Video Tutorial Metallic Bonding
Quick Notes Metallic Bonding
- In metallic bonding, positive metal ions are attracted to negatively charged delocalised electrons, forming a dense and rigid structure.
- Positively charged metal ions form because outermost electrons are held weakly by the metal atoms nucleus and are able to drift out of their orbitals, becoming delocalised and leaving a positively charged metal ion behind.
- Delocalised electrons from metal atoms have 'nowhere to go' and stay attracted to the positive charge of metal ions.
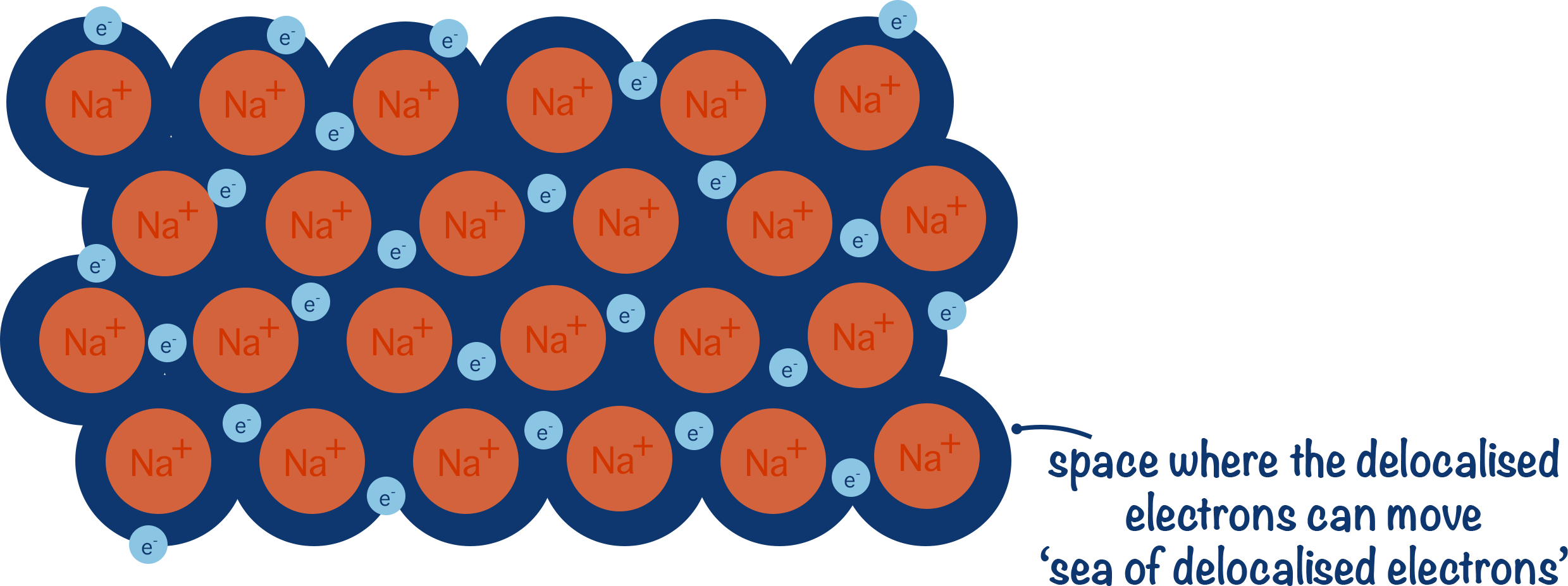
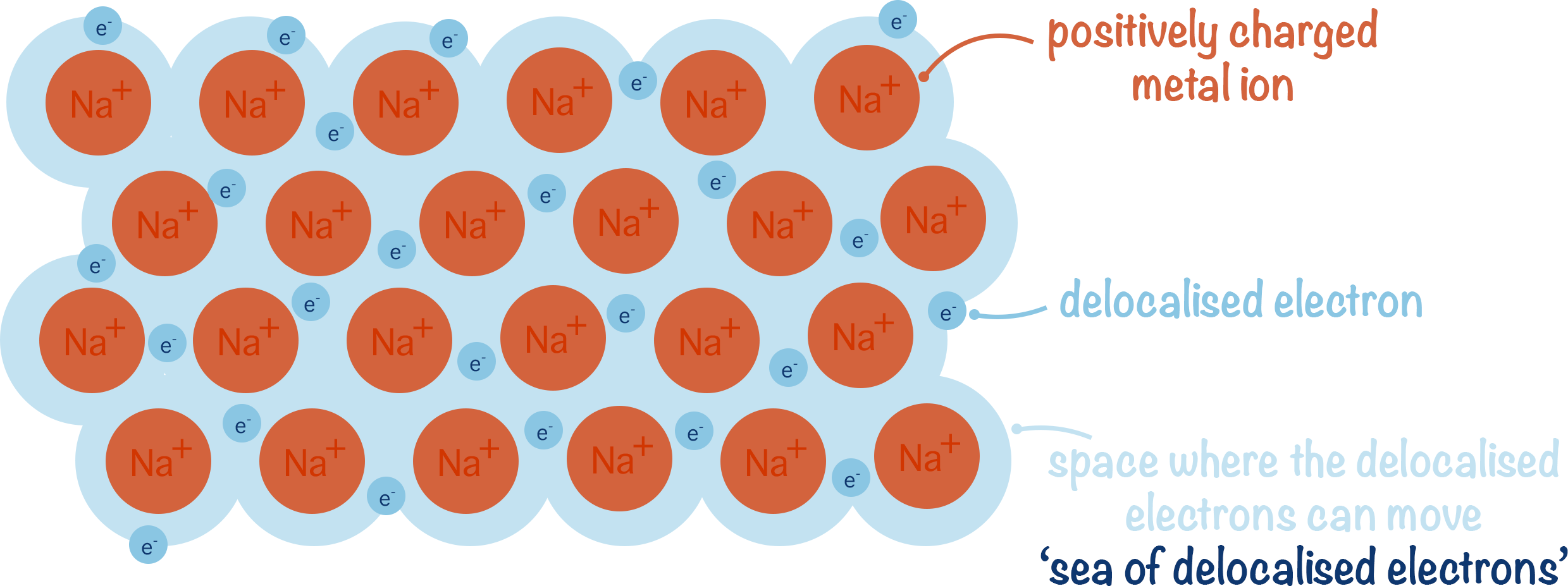
- This creates a 'sea of delocalised electrons' between metal ions.
- Free movement of delocalised electrons enable metals to conduct electricity and heat easily.
- Delocalised electrons from metal atoms have 'nowhere to go' and stay attracted to the positive charge of metal ions.
Full Notes Metallic Bonding
It is seems obvious, but when talking about metallic bonding, we are talking about a specific type of atomic bonding that only occurs in metals.
Metal atoms easily form positive ions as their outermost electrons have a weak attraction to the atoms nucleus. This weak attraction means they can be easily 'pulled' away from the atom, leaving a positively charged metal ion behind.
