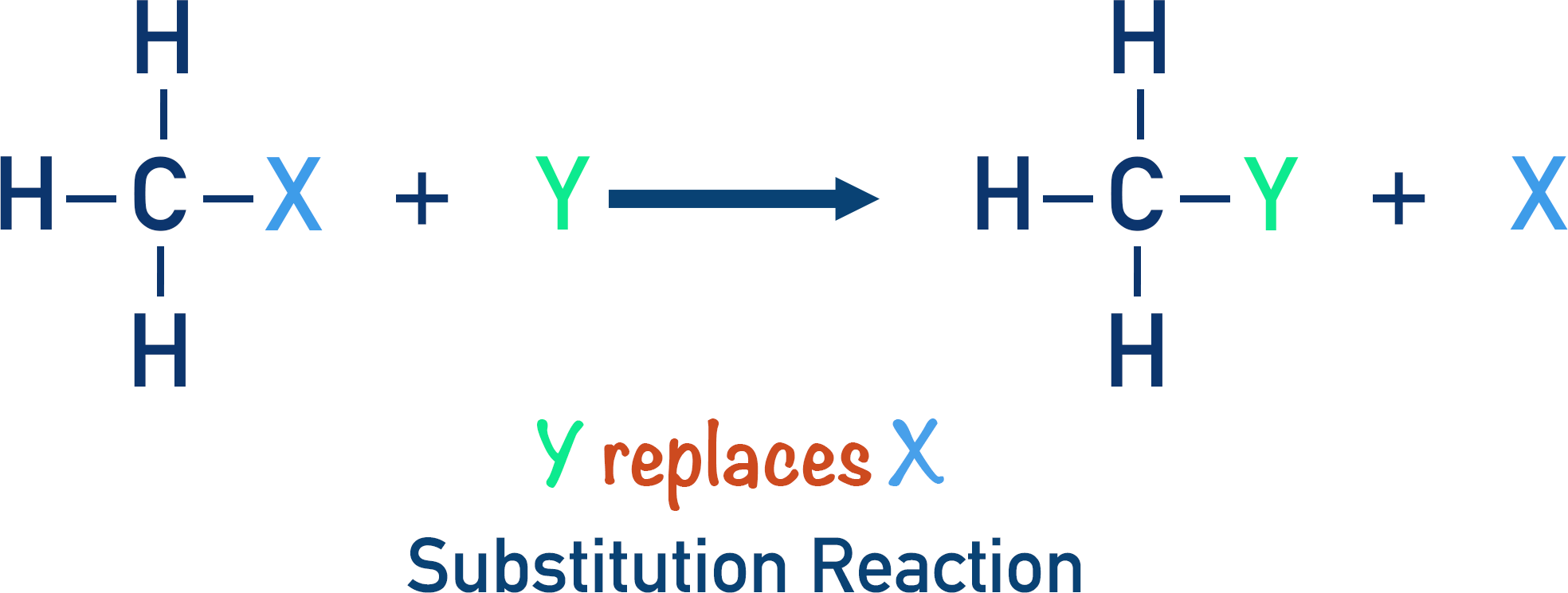
Alkanes can react with halogens in the presence of ultraviolet (UV) light. A halogen is substituted for a hydrogen atom in the alkane to form a halogenoalkane.
The process happens in stages. These stages link together to gives us a ‘mechanism’ that describes how products are formed from the reactants.
