Video Tutorial Boltzmann Distribution Curves
Quick Notes Boltzmann Distribution Curves
- Maxwell-Boltzmann distribution curves show how available energy is distributed among molecules in a gaseous system.
- At a given temperature, only a certain amount of energy is available for all molecules of gas within a system.
- Individual molecules have different energies and are constantly colliding, transferring energy with other molecules – either gaining or losing energy.
- The effects of changing concentration, temperature and the use of a catalyst on the energies of particles can be shown.
Full Notes Boltzmann Distribution Curves
At a particular temperature, there is only so much energy available for molecules in a gaseous system. This energy is never evenly spread out between the molecules; at any given moment, some molecules will have more energy than others. Maxwell-Boltzmann distribution curves show how available energy is distributed among the molecules in a system. The distribution curve applies to gases at a constant temperature.
Note, as molecules are constantly colliding with each other, they are constantly transferring energy. This means the energy of a gas molecule is never fixed, it is constantly changing. Maxwell-Boltzmann distribution curves show average distributions.
The distribution curves can be used to show how many molecules have the required activation energy in a system.
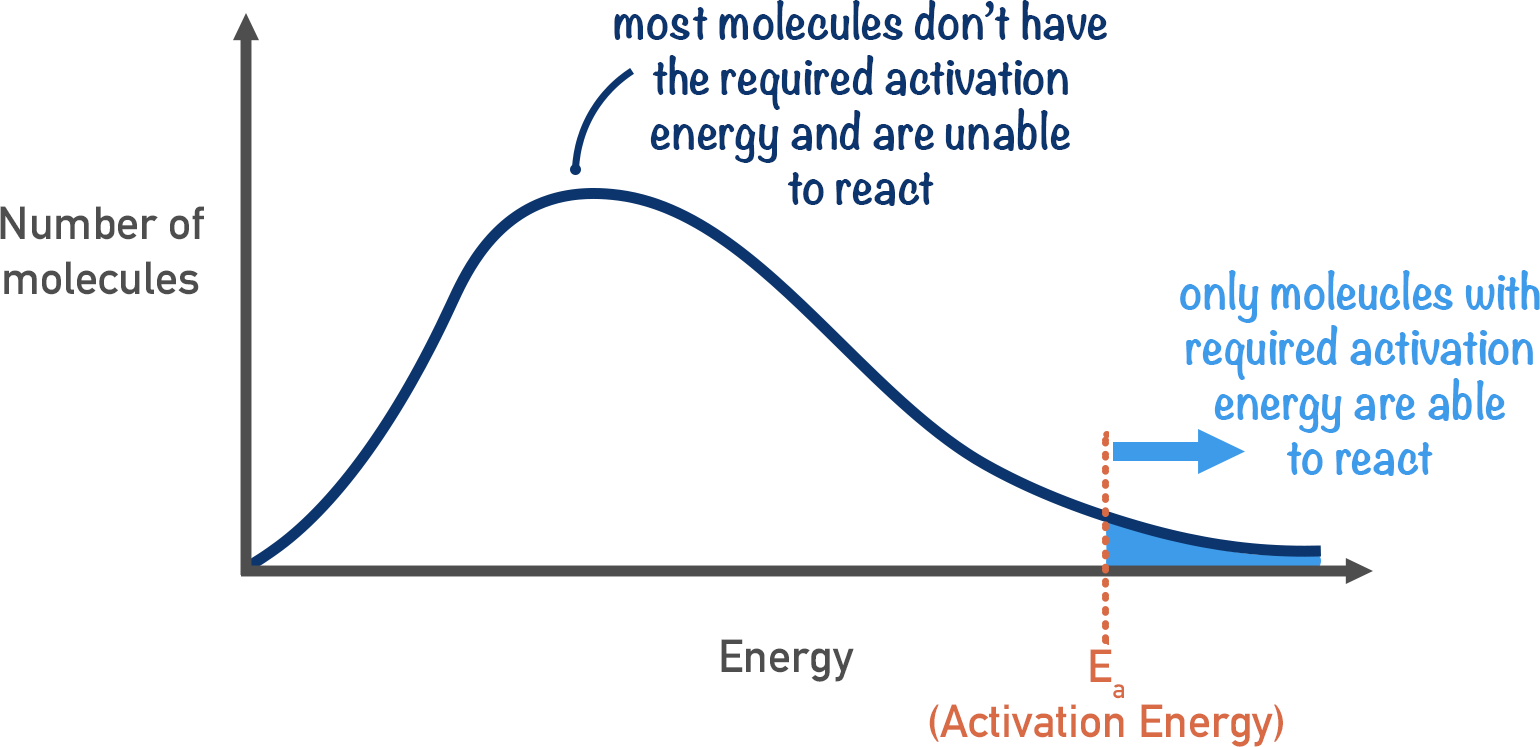
All particles in a system have energy, but some have more energy than others. Particles with the activation energy required for a reaction can successfully collide and react.
