Video Tutorial Hydrogen Bonding
Quick Notes Hydrogen Bonding
- Hydrogen bonds are a specific type of intermolecular force.
- Hydrogen bonds exist between molecules that have an N-H, O-H or F-H bond.
- Nitrogen, oxygen and fluorine are highly electronegative elements, so covalent bonds between atoms of them and hydrogen are highly polar.
- A lone pair of electrons from a partially negative nitrogen, oxygen or fluorine gets attracted to a partially positive hydrogen atom from the same bond type in another molecule.
- Substances that can form hydrogen bonds have higher melting points than similar-sized substances that are unable to.
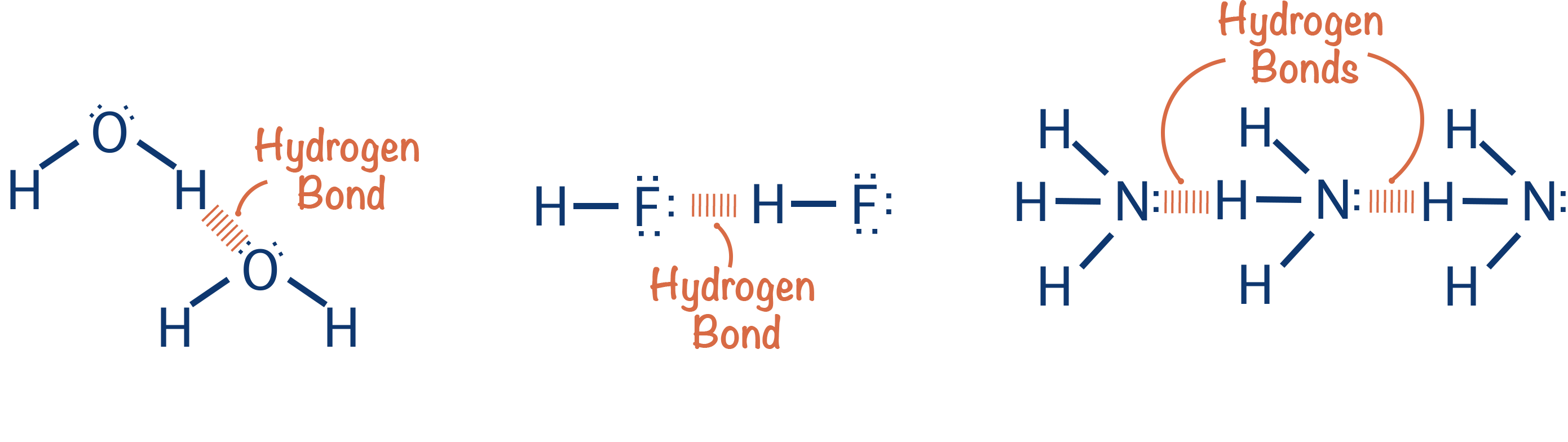
Full Notes Hydrogen Bonding
Hydrogen bonding describes a type of permanent dipole-dipole, intermolecular force.
Nitrogen, oxygen and fluorine are highly electronegative elements whereas hydrogen has a very low electronegativity (see Electronegativity). When a hydrogen atom covalently bonds with a nitrogen, oxygen or fluorine atom, the bond formed is highly polar.