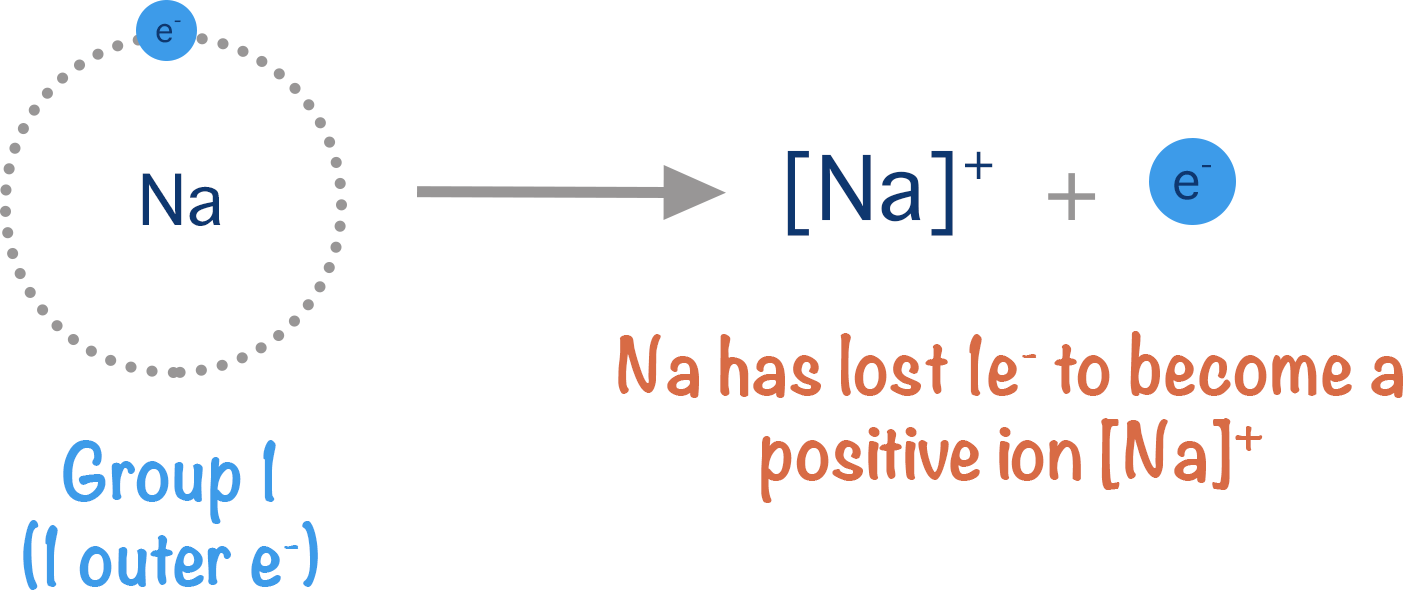
In metallic bonding, metal atoms don't actually ‘lose’ electrons to become positive ions as there is nothing else available to 'take' the electrons.
Instead, due to their weak attraction to the atoms nucleus, the outermost electrons drift away from the metal atoms, temporarily leaving their atomic orbitals. They are now free to move in a much larger region of space and are described as delocalised. As they have a negative charge and the metal ion left behind has a positive charge, there remains attraction between the delocalised electrons and the metal ion meaning the delocalsied electrons remain close to the ions.