Increasing Temperature
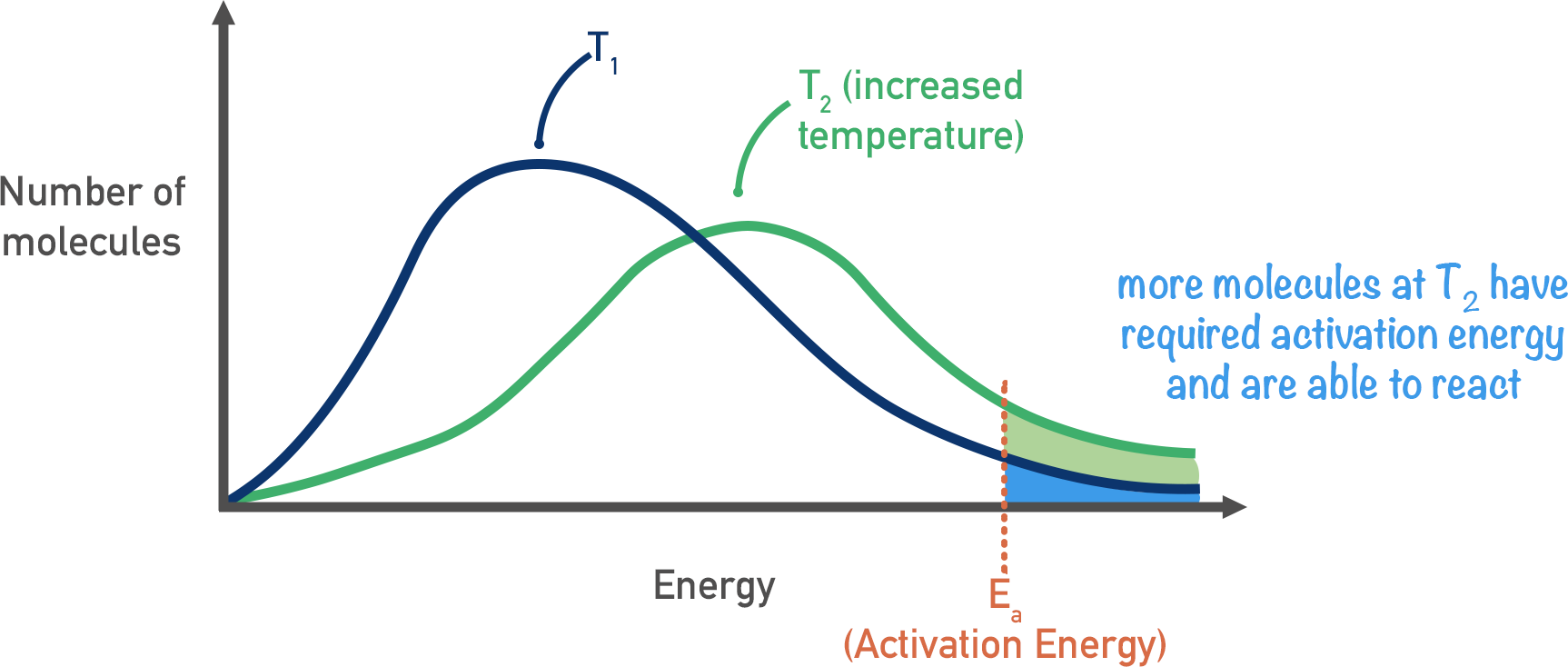
By increasing the temperature of a system, the energy of the particles increases. So, more particles have the required activation energy.