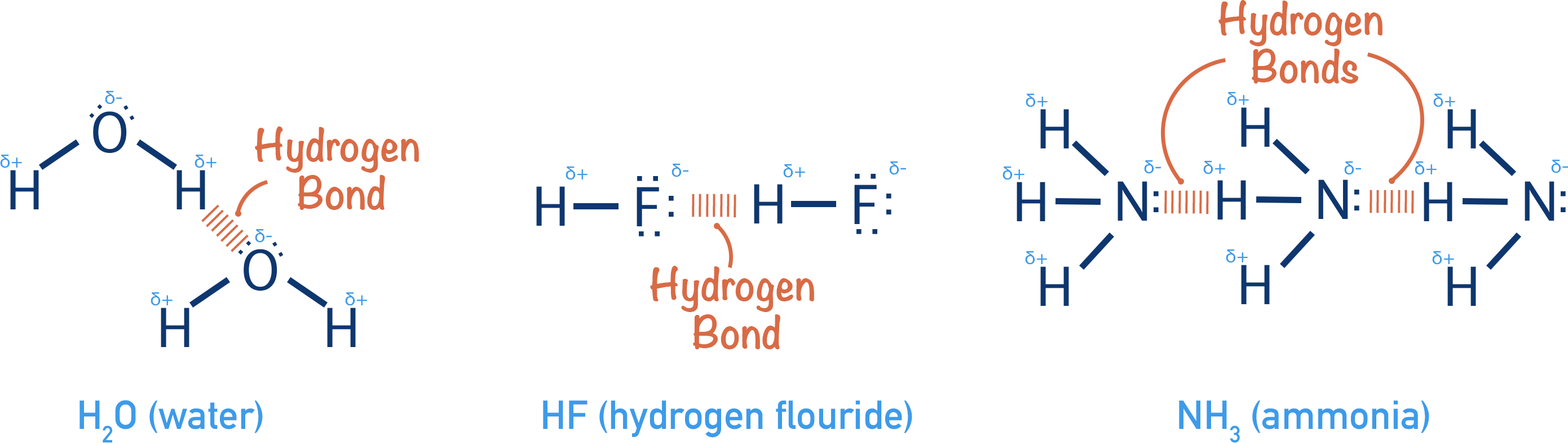
This high polarity gives rise to permanent dipole-dipole forces between partially negative nitrogen, oxygen or fluorine atoms from one molecule and partially positive hydrogen atoms from a neighbouring molecule. These forces are called hydrogen bonds.
Hydrogen bonds only occur between molecules that contain nitrogen-hydrogen (N-H), oxygen-hydrogen (O-H) or fluorine-hydrogen (F-H) bonds.
