Quick Notes Intermolecular Forces
- Intermolecular forces hold molecules together in molecular substances.
- Temporary induced dipole-dipole forces (London dispersion forces) exist between all molecules.
- Unequal electron distribution around a molecule creates an instantaneous dipole that can induce a dipole on a neighbouring molecule.
- Two opposite dipoles from different molecules are attracted to each other. This creates a weak force of attraction between the two molecules.
- They are the weakest type of intermolecular force.
- The bigger the molecules, the greater the intermolecular forces that can arise between them, giving larger molecules higher melting and boiling points.
- Partial charges are shown using δ+ (partial positive charge) and δ- (partial negative charge).
Full Notes Intermolecular Forces
Intermolecular forces exist between molecules because of unequal electron distribution within molecules. Think of the covalent bond between two atoms in a molecule of hydrogen (H2).
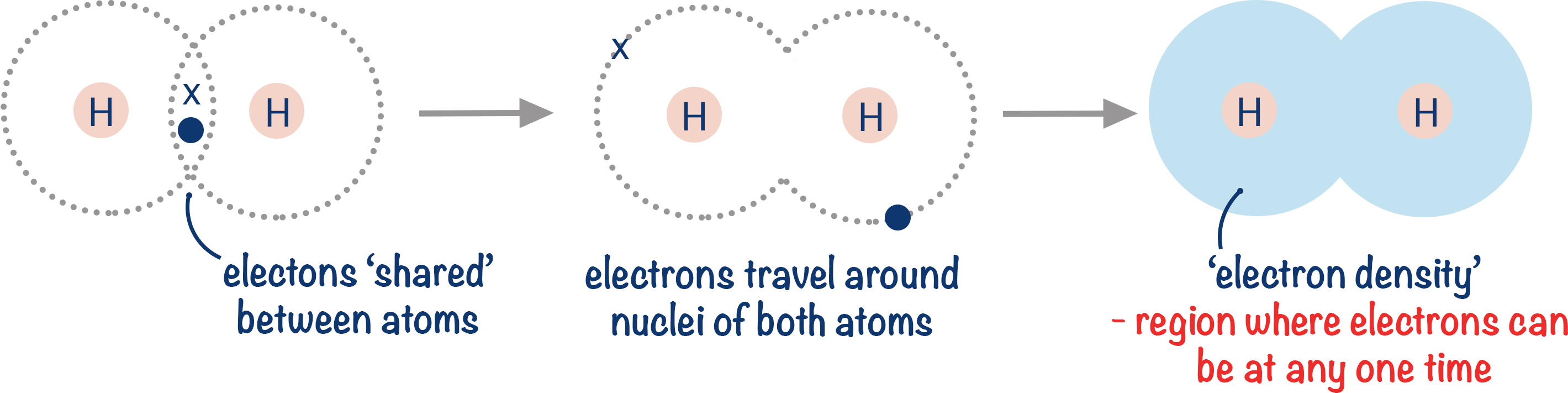
We can see electrons exist around the whole molecule. Electrons move very quickly. So, at any one time, the electrons may not be perfectly shared between the two atoms. Electrons have a negative charge. If we have more electrons on one side of the H2 molecule than the other: that side of the H2 becomes partially negatively charged, and the other side of the H2 becomes partially positively charged. Note, these charges are partial as there is still a covalent bond – one atom doesn’t actually gain an electron, it just has a greater ‘share’ of the electrons for a very short period of time.
Partial charges are shown using δ+ (partially positive) and δ- (partially negative). Partial charges are smaller than ionic charges and, at A-level, have no numerical value.
