Video Tutorial Ionic Bonding
Quick Notes Ionic Bonding
- Ionic bonding describes the electrostatic attraction that holds oppositely charged ions together.
- The strength of an ionic bond is based on the charge and size of the ions involved:
- Greater the charge = stronger the bond.
- Smaller the ionic radius = stronger the bond
- To get a stable electron configuration, atoms lose or gain electrons.
- Metals lose electrons and become positively charged ions.
- Non-metals (usually) gain electrons and become negatively charged ions.
- Oppositely charged ions arrange themselves in a solid to maximise attraction of opposite charges and minimise repulsion to like charges, forming a repeating lattice structure.
Full Notes Ionic Bonding
The most stable configuration for most atoms is to have eight electrons in their outermost shell (octet rule). Note, transition metals are an exception to this, (see Transition Metals). The reactivity of an element can often be linked to how easily an atom can ‘get’ to having eight electrons in its outermost shell. This explains why the noble gases (group VIII in the periodic table) are so unreactive, as they do not want to change their electron number.
To get a full outer shell, atoms can lose or gain electrons and become charged ions.
Electrons are negatively charged (e-). So, if an atom loses an electron, it becomes a positively charged ion; if an atom gains an electron, it becomes a negatively charged ion. Oppositely charged ions attract each other and form ionic compounds (compounds made of ions).
A common example is the reaction between sodium and chlorine.
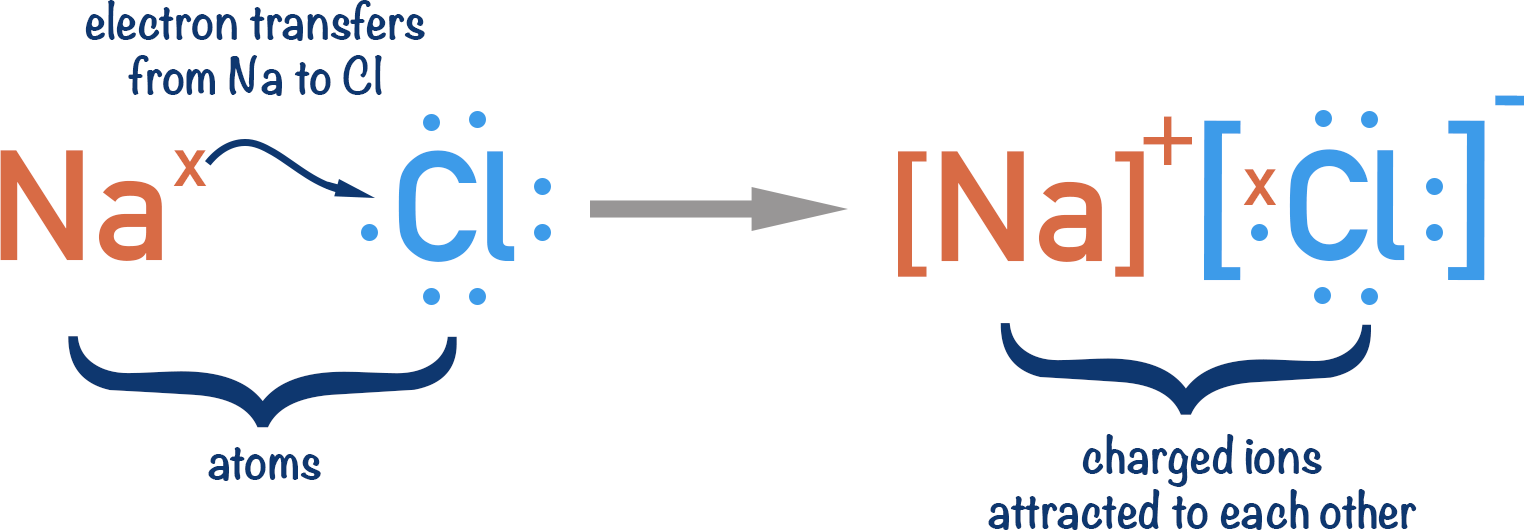
Sodium has one electron in its outer shell and chlorine has seven electrons in its outer shell. In order for both sodium and chlorine to have full outer shells, sodium ‘gives’ an electron to chlorine.