How do hydrogen bonds form?
A hydrogen atom is made up of just one electron and one proton.
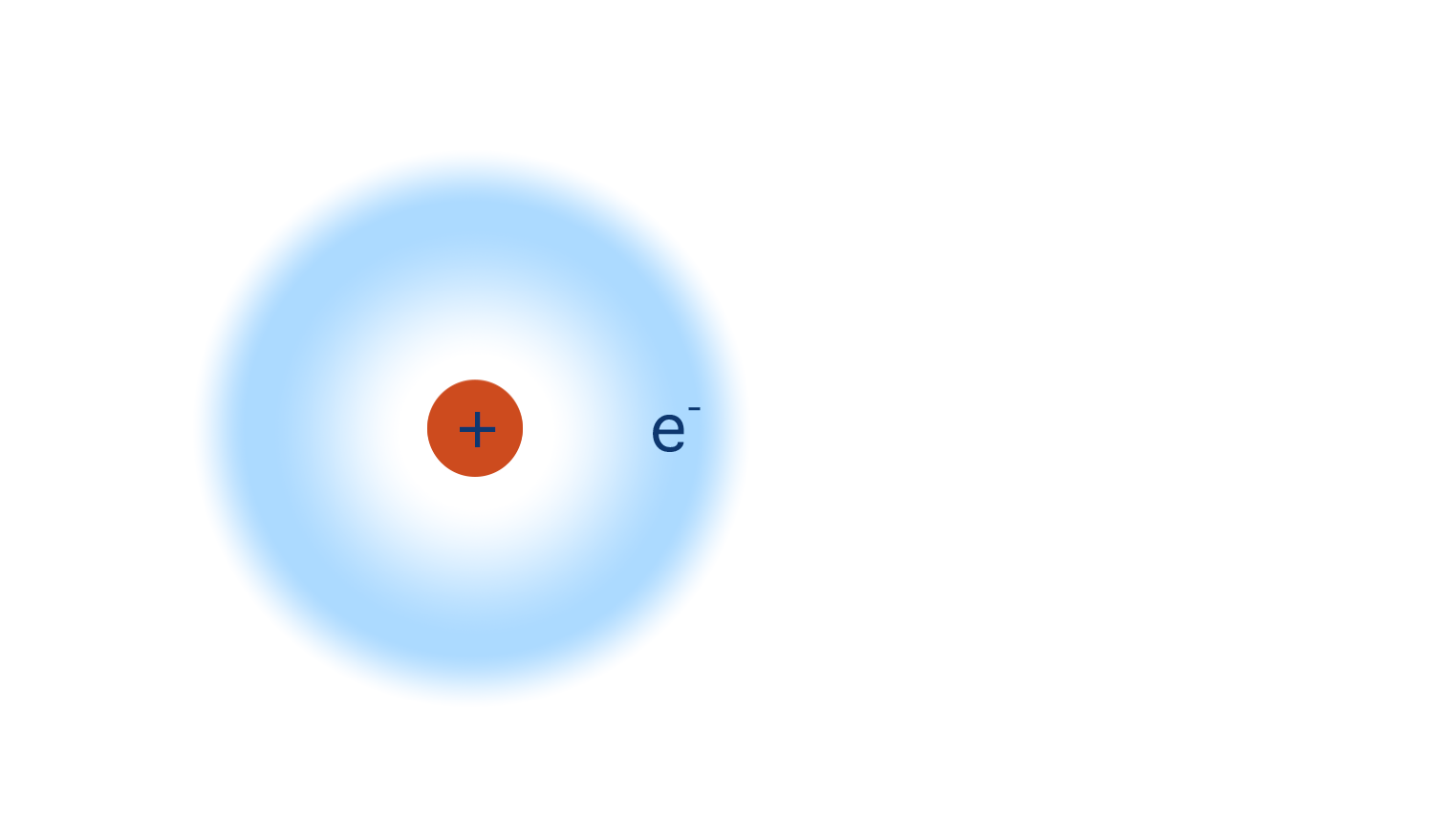
When a hydrogen atom is covalently bonded to a very electronegative element (such as nitrogen, oxygen or flourine), its electron is pulled away from it and towards the more electronegative element.
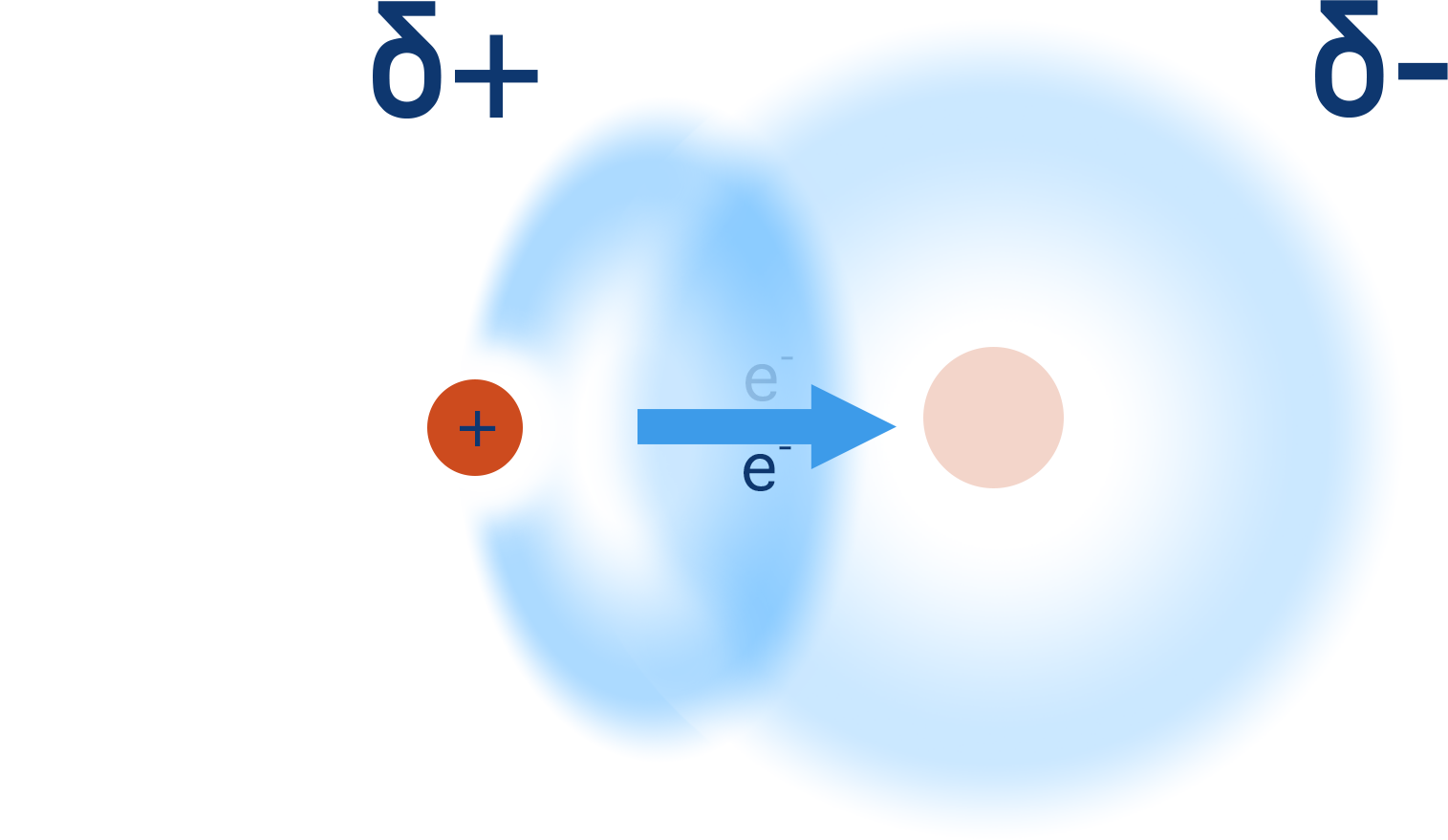
