Video Tutorial Covalent Bonding
Quick Notes Covalent Bonding
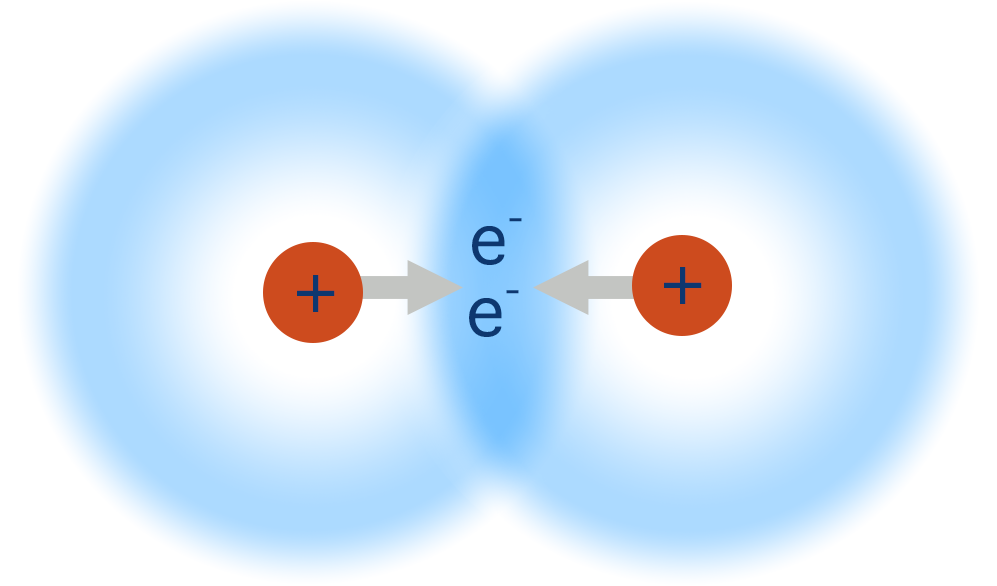
- A covalent bond is the sharing of one pair of electrons between two atoms.
- Two atomic orbitals from two atoms overlap and create a new area of electron density between the two atoms.
- The positively charged nuclei of both atoms get attracted to the shared pair of negatively charged electrons and this pulls them closer together.
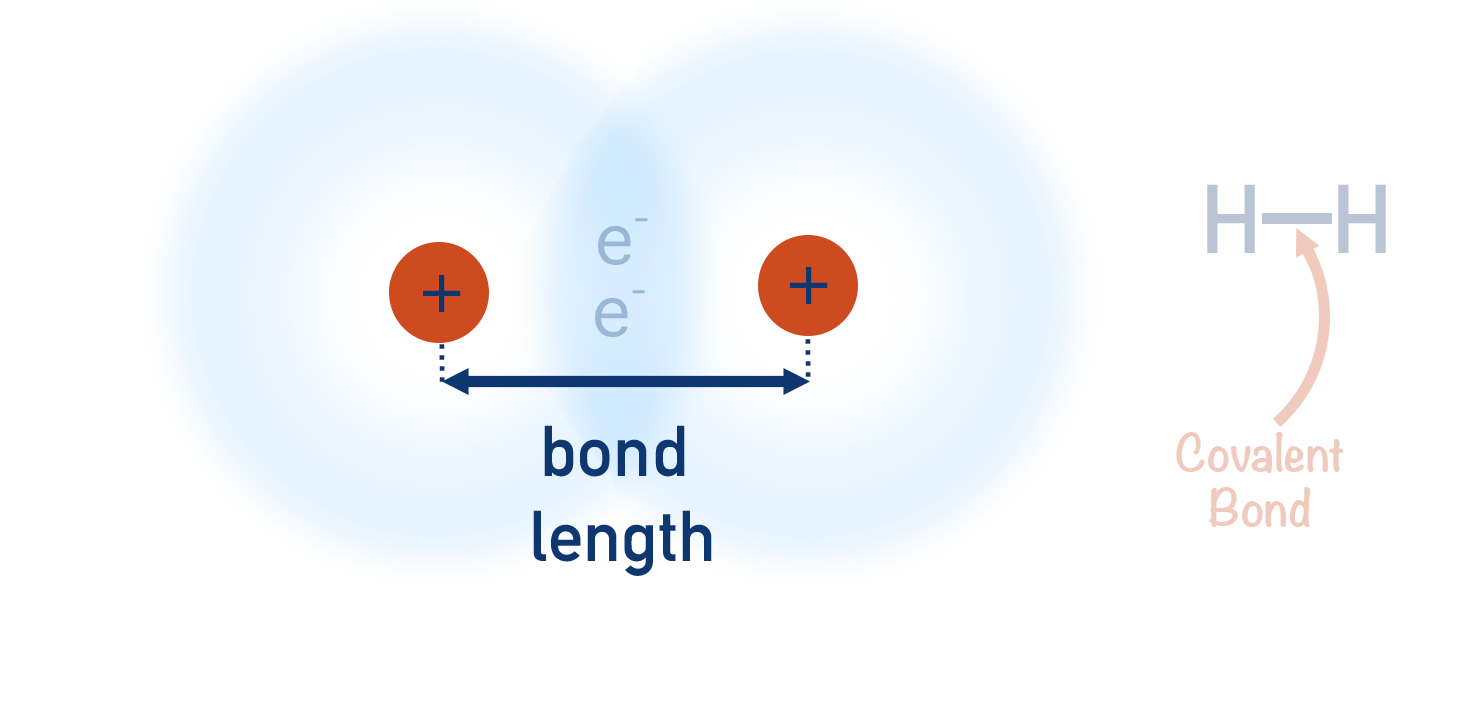
- The distance between the centre of each nuclei is called a ‘bond length’.
- Non-metal atoms form covalent bonds with other non-metal atoms.
- As covalent bonds occur between atoms, they are atomic bonds.
Full Notes Covalent Bonding
Atomic Structure Recap
Atoms have electrons that travel around a nucleus in ‘orbitals’. Each orbital can have a maximum of one pair of electrons in. These orbitals exist in shells, and each shell is a different distance from the nucleus (1 being the closest, 2 the next closest, etc.).