In a pure metal, there are lots of metal atoms all with this arrangement of delocalised electrons. The delocalised electrons from one atom get attracted to other positively charged metal ions and at the same time positively charged metal ions present are attracted to the delocalised electrons , pulling the ions close together.
How does metallic bonding form?
Outer electron from metal atom has a weak attraction to the nucleus and 'drifts' out of its orbital.
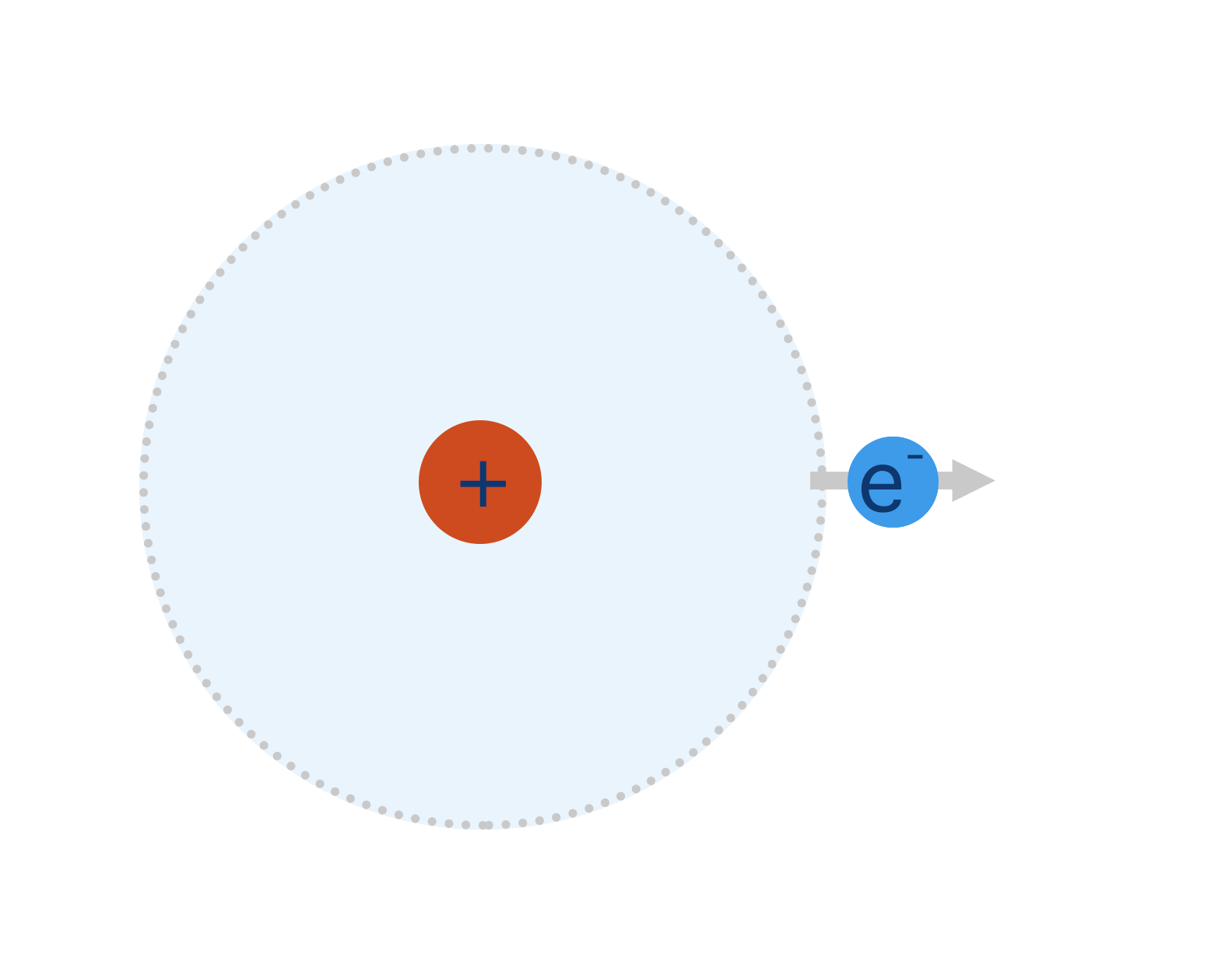
The electron is no longer forced to stay in its orbital shape and can move around in a much larger area and is described as being delocaised.