This not only creates a polar bond and leaves the hydrogen with a partial positive charge (δ+) but it also leaves the nucleus (proton) of the hydrogen atom 'exposed' on one side.
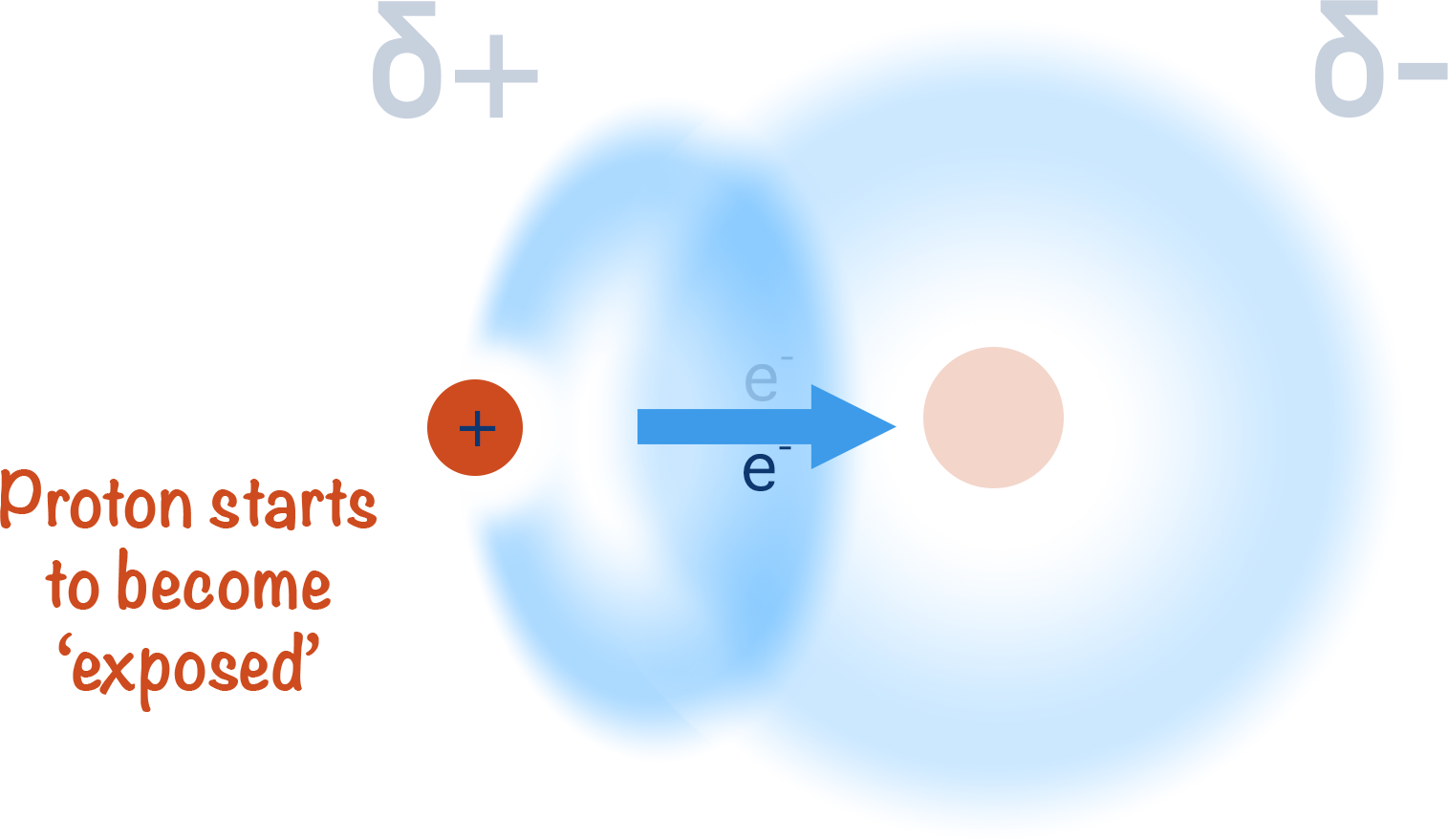
Unlike with other atoms that are able to form a covalent bond, there arn't any further electrons around the nucleus of the hydrogen atom. This means as the bonded electron gets pulled away from it, one side of