Video Tutorial Orders of Reaction
Quick Notes Orders of Reaction
- Changing the concentration of reactants in a reaction can affect the rate of the reaction.
- The orders of a reaction describe how much the rate of a reaction changes when the concentrations of reactants are changed.
- The order of a reaction with respect to a reactant describes how much the rate of a reaction changes when the concentration of that reactant is changed by a given factor.
- Zero order means changing the concentration of the reactant has no effect on the rate of reaction.
- First order means changing the concentration of the reactant by a given factor will change the rate of the reaction by the same factor.
- Second order means changing the concentration of the reactant by a given factor will change the rate of the reaction by the same factor squared (x2).
Full Notes Orders of Reactions
The rate of a reaction is a measure of how fast a reaction is occurring. Changing the concentrations of reactants in a reaction can alter how fast the reaction happens, but not always by the factor we have changed the concentration by. For example, sometimes a reactant's concentration is doubled and yet the reaction rate doesn’t change at all!
How much the rate of a reaction is determined by the concentrations of its reactants is described by its ‘order of reaction’.
Orders of a reaction just link changes in concentrations of reactants to changes in the rate of a reaction. The order of a reaction ‘with respect to…’ just means how changing the concentration of a particular reactant affects the rate of the reaction (independent of other reactants).
There are three types of orders you need to know for A-level Chemistry – zero order, first order and second order.
For example, in the following reaction:

Let’s say we are measuring the rate of this reaction, and we want to see how changing the concentrations of each reactant affects the overall rate.
Zero Order
If we double the concentration of A and the rate does not change, then the order of the reaction with respect to A is zero order.
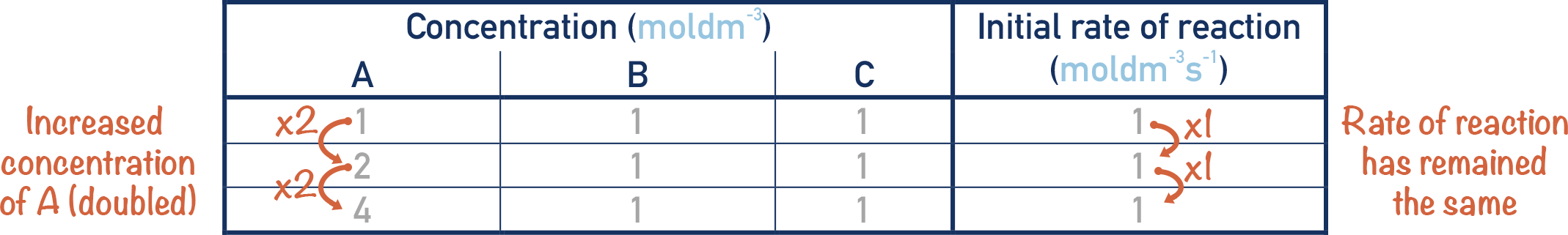
It does not matter what we change the concentration of A to, the rate will always be the same.
First Order
If we double the concentration of B and the rate also doubles, then the order of the reaction with respect to B is first order.
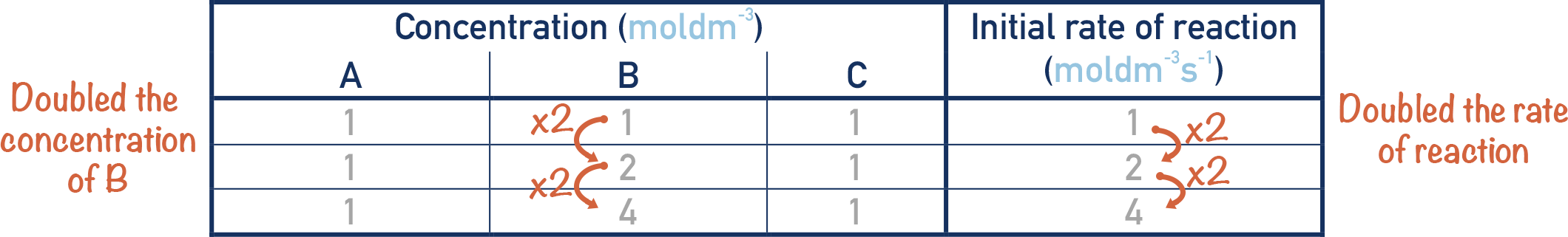
Whatever factor we change the concentration of B by, the rate will change by the same factor (i.e. doubling the concentration of B results in a doubling of the rate, halving the concentration of B results in a halving of the rate, and so on).
Second Order
If we double the concentration of C, and the rate quadruples, then the order of the reaction with respect to C is second order.
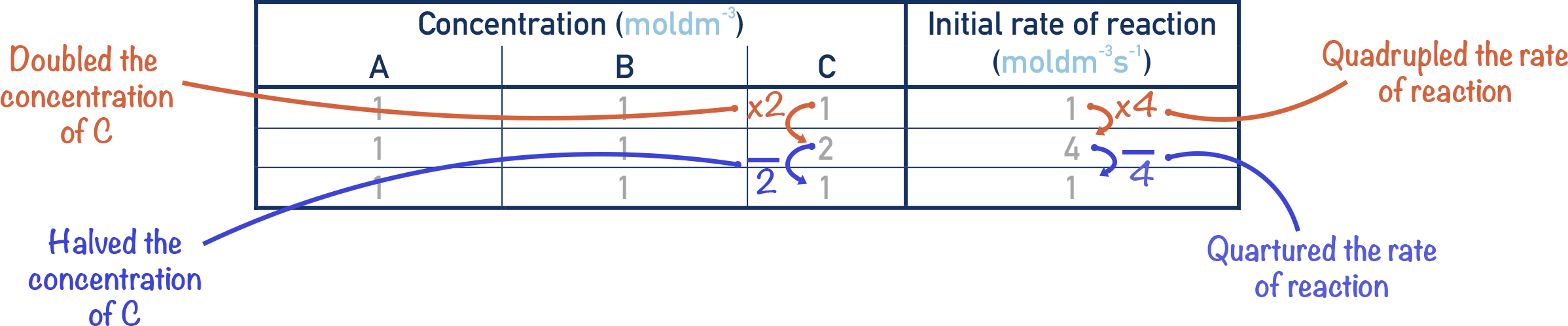
Here, the rate of a reaction is changed by a factor that is the change in concentration squared. If we double the concentration of C, then the rate would change by 22 = 4. If we halved the concentration of C, then the rate would change by 0.52 = 0.25 (the rate would be quartered).
We’ve launched our new site! 🎉
Course-specific notes with built-in search!
AP • A-Level (AQA • CIE • Edexcel • OCR) • IB • NCERT 11 + 12
over 750+ new pages and 3,500 images.
Visit the new homepage