With covalent bonding, we are concerned with the outermost electrons and shell of an atom.
In a covalent bond, a pair of electrons is shared between two atoms. The positively charged nuclei of both atoms get attracted to the shared pair of negatively charged electrons and this pulls them in closer together.
For example...
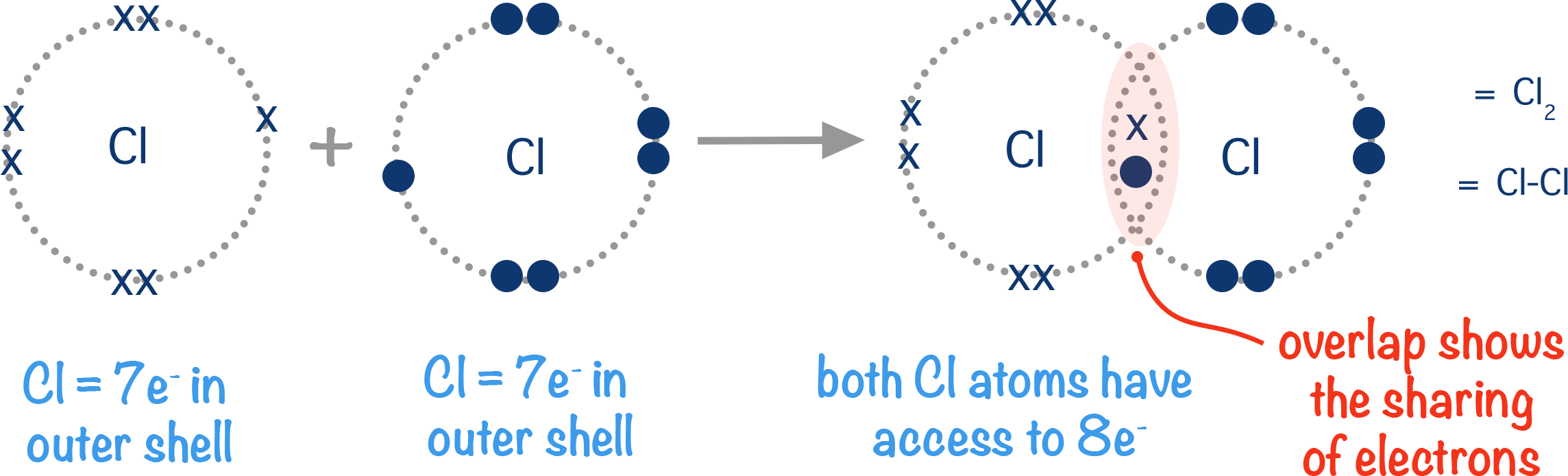
Chlorine atoms have seven electrons in their outer shell. This means they have one electron in a half filled orbital. To become stable, they need to gain access to one more electron to 'fill' this orbital. Two chlorine atoms can each share their one electron from a half filled orbital with the other atom. This forms a covalent bond and now, each atom has ‘access’ to a full orbital.