the atom has no electrons around it and this exposes the single proton in the nucleus.
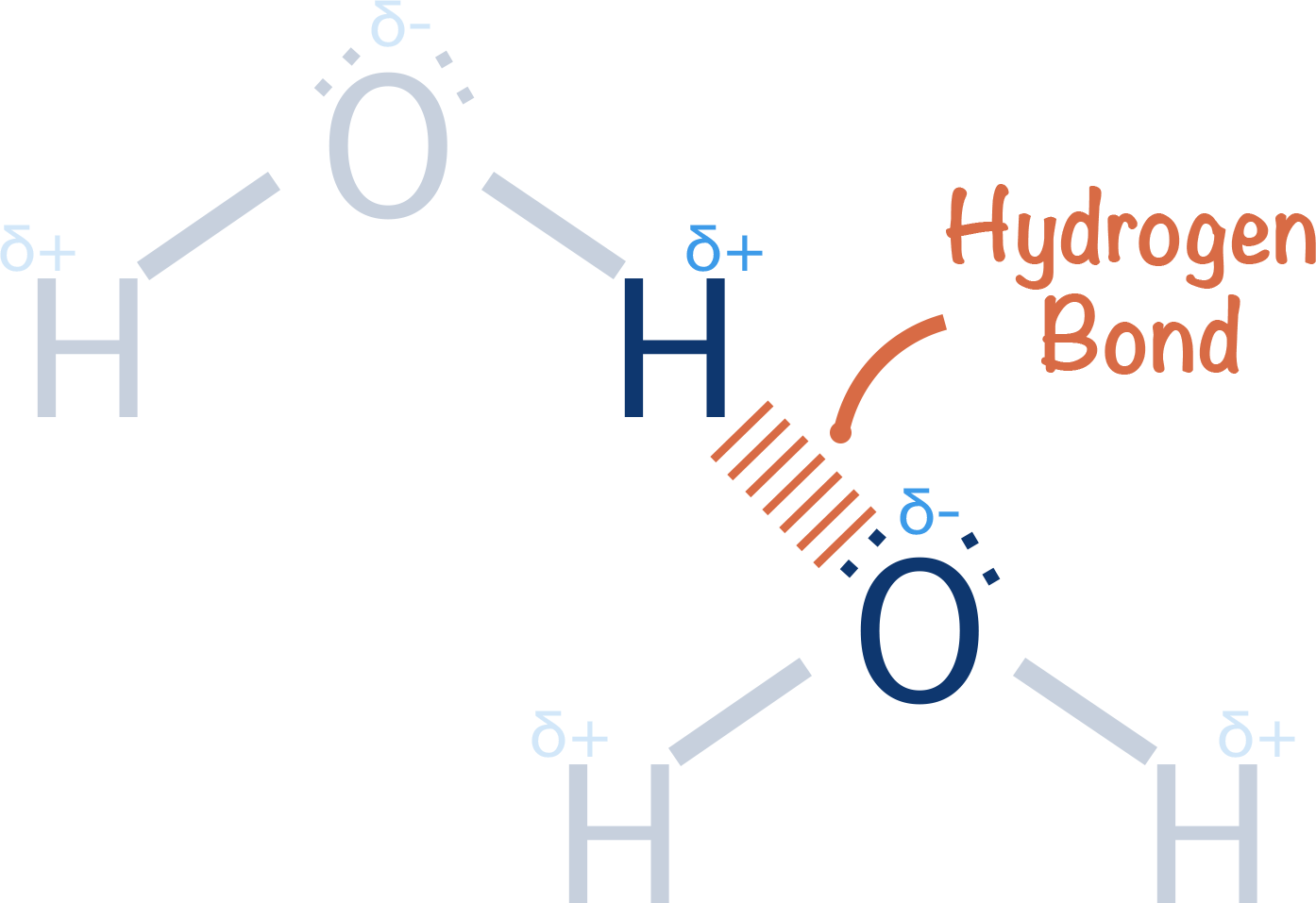
A lone pair of electrons from a nitrogen, oxygen or flourine atom from a similiar bond from a neighbouring molecule will be attracted to this partially exposed proton - giving rise to a 'hydrogen bond'.
Hydrogen Bonding vs Permanent Dipole-Dipole Forces
Molecules that are able to form hydrogen bonds have higher melting and boiling points than similar molecules that are unable to.
For example...
Nitrogen and phosphorus are both group V (5) elements and form three covalent bonds with hydrogen atoms, forming ammonia (NH3) and phosphine (PH3). Ammonia has a higher melting point than phosphine, even though its a smaller molecule and you would expect phosphine to have the higher melting point.