Increasing Concentration
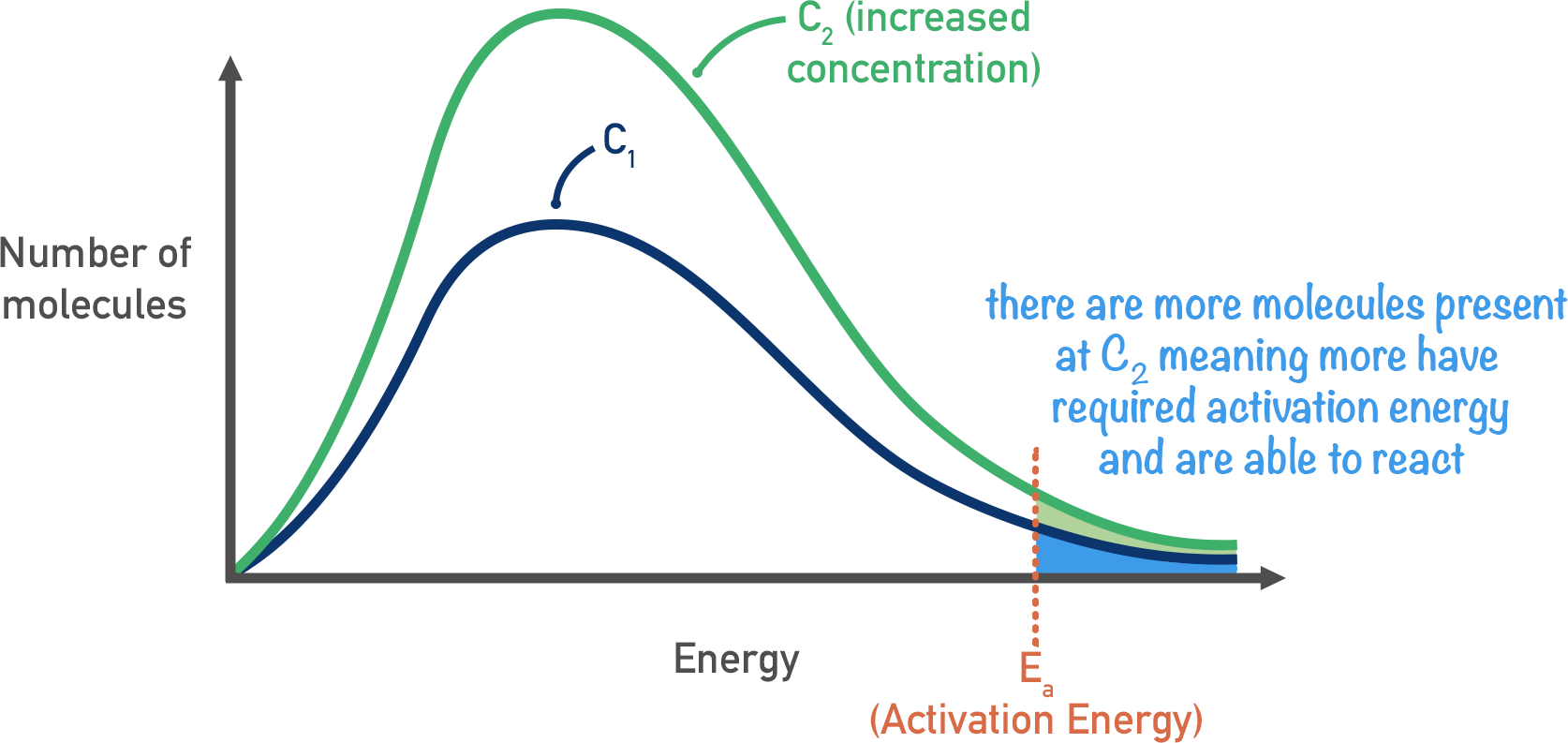
By increasing the concentration of molecules within a system, the actual energy of the molecules doesn’t change. The proportion of molecules with the required activation energy is the same, there are just more of them! Because of this, more particles in the system have the required activation energy and react.