How does a covalent bond form?
When a covalent bond forms, two half filled atomic orbitals from two atoms overlap and create a new area of electron density between the two atoms, called a bonding orbital. Each half filled atomic orbital contained one electron each, meaning the new bonding orbital now contains a full pair of electrons.
As the two nuclei move closer together, they start to repel each other, meaning if they get too close, the repulsion force would be stronger than the attractive force to the shared electrons. Other electron orbitals in both atoms will also start to repel the bonding electrons. As a result, the two nuclei stay at a certain distance from each other - close enough to maximise their attraction to the negatively charged electrons and at the same time, far enough away from each other to minimise the repulsion between them both. The distance between the centre of each nuclei to each other is called a ‘bond length’.
The simplest example of a covalent bond forming is between two hydrogen atoms, as outlined below.
Forming of a covalent bond between two hydrogen atoms
We can use the coming together of two hydrogen atoms to show how a covalent bond can form.
A single atom of hydrogen has one electron and a nucleus of one proton. This means it has one electron in its outer shell - in an s-shaped orbital. The electron is attracted to the positively charged nucleus (proton) and is free to exist anywhere in its s-orbital.
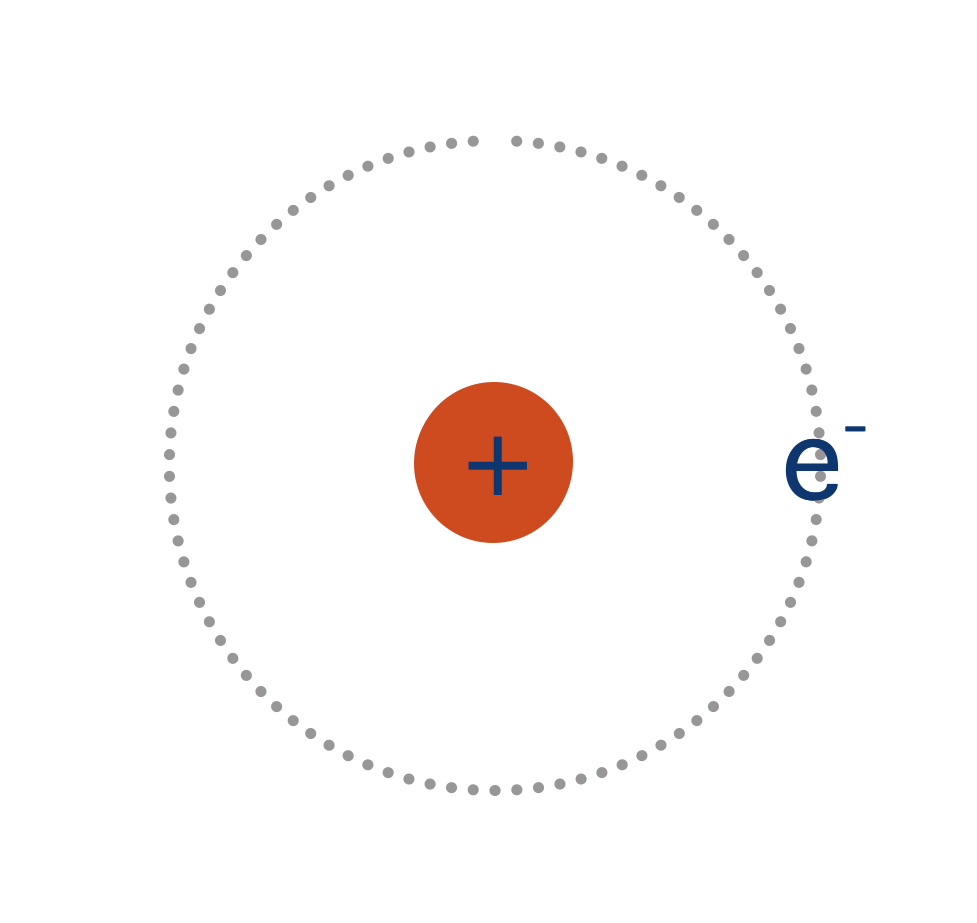
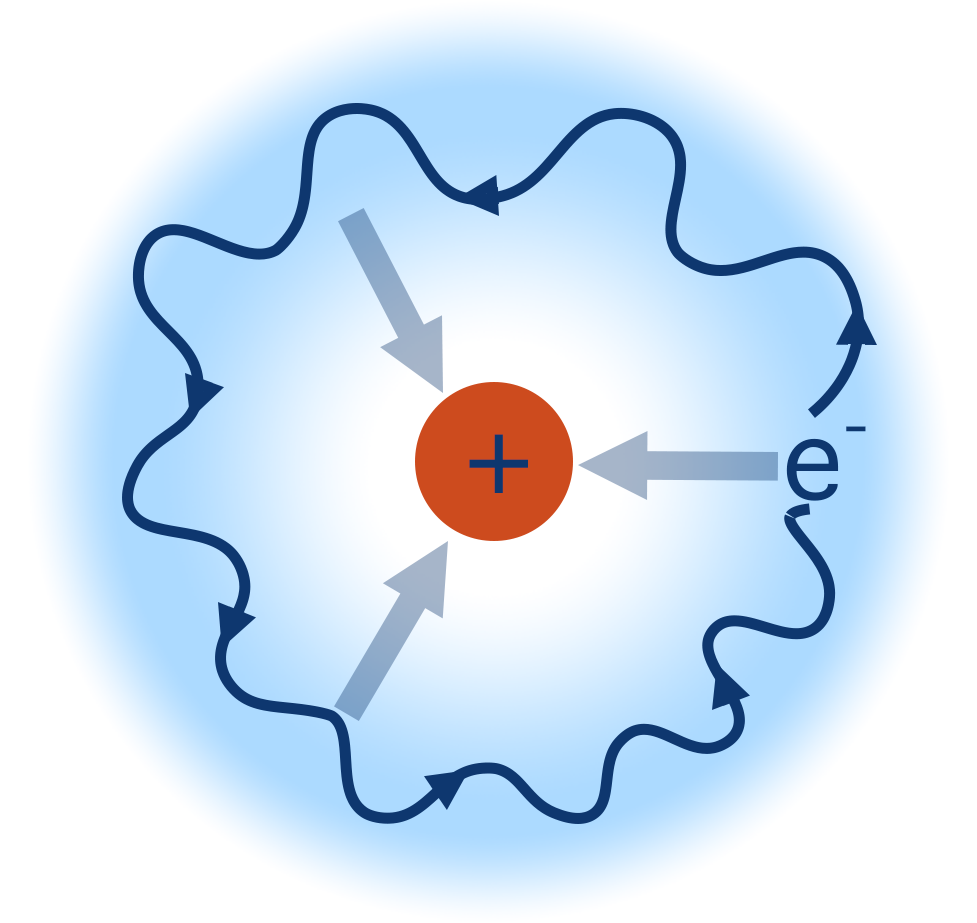
As for any atomic orbital, the s-orbital is able to hold two electrons, and so at the moment is ‘half filled’.
If another hydrogen atom, also with one electron in its 1s orbital, came close to the first hydrogen atom, the 1s orbitals from both atoms can start to overlap as each can ‘take’ another electron. Remember, one orbital can hold a pair of electrons.