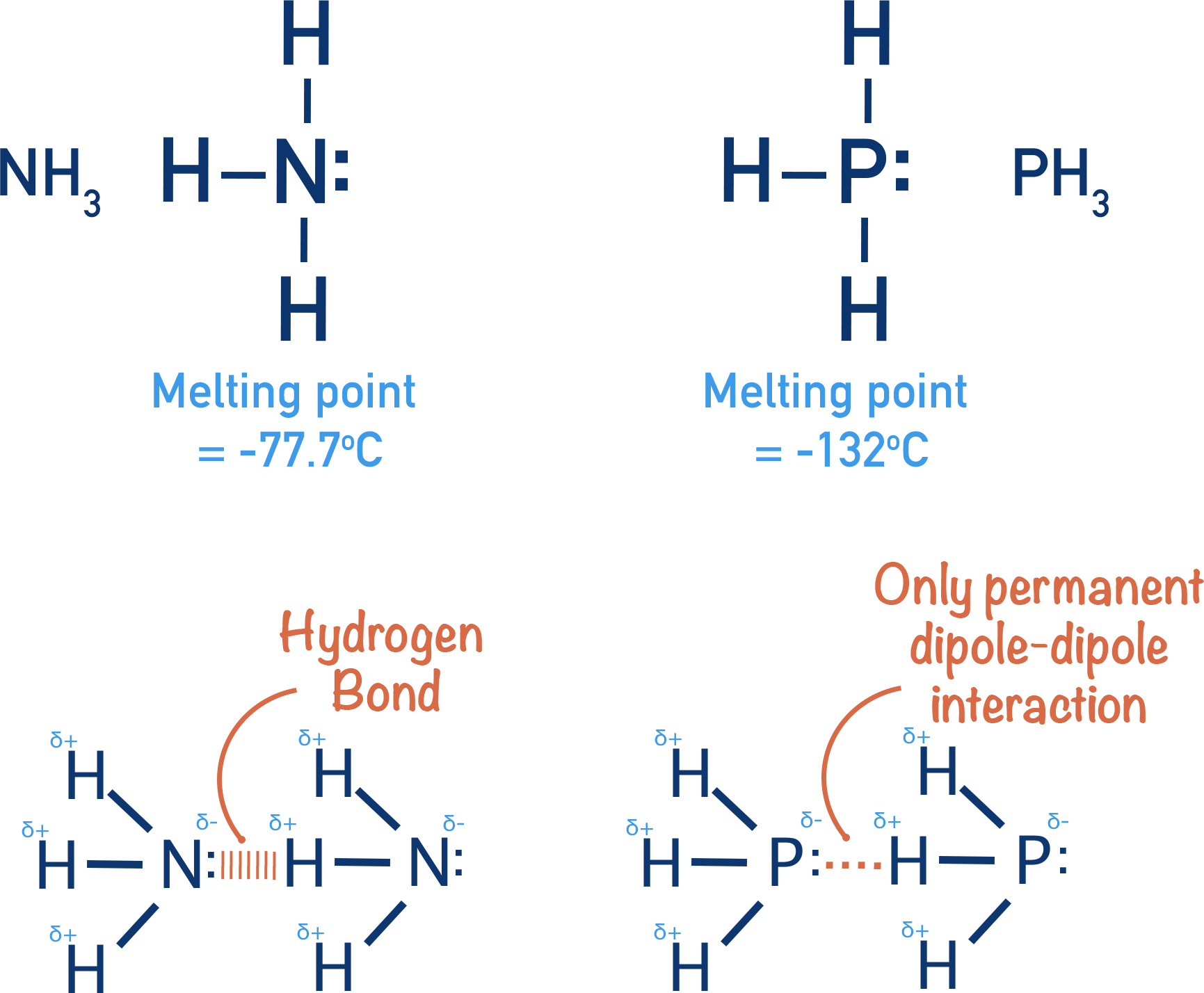
Ammonia has a higher melting point than phosphine as temporary induced dipole-dipole forces, permanent dipole-dipole forces and hydrogen bonds are able to form between ammonia molecules.
In phosphine, only temporary induced dipole-dipole forces and permanent dipole-dipole forces are able to form. Weaker forces of attraction between molecules means less energy is required to overcome the intermolecular forces in phosphine than ammonia, giving ammonia the higher melting point of the two.
The same thing can be seen with water (H2O) and hydrogen sulfide (H2S) - both group VI (6) elements and hydrogen fluoride (HF) and hydrogen chloride (HCl) - both group VII (7) elements.
Water has a higher melting point than hydrogen sulfide, even though it is a smaller molecule and its temporay induced dipole-dipole forces would be weaker than in hydrogen sulfide.