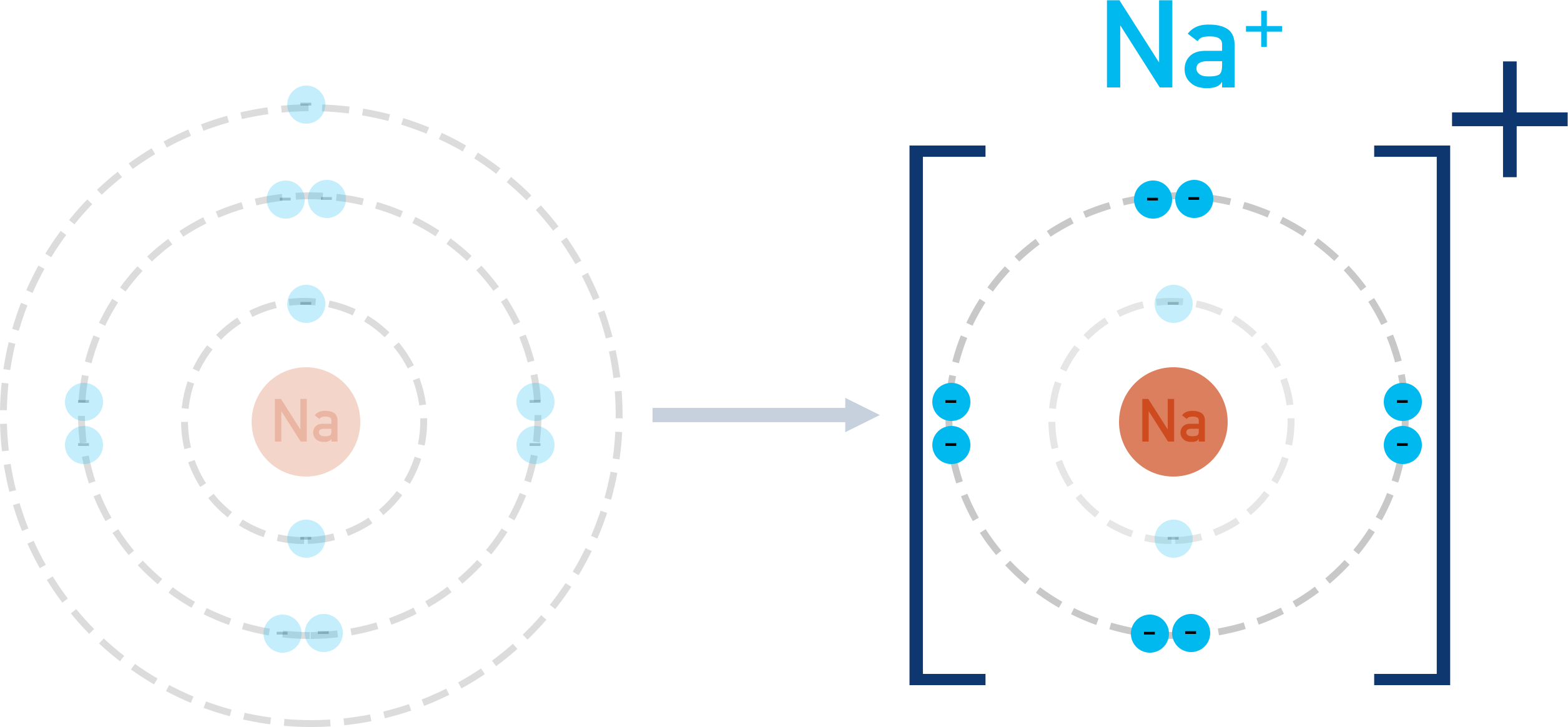
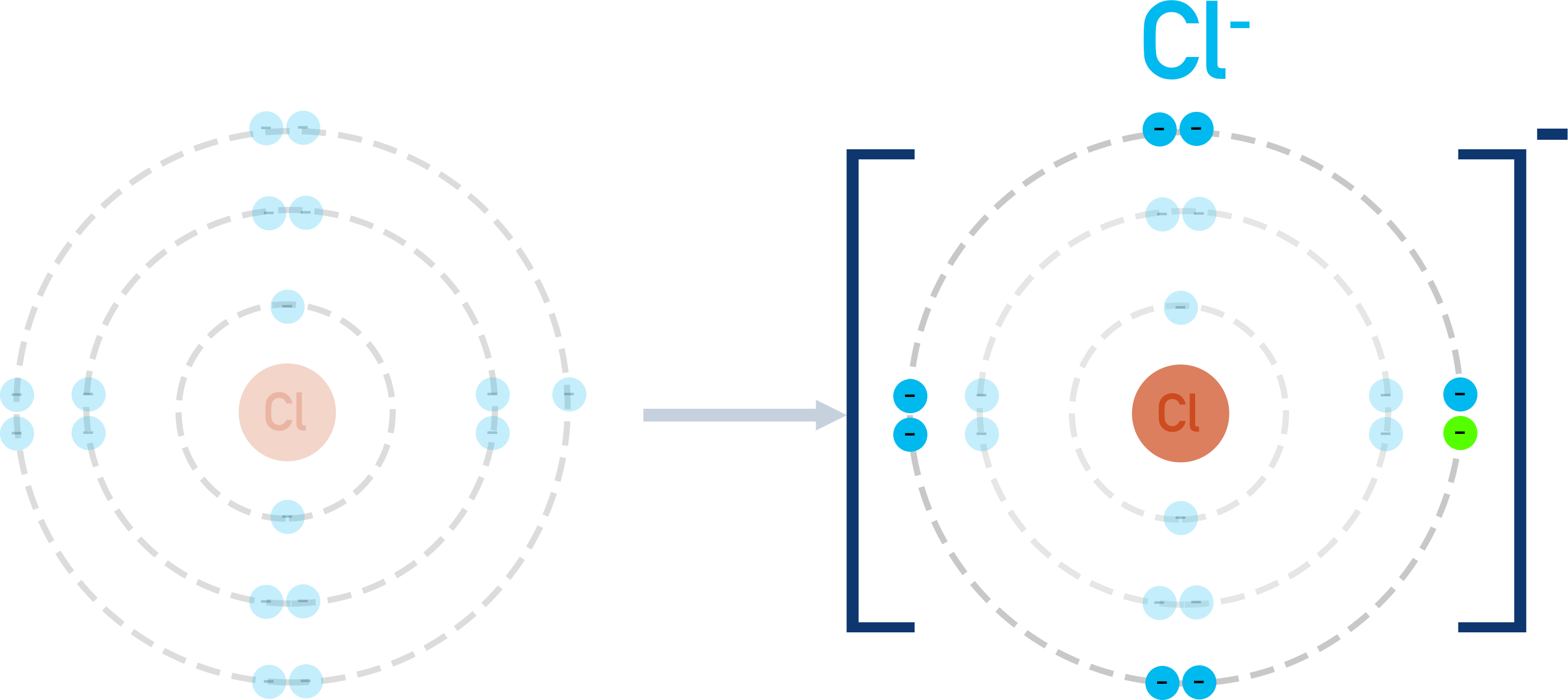
In the process, sodium becomes a positively charged ion (because it has lost a negatively charged electron); chlorine becomes a negatively charged ion (because it has gained a negatively charged electron).
Positively charged and negatively charged ions are attracted to each