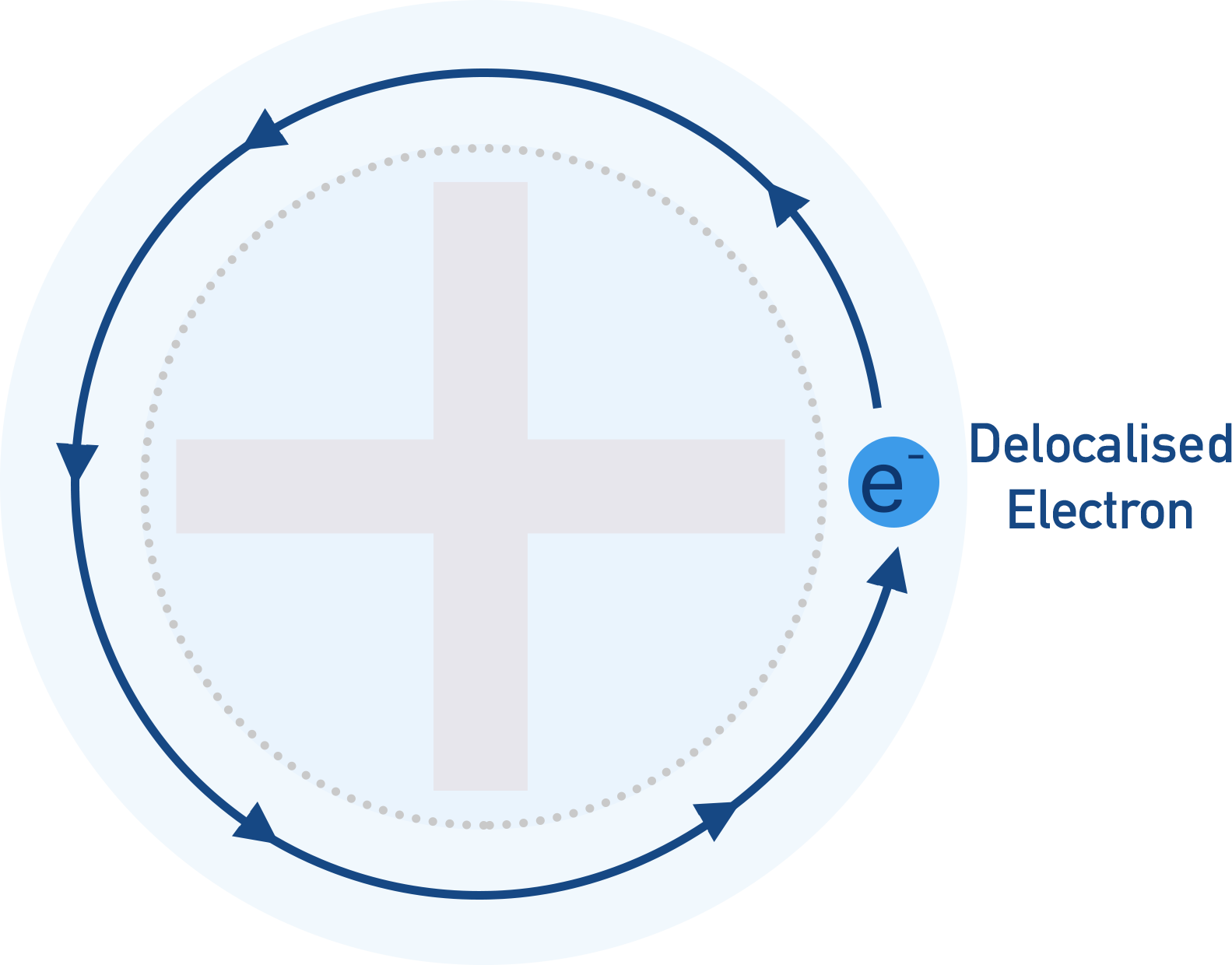
The negative charge of the delocalised electron and the positive charge of the metal ion left behind means there is a force of attraction that keeps the delocalsied electron close to the ion.
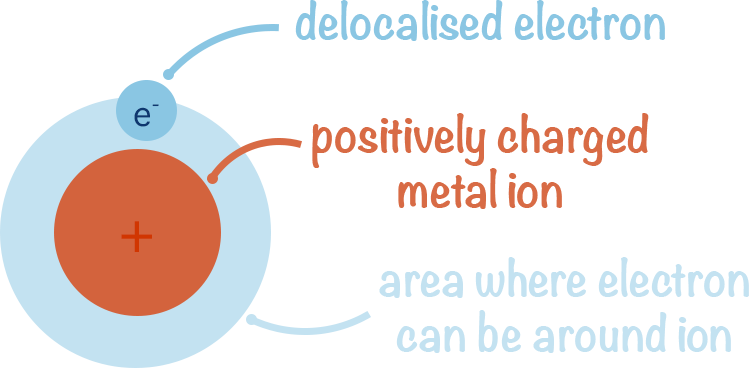
In a pure metal, there are lots of delocalised electrons and metal ions. The positively charged ions attract delocalised electrons from other atoms and this means the electrons can 'flow' between metal ions and temporarily join their atomic orbitals before then being attracted into another ions orbital.