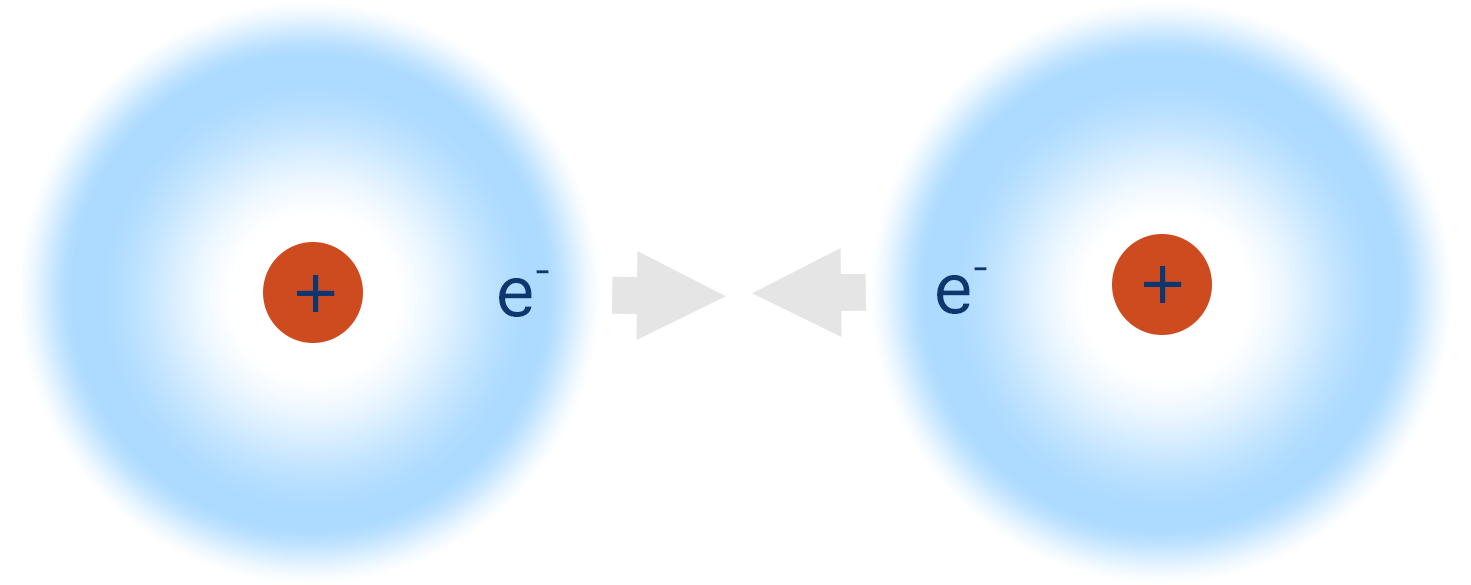
As the orbitals continue to overlap, there will come a point where an electron in one hydrogen atom will be just as attracted to the positive nucleus from the other hydrogen atom as it is to its ‘own’ nucleus.
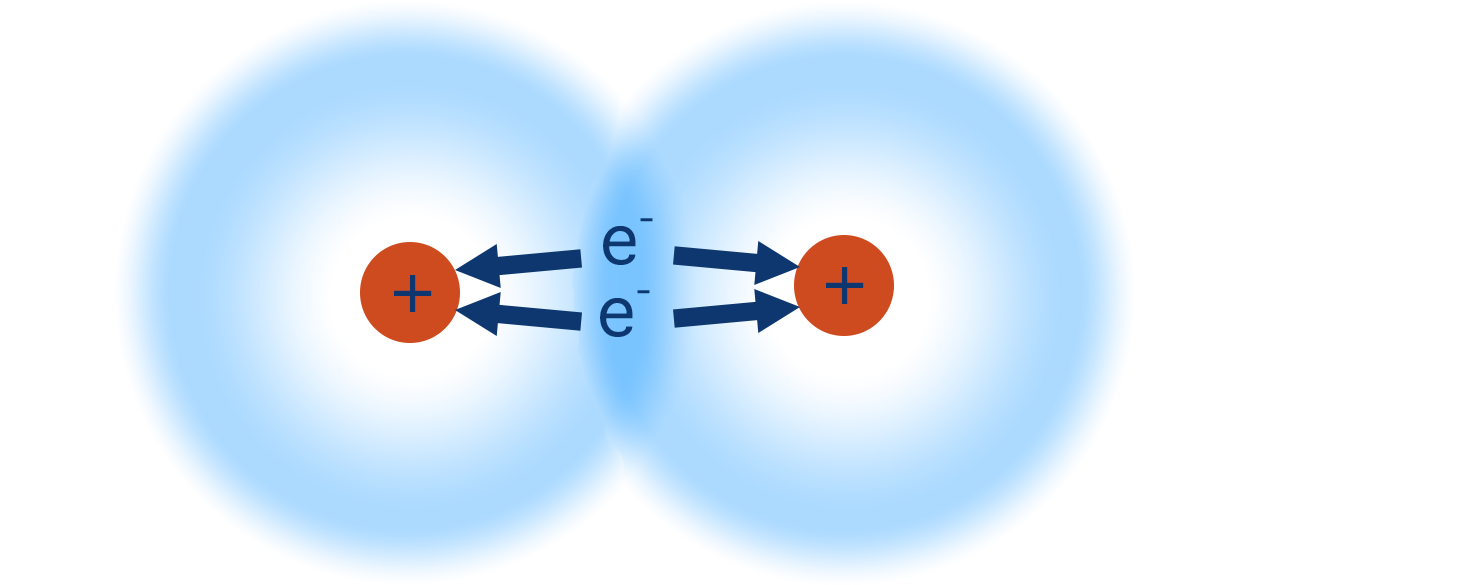
At the same time, the positively charged nuclei of both hydrogen atoms will be attracted to the increased electron density between them, pulling them inwards and closer together.