other (electrostatic attraction). With the above example, the positive sodium ion is attracted to the negative chloride ion. Unless given enough energy to overcome this attraction, the ions move towards each other, forming the compound ‘NaCl’.
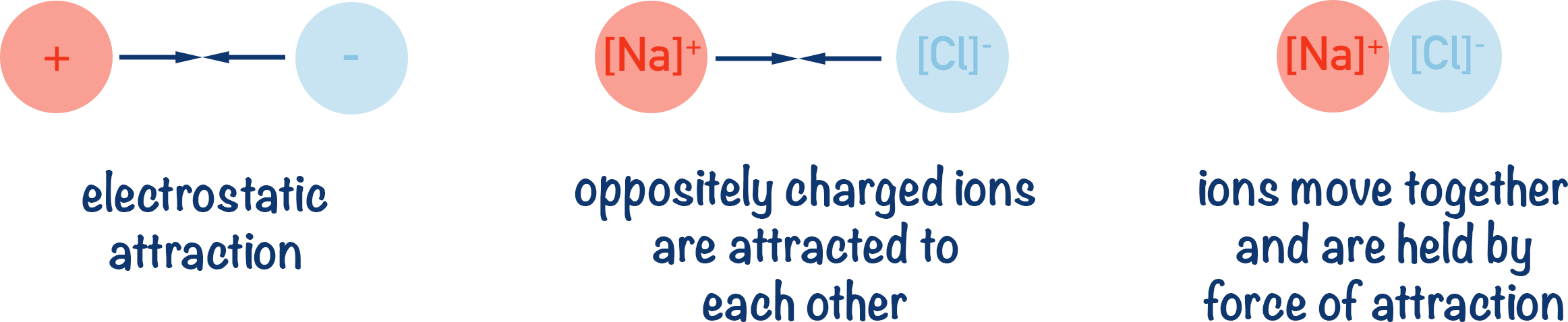
The bonding between the oppositely charged ions is called ‘ionic bonding’.