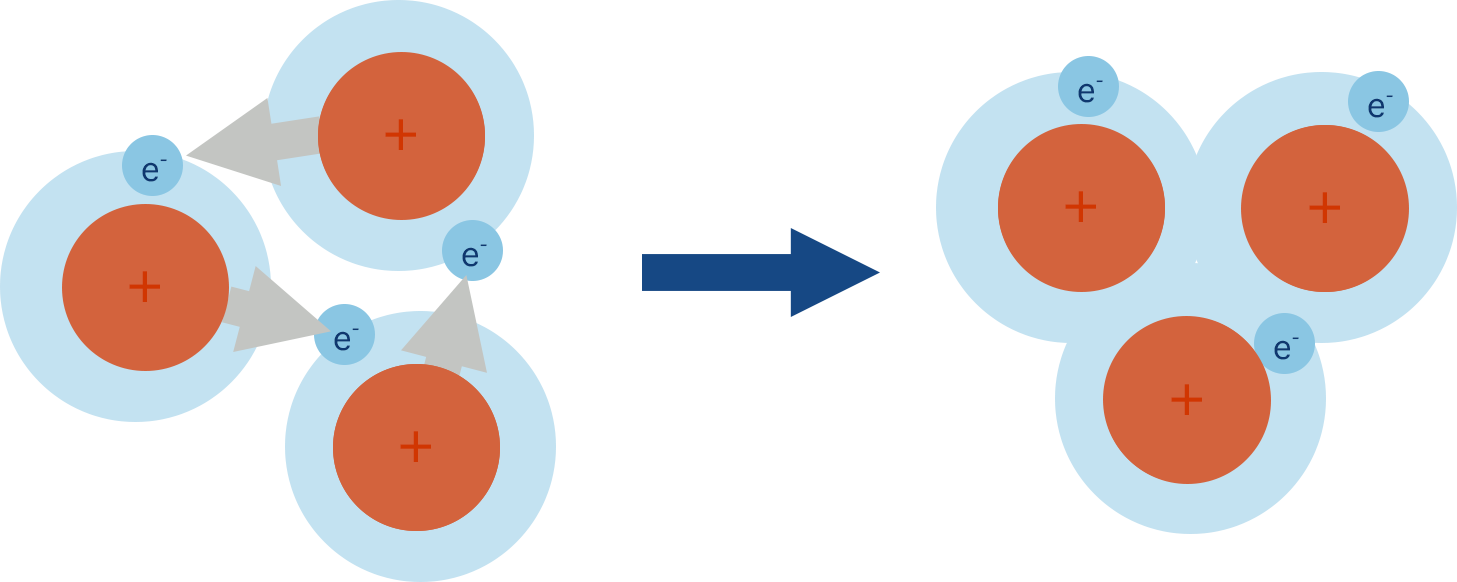
This arrangement pulls in the ions in the metal together and creates a dense, tightly packed lattice structure.
Metallic Bonding Strength
The strength of bonding in a metal is based on the forces of attraction between the metal ions and delocalised electrons present.
To break apart a metallic structure, these forces of attraction need to be overcome. The stronger the forces of attraction, the harder it is to break the structure and the stronger the metallic bonding.
The strength of attraction will be based on two things:
The greater the charge of the ion, the stronger the forces of attraction, giving stronger metallic bonding. Note also that a greater charge of ion means more delocalised electrons present, further increasing attraction forces.
The smaller the metal ions, the stronger the forces of attraction as there is less distance between the positive charge (nucleus of metal ion) and negative charge (delocalised electrons).
For example...
Sodium ions (Na+) are larger and have a smaller charge than aluminium ions (Al3+).
As a result, in a sample of sodium there will be weaker attraction between the positive metal ions and delocalised electrons compared to in a sample of aluminium.
This leads to stronger metallic bonding in aluminium and explains why alumninium is more dense (tightly packed) and stronger as a solid than sodium.