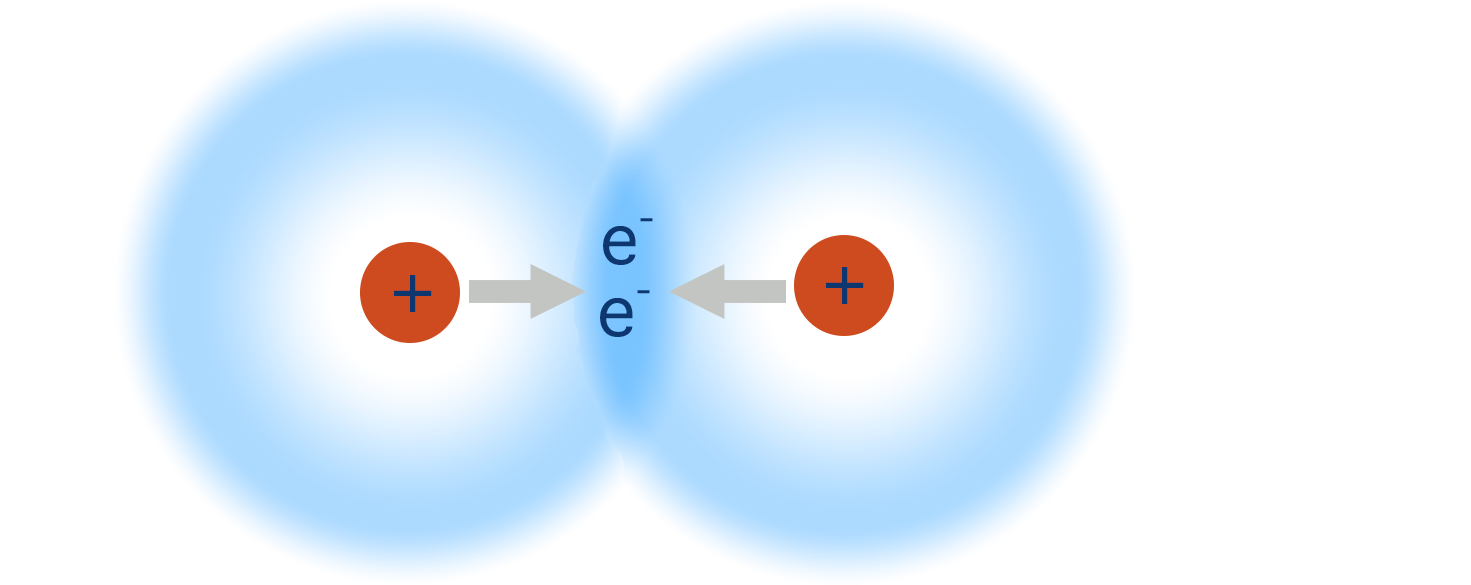
As this happens, the orbitals from both hydrogen atoms start to ‘merge’ creating a new orbital, called a ‘bonding’ or ‘molecular’ orbital. Now, both electrons can exist anywhere within this new orbital. Each hydrogen atom also has ‘access’ to two electrons and as a result can be considered as having a full outer shell.