The stronger the ionic charge (the charge on the ion), the stronger the ionic bonding.
The group number (I to VIII) in the periodic table tells us how many electrons an atom needs to lose or gain to get to a full outer shell, so it can be used to predict the charge of the ion formed.
Ionic Bond Strength
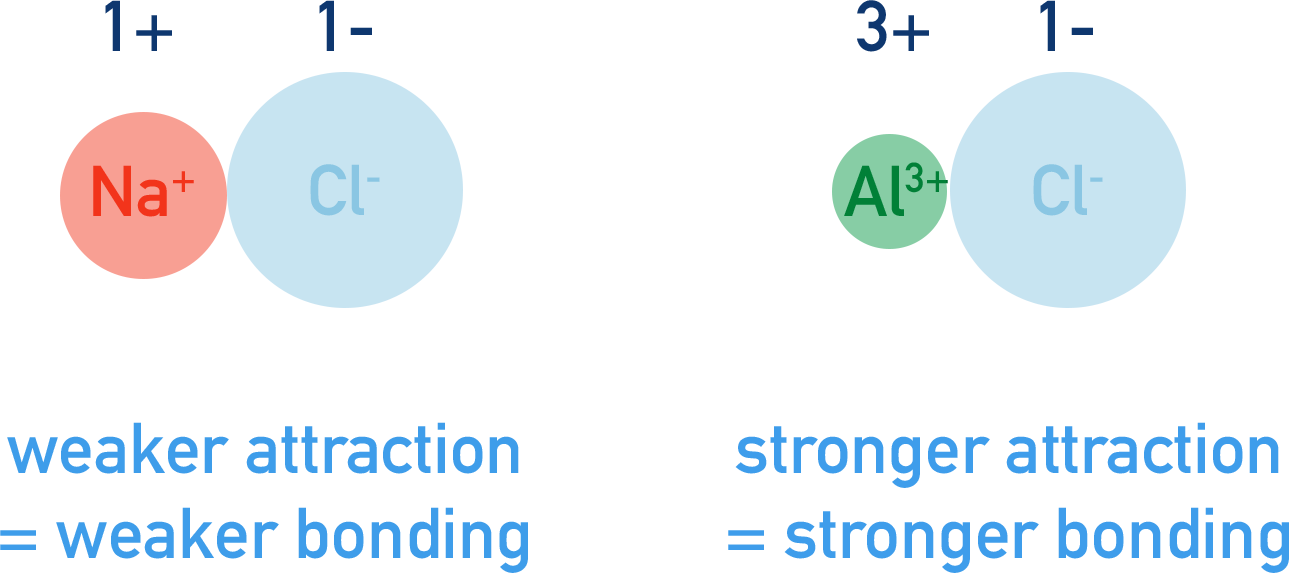
For ionic bonding, the greater the charge of an ion and the smaller its ionic radius, the stronger the attraction it has to oppositely charged ions.
The strength of an ionic bond is based on the attraction between the ions forming the bond. Stronger forces of attraction mean a stronger bond as more energy is needed to pull the ions apart and overcome the attraction between them.