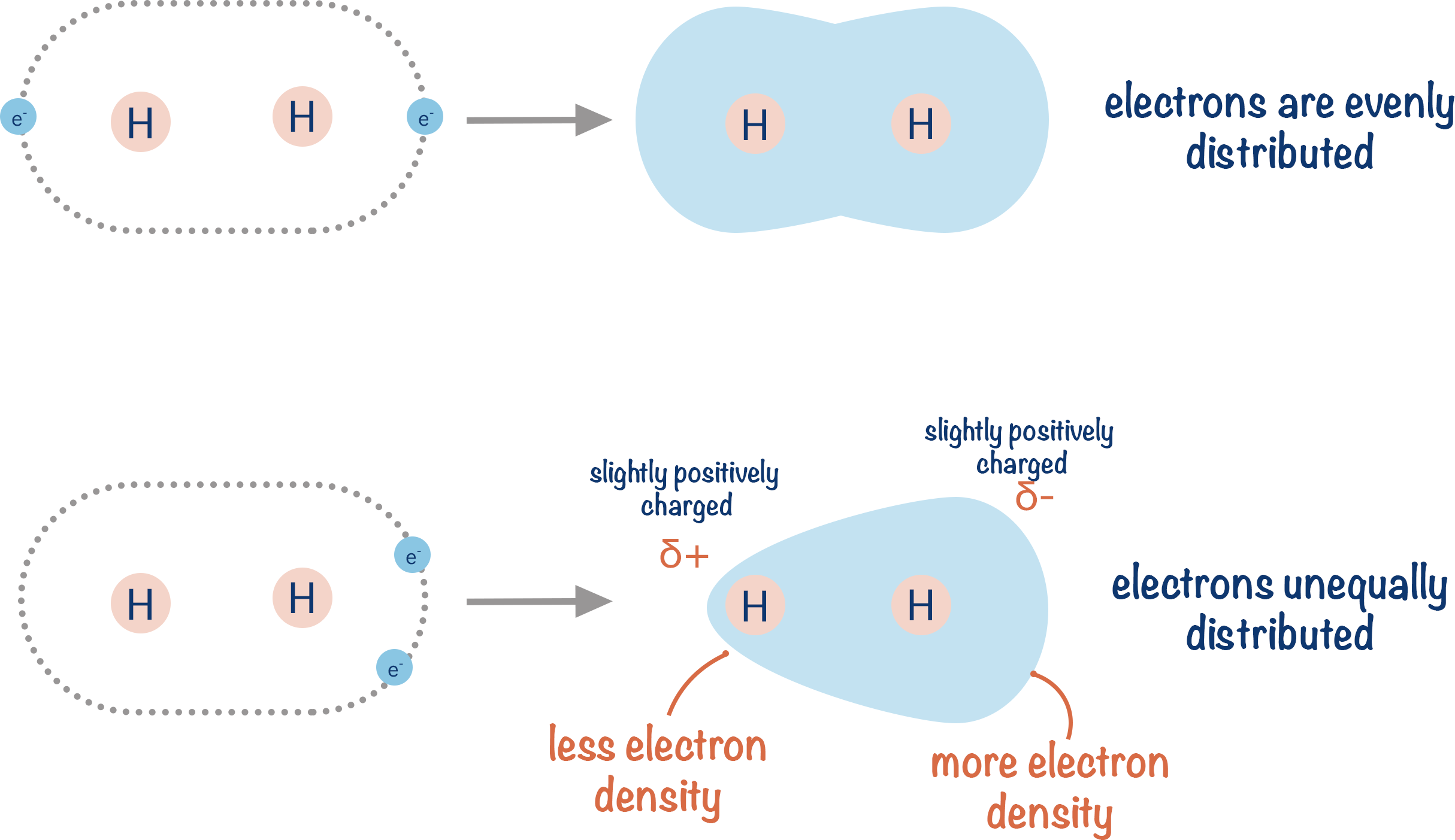
This is called a temporary dipole. It is instantaneous! This means it doesn’t exist for very long as electrons are constantly moving. But, if you have millions of molecules in a small area, you will always have large numbers of dipoles arising at any one time.
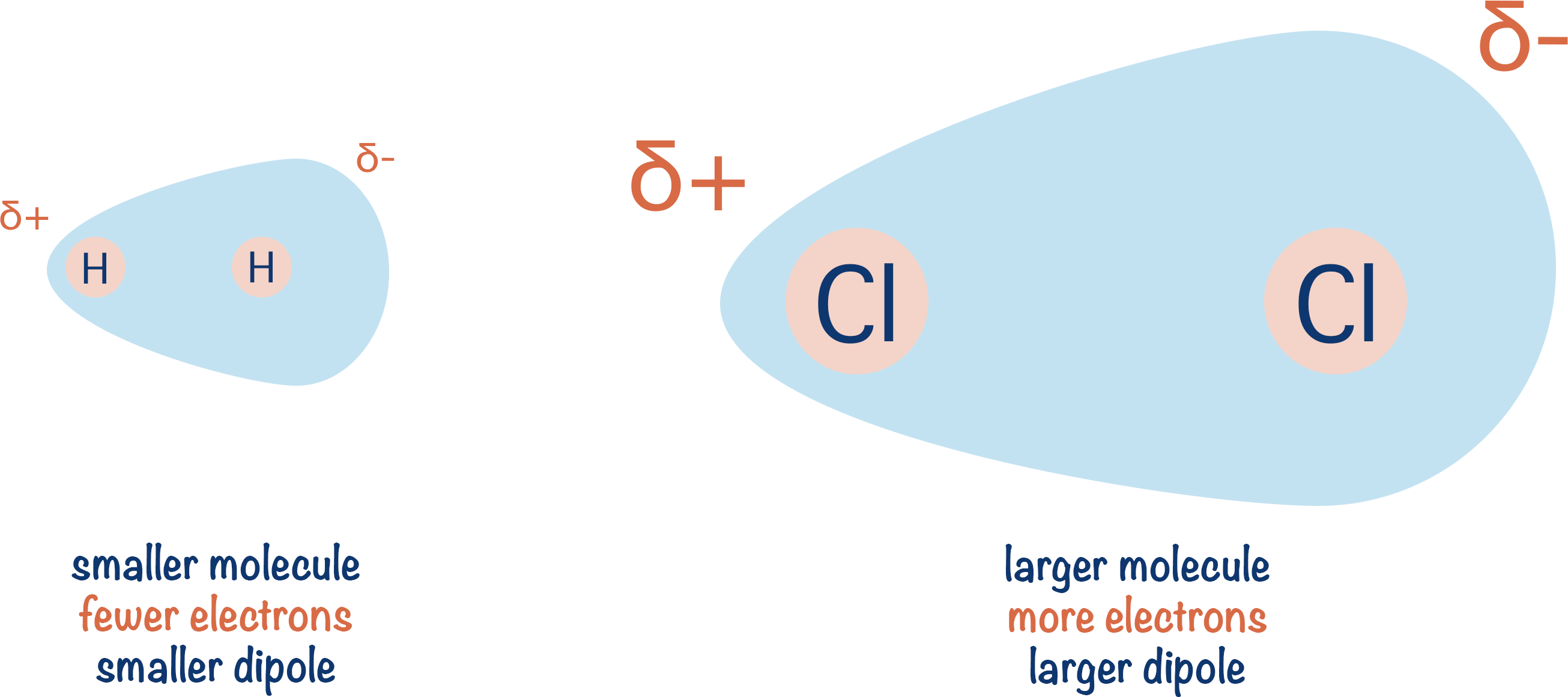
The bigger the molecule or bonding atoms, the larger the dipole because there are more electrons moving around. If more electrons move to one side than the other: a greater negative effect will be felt on that side, and a greater positive effect will be felt on the other side.
Inducing a Dipole
If a molecule with a dipole is lined-up with another molecule, it can ‘induce’ a dipole on that neighbouring molecule. This is because electrons held within the neighbouring molecule will either be attracted to the very slightly positive end of the first molecule or repelled by the very slightly negative end.
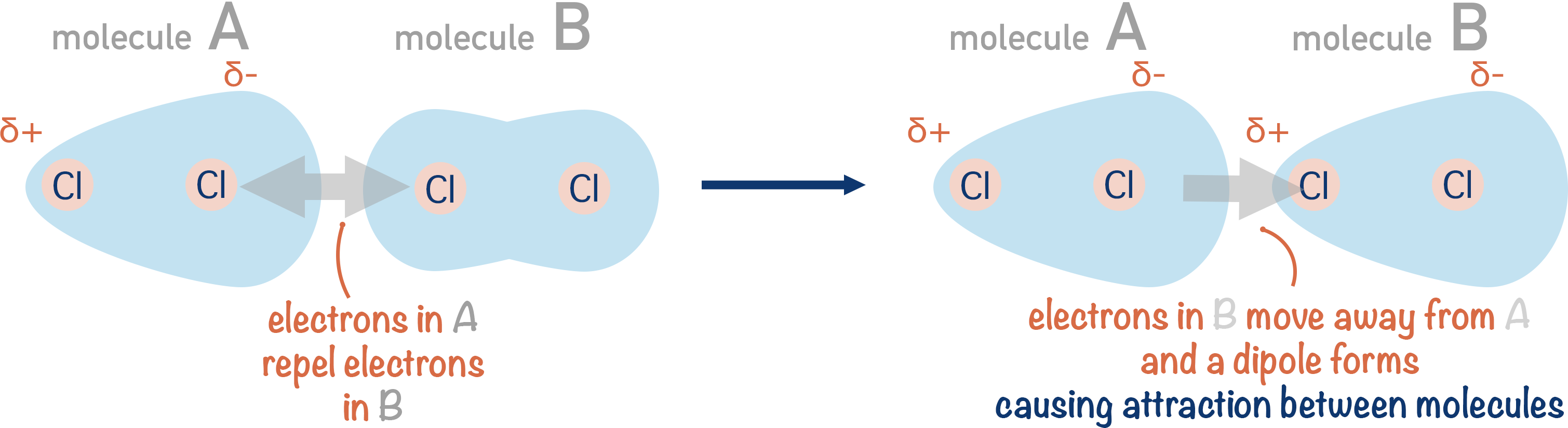
This electrostatic attraction gives rise to very weak forces between molecules. These are a type of intermolecular force, which arise between all molecules. We call these forces temporary, induced dipole-dipole interactions. Sometimes they are referred to as van der Waals or London dispersion forces.
The larger the molecules, the greater the strength of temporary, induced dipole-dipole interactions. This means larger molecules have higher melting and boiling points than smaller molecules (assuming no other intermolecular forces are present).
Are London dispersion forces van der Waals forces?
This is a common question. Van der Waals forces actually refer to all intermolecular forces that are based on attraction between opposite dipoles in molecules.
London dispersion forces are a specific type of van der Waals force. Many exam boards require you to be more specific than just stating ‘van der Waals forces’. As a result, if you are referring to the type of intermolecular force covered here: refer to ‘London dispersion forces’ or ‘temporary, induced dipole-dipole forces’ (depending on your specification).