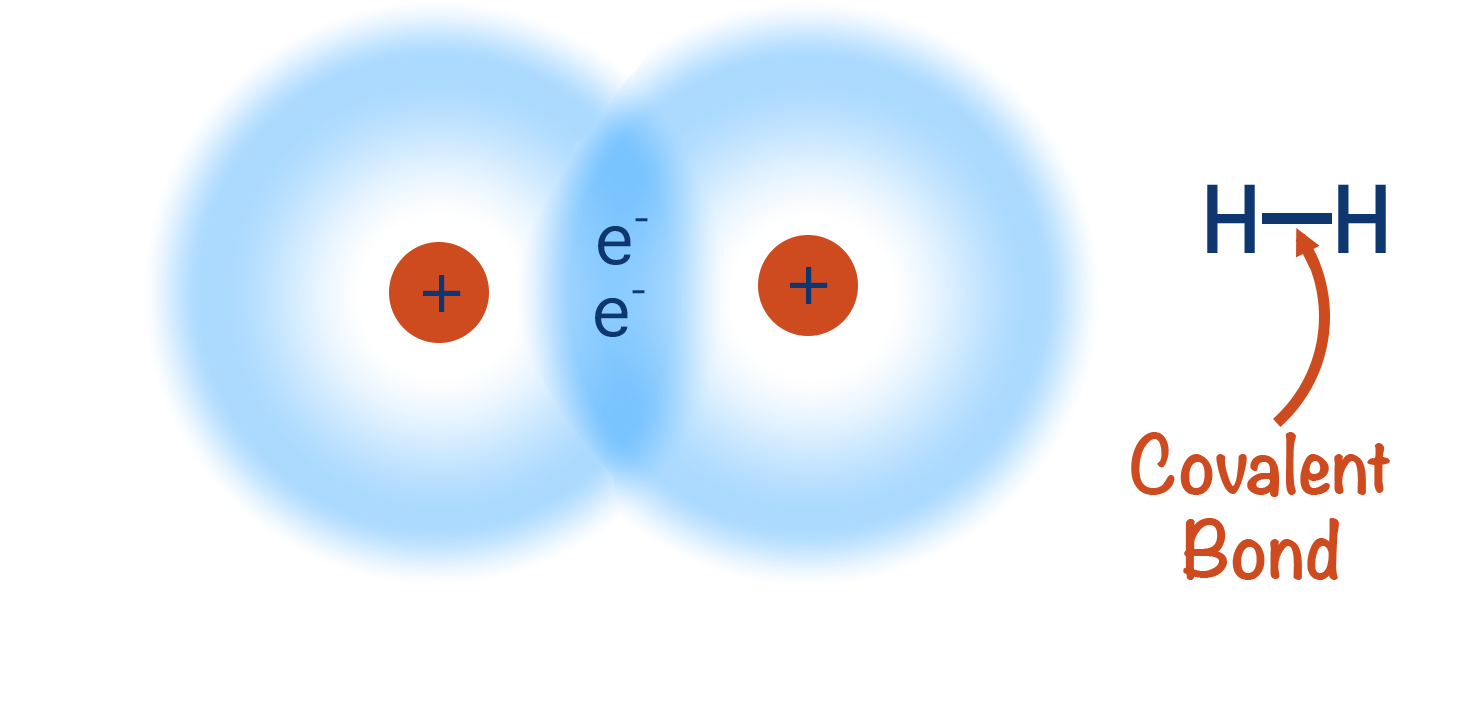
The positively charged nuclei of both hydrogen atoms get attracted to the electron density in the middle of the orbital (as this is where the electron density is highest) and this keeps the two atoms close together - this what is meant by a ‘covalent bond’. The nuclei also repel each other, meaning there is a balance between the attraction to the electron density and this repulsion, preventing the two nuclei from getting ‘too’ close. The distance between the two nuclei is called a bond length.

Further Points
Covalent bonds form between non-metal atoms. Covalent bonds between metal atoms and non-metal atoms do sometimes occur but usually, the high difference in electronegativity (see electronegativity) of the metal and non-metal means the bond ends up being ionic.
The group number (I to VIII) of an element in the periodic table tells us how many electrons an atom of that element has in its outermost shell, which can tell us how many bonds the atom can make.
For example, carbon is in group IV (4). This means carbon wants to gain access to another four electrons. Making four covalent bonds with other atoms would give the carbon atom access to a full outer shell. This is why carbon can make four bonds.
Sometimes, atoms share more than one pair of electrons. This leads to the formation of double and triple covalent bonds. See Sigma and Pi Bonding for more detail on double bonds.
We’ve launched our new site! 🎉
Course-specific notes with built-in search!
AP • A-Level (AQA • CIE • Edexcel • OCR) • IB • NCERT 11 + 12
over 750+ new pages and 3,500 images.
Visit the new homepage