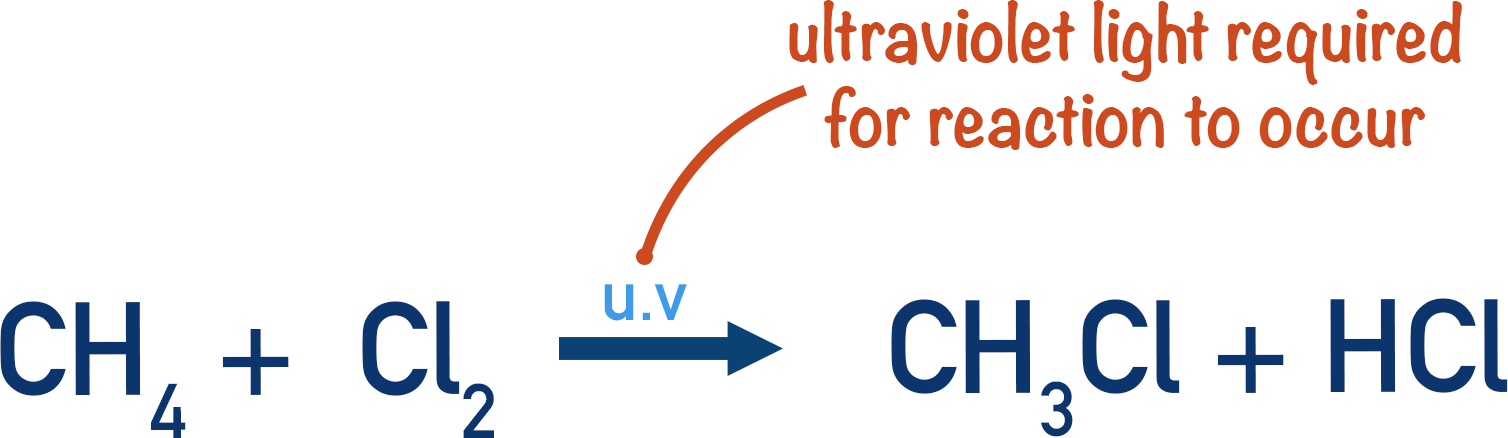
Methane with Chlorine Substitution
Initiation step
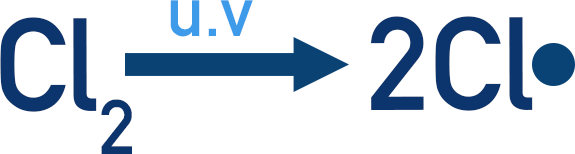
A halogen and an alkane will not react without UV light. The UV light is required to form a halogen radical.
In this example, the UV light gives energy to the covalent bond in Cl2, so that it undergoes homolytic fission (see above). Producing two Cl• radicals.
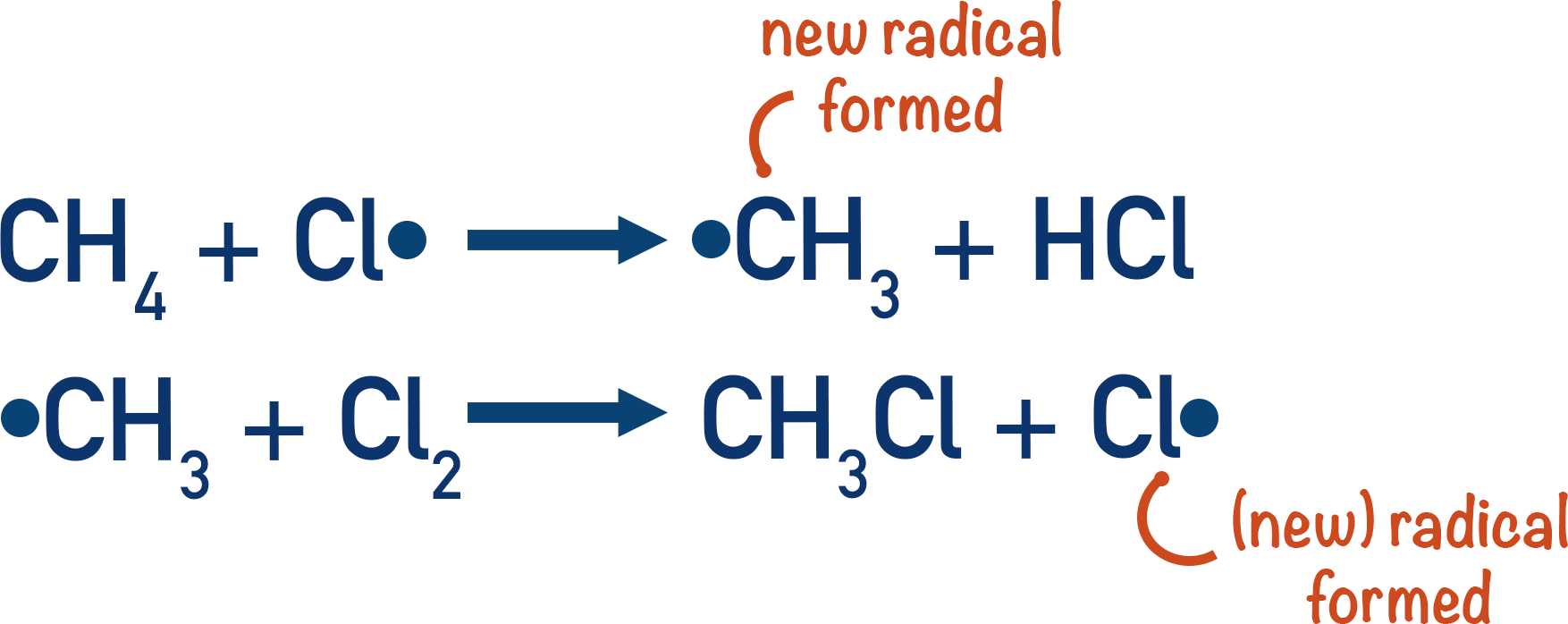
Propagation step
Once radicals have been produced in the initiation step, they are free to react with the alkane present (in this case methane).