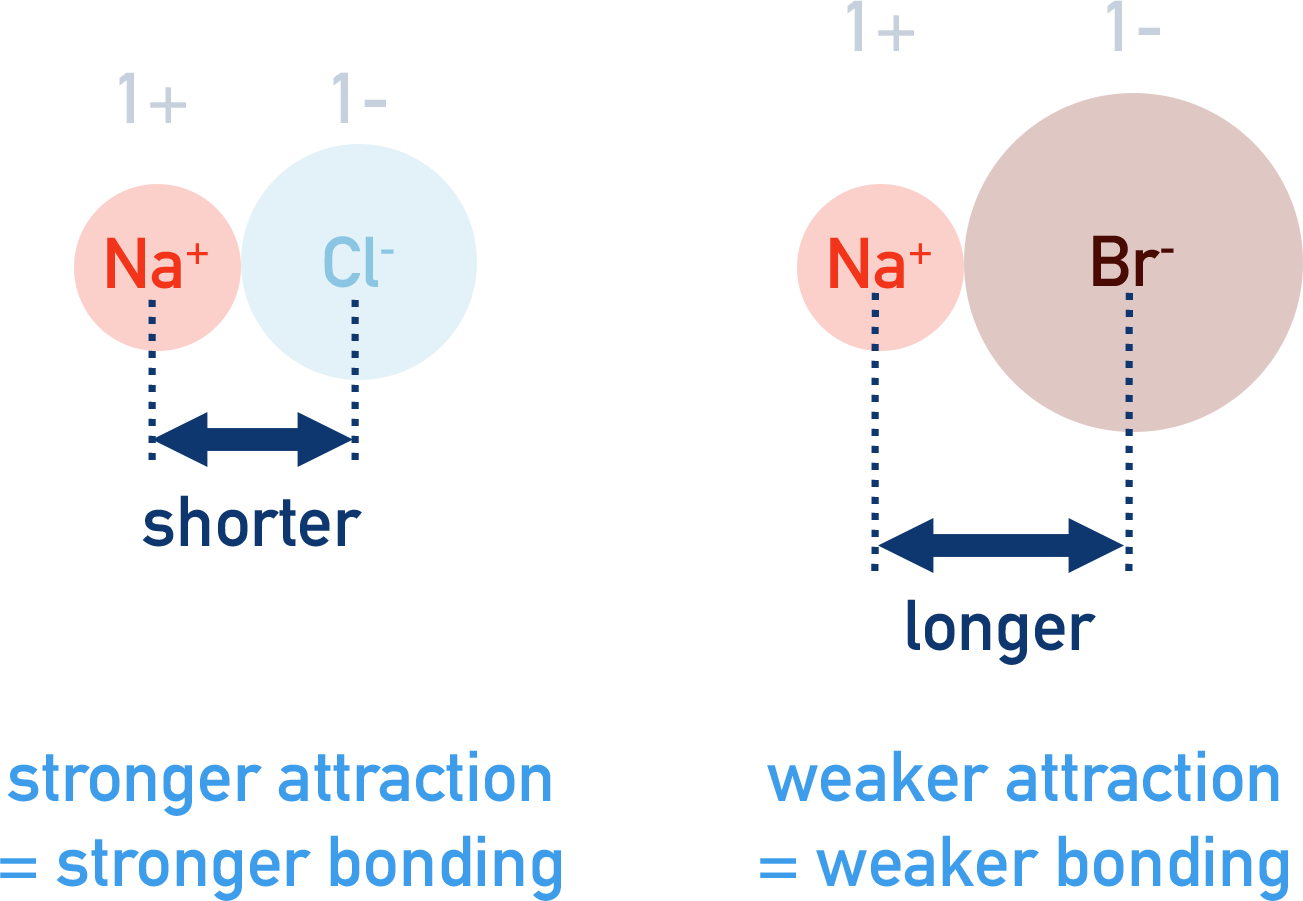
Note, the smaller the ionic radius, the shorter the 'distance' between opposite charges meaning a stronger force of attraction.
For example..
Sodium ions have a charge of 1+ and aluminium ions have a charge of 3+. When sodium and aluminium ions bond with chloride ions (Cl-), the force of attraction between the aluminium and chloride ions will be much stronger than between the sodium and chloride ions. This is because of the greater charge of the aluminium ions.
Chloride (Cl-) ions are smaller than bromide (Br-) ions giving them a smaller ionic radius. When sodium (Na+) ions bond with chloride and bromide ions, the strength of attraction between Na+ and Cl- will be greater than between Na+ and Br- as there is less distance between the 'centre' of each ion, giving stronger attraction and leading to stronger bonding.