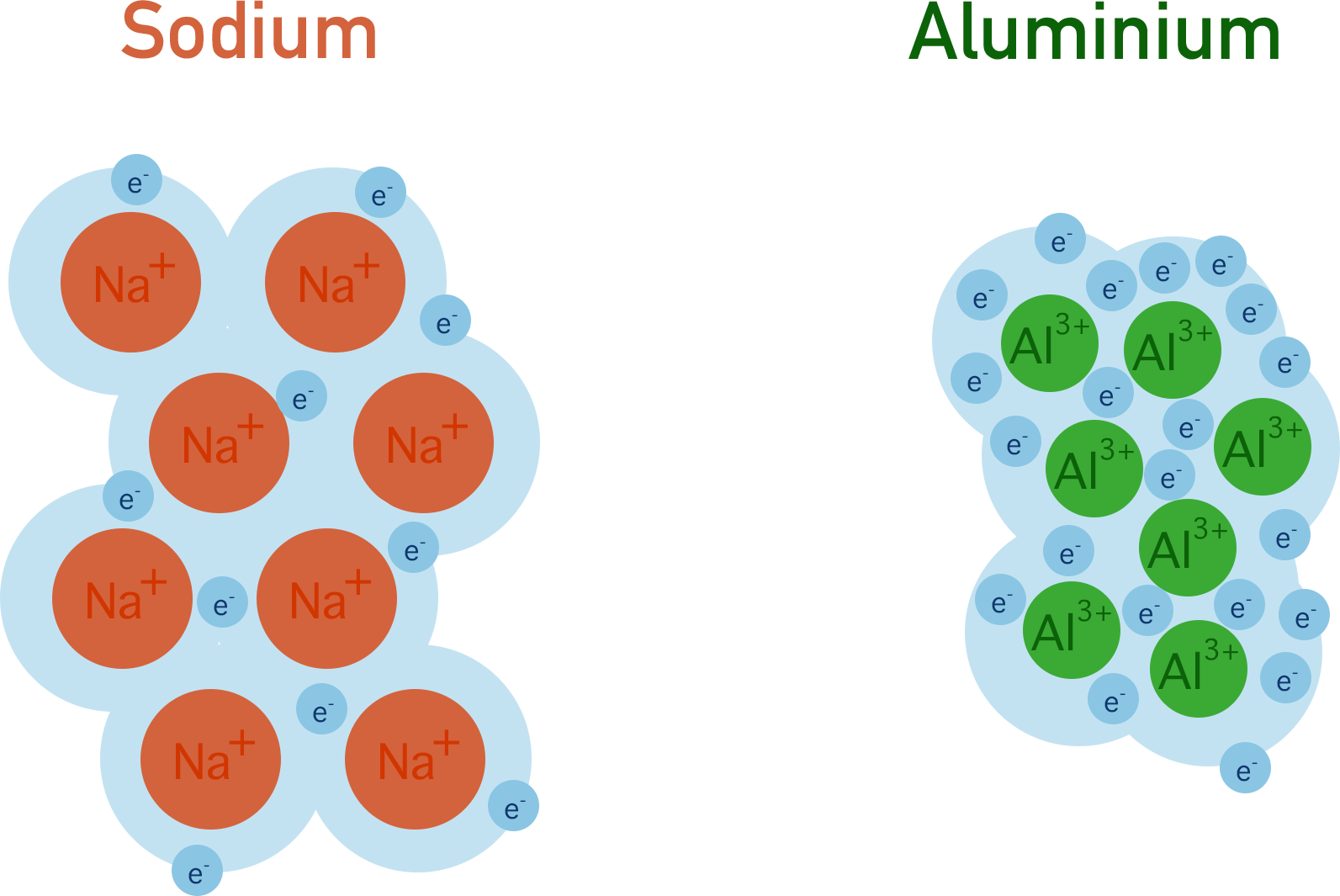
The melting point of sodium is 97.7oC and the melting point of aluminium is 660.3oC. This difference can be explained by the stronger metallic bonding in aluminium compared to in sodium. More energy is needed to overcome the forces of attraction in aluminium, giving a higher melting point.
Metallic Bonding Properties
The arrangement of ions and presence of delocalised electrons in metallic structures give metals some interesting properties. Metals are good conductors of electricity and heat.
As delocalised electrons in a metal are free to move, they can be forced to move in a particular direction, enabling an electrical current to move through the metal.