As radicals contain an unpaired electron, unless a radical is reacting with another radical, the products produced must also contain an unpaired electron. New radicals are therefore produced when a chlorine radical reacts with an alkane.
These new radicals can then react again, producing the desired product and the same species that originally formed the starting radical (in this case, Cl2).
Termination step
When two radicals react together, two unpaired electrons come together and a covalent bond forms. This means a neutral product ( that isn't a radical) get produced and the chain reaction ends at this point. Any two radicals can react to end the chain process at any time.
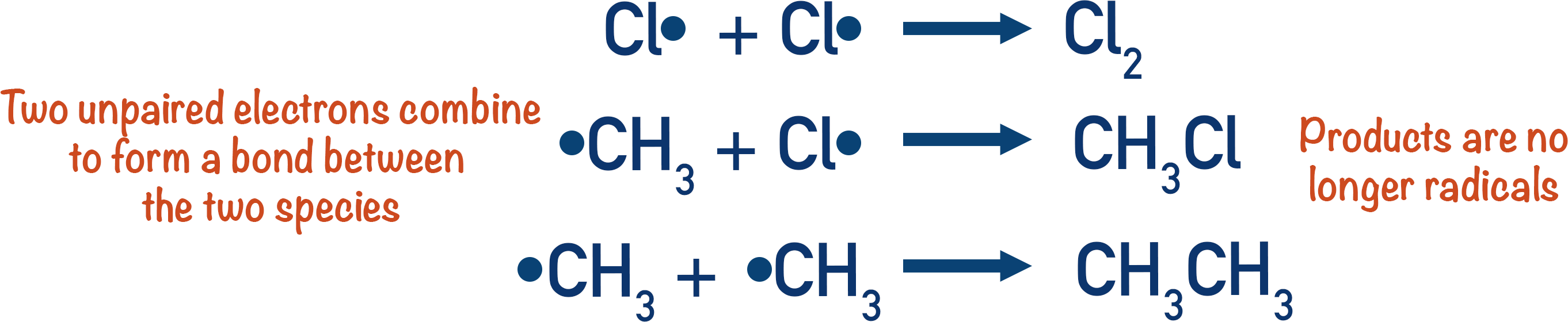
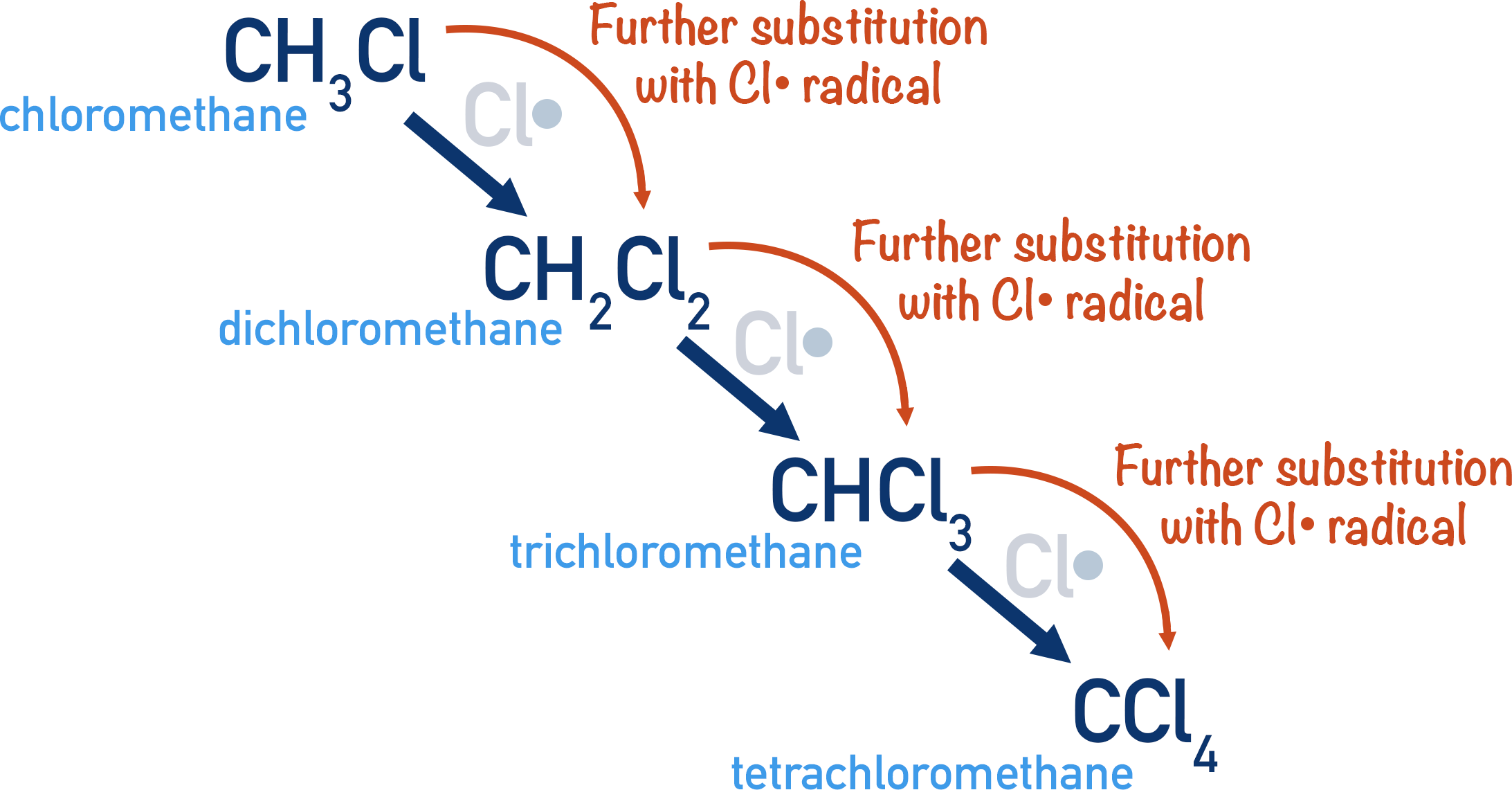
It is important to note that further substitution reactions can happen between the halogenoalkane formed and other chlorine radicals, giving di-chloromethane, tri-chloromethane and tetra-chloromethane (carbon tetrachloride).
We’ve launched our new site! 🎉
Course-specific notes with built-in search!
AP • A-Level (AQA • CIE • Edexcel • OCR) • IB • NCERT 11 + 12
over 750+ new pages and 3,500 images.
Visit the new homepage