Using a Catalyst
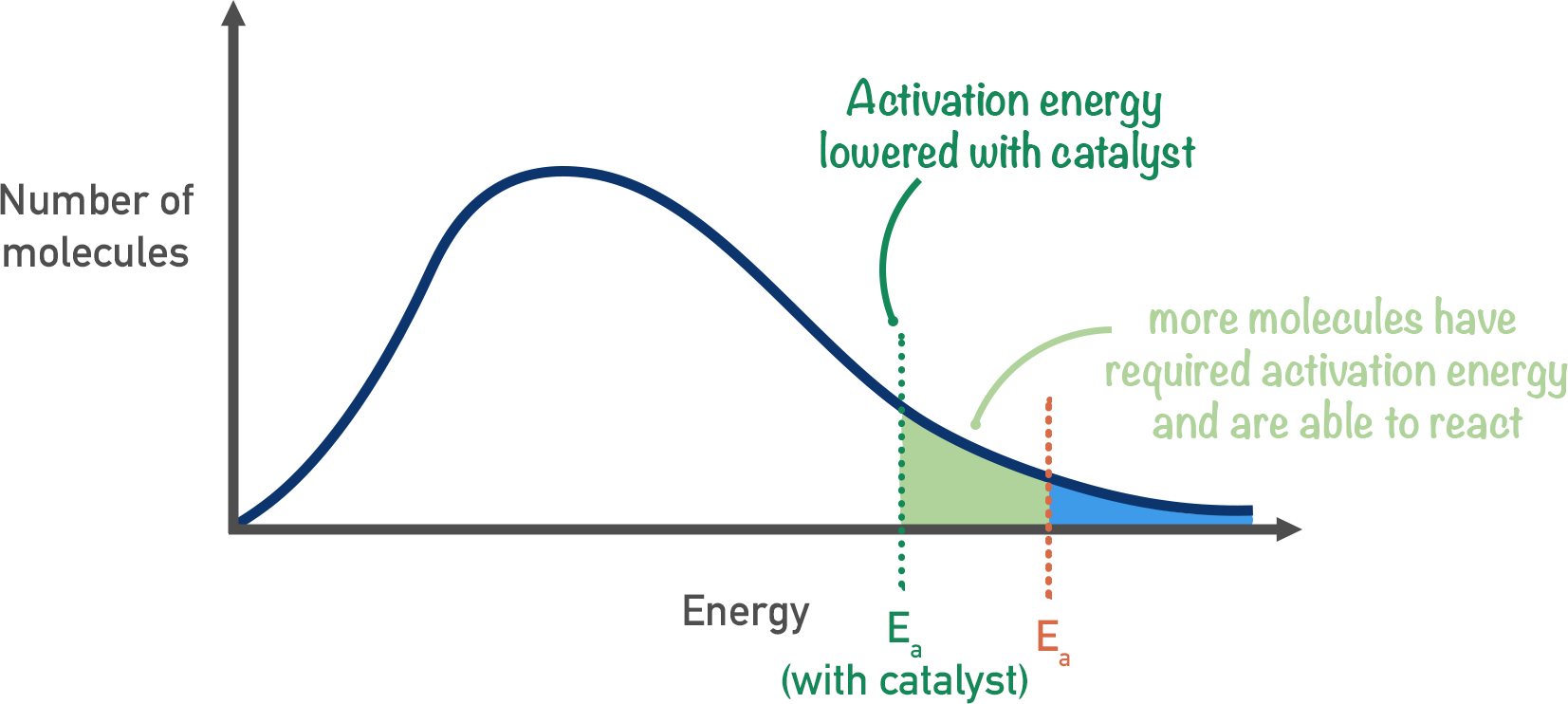
By using a catalyst, the energy of particles in the system remains the same, but the activation energy required for a successful collision is lowered. As a result, more particles are able to successfully collide.
We’ve launched our new site! 🎉
Course-specific notes with built-in search!
AP • A-Level (AQA • CIE • Edexcel • OCR) • IB • NCERT 11 + 12
over 750+ new pages and 3,500 images.
Visit the new homepage