Ionic Lattice
The strength of attraction between oppositely charged ions is strong.
There are also forces of repulsion between like charges. Two positively charged ions, for example, will repel each other and there will be force 'pushing' them apart.
Unless a group of ions are given enough energy to overcome these forces, they will end up arranging themselves to maximise attraction between the opposite charges and minimise the repulsion between the same charges. This leads to the formation of regular, repeating structures called ionic lattices.
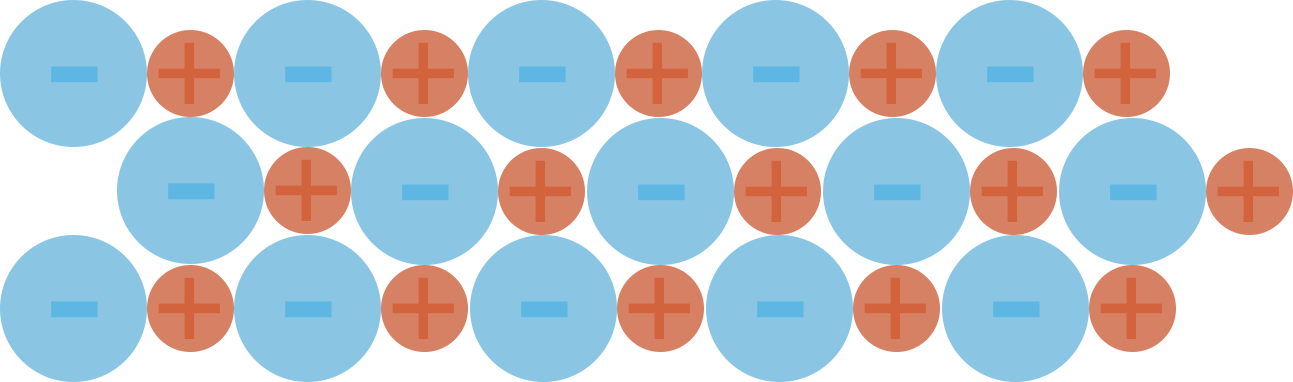
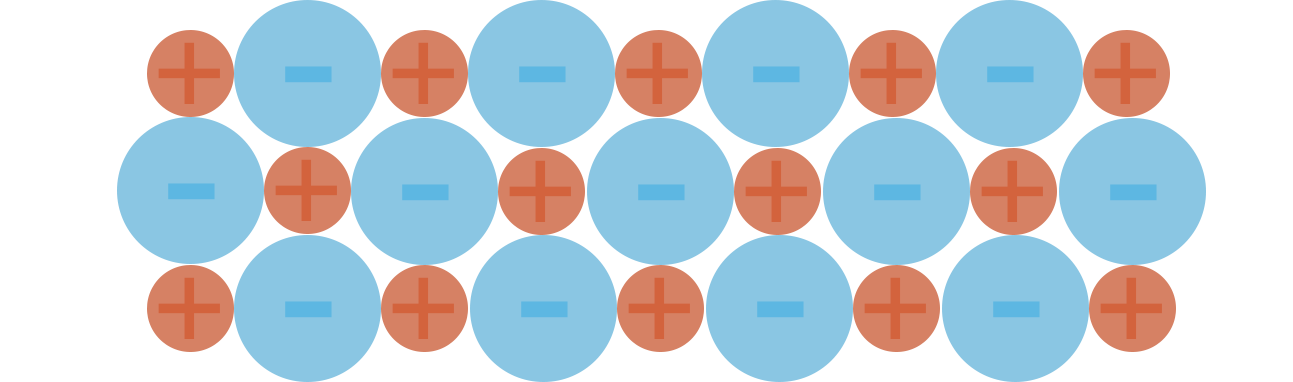
Ionic Lattice Example (2D)
For example...
With sodium chloride, the positive sodium ions want to be as far away from each other as possible and the negative chloride ions also want to be as far away from each other as possible.
To achieve this, the ions arrange themselves in a regular pattern.