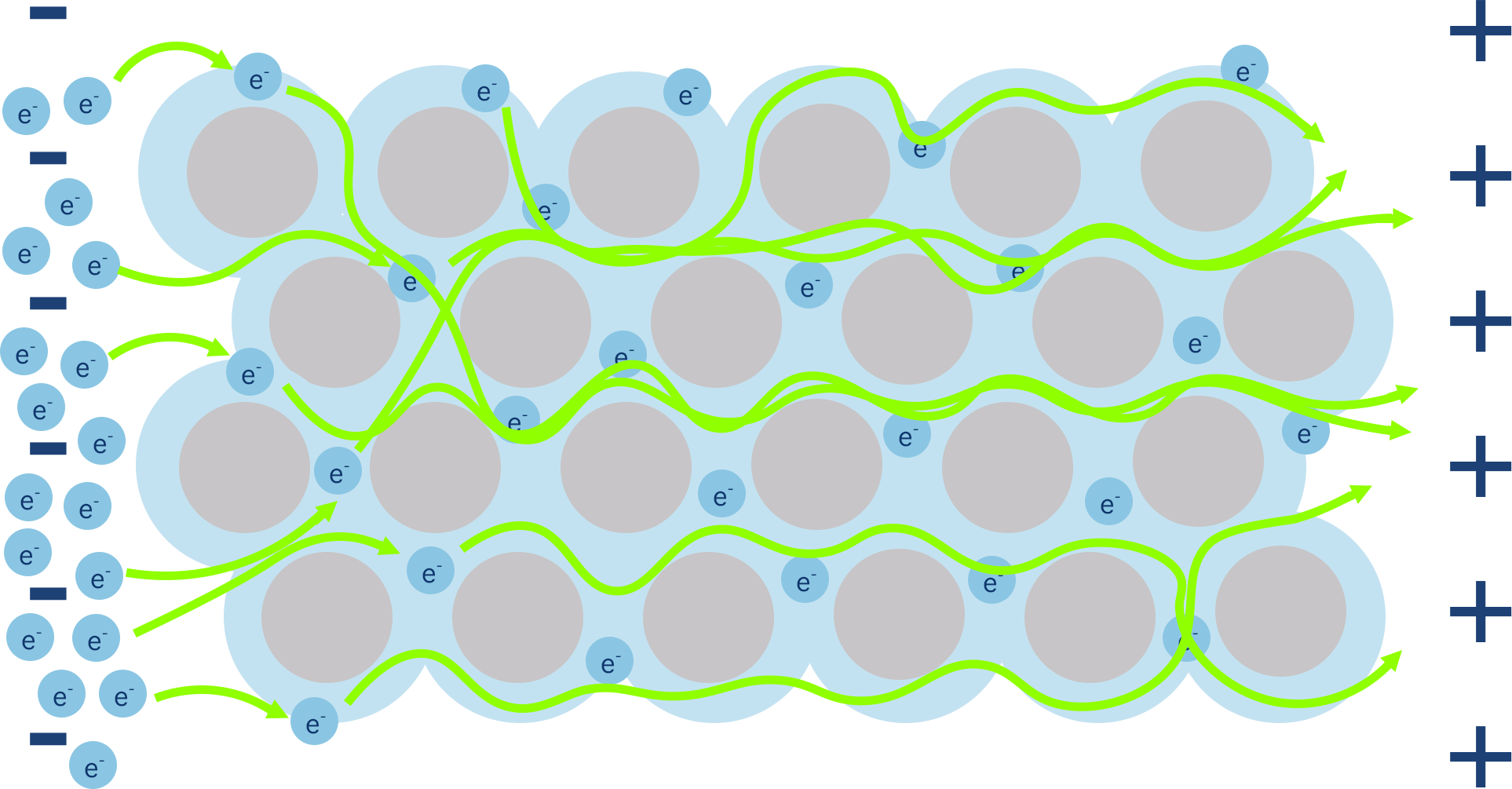
The presence of delocalised electrons also enables heat to move through a solid metal very quickly - the electrons are free to 'move' the extra energy through the solid as it is heated.
Metallic bonding is very strong, which is why metals have high melting points and are used for strength in applications.
The greater the charge of the metal ion, the stronger the metallic bonding. In aluminium, for example, each aluminium atom forms an Al3+ ion. Even though each aluminium ion has a low mass (Al has an Ar of 27), the attraction between the 3+ ions and the delocalised electrons is very strong, giving aluminium a very high strength to weight ratio.
We’ve launched our new site! 🎉
Course-specific notes with built-in search!
AP • A-Level (AQA • CIE • Edexcel • OCR) • IB • NCERT 11 + 12
over 750+ new pages and 3,500 images.
Visit the new homepage