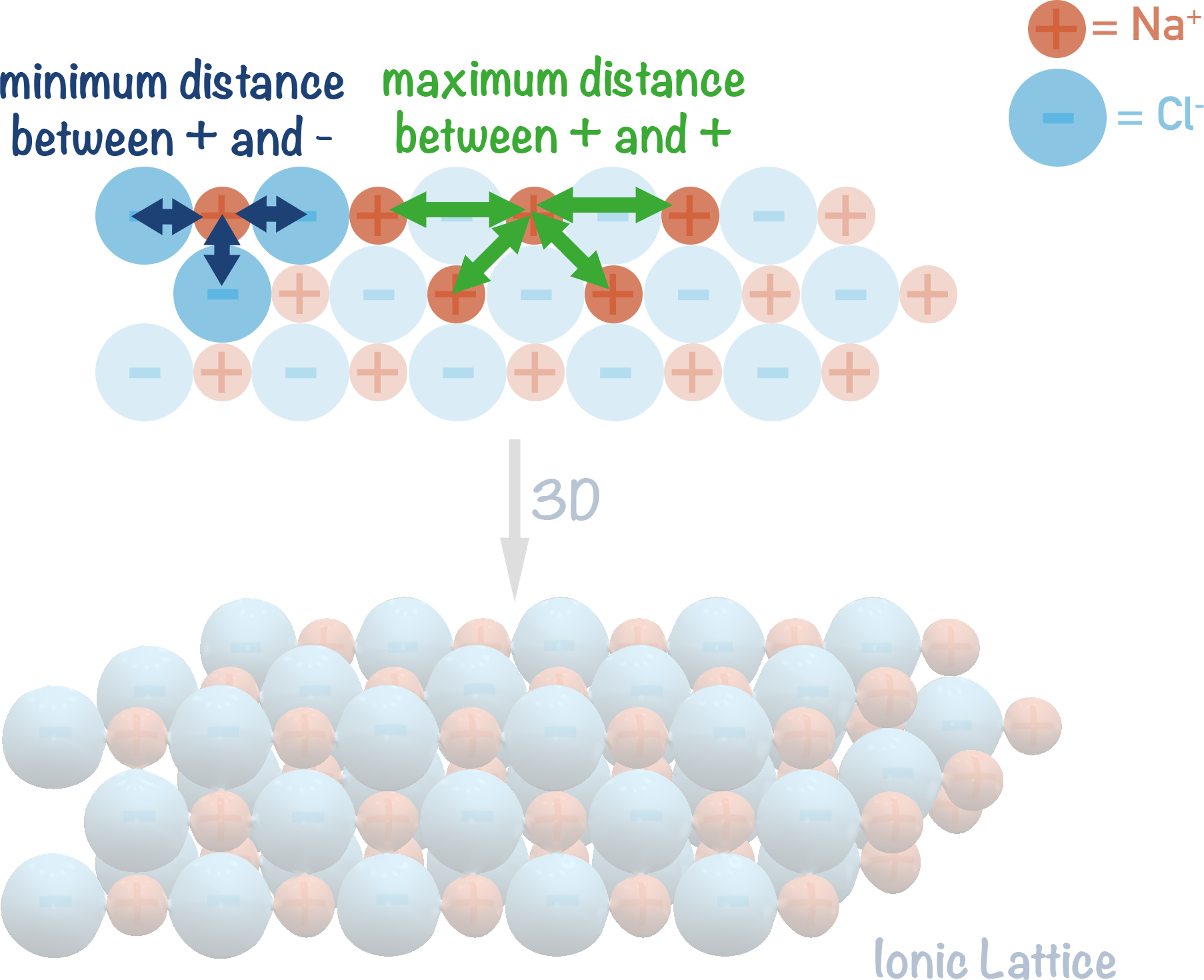
Lattice structures are three dimensional, meaning each ion is attracted to several other ions in the structure, making the ionic lattice very strong and hard to break. This is why ionic compounds usually have very high melting and boiling points. The charged ions within a lattice are also in 'fixed' positions and unable to move freely, meaning they are unable to conduct electricity. As a lattice melts, the charged ions within it are able to move and this gives them the ability to conduct electricity through the compound. This is why ionic compounds are able to conduct electricity in molten state but unable to as solids.
We’ve launched our new site! 🎉
Course-specific notes with built-in search!
AP • A-Level (AQA • CIE • Edexcel • OCR) • IB • NCERT 11 + 12
over 750+ new pages and 3,500 images.
Visit the new homepage