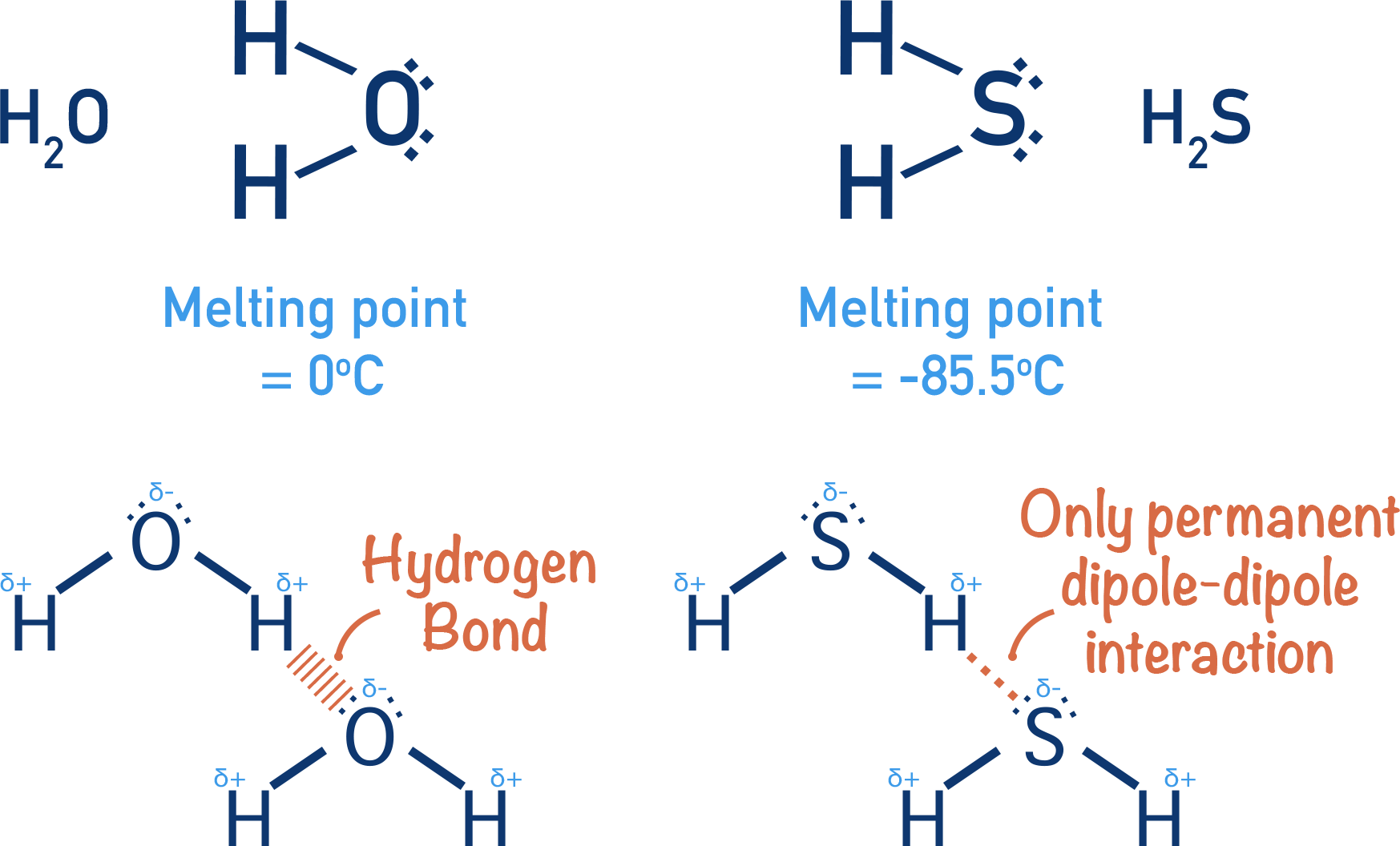
Hydrogen flouride has a higher boiling point than hydrogen chloride, even though it is a smaller molecule and its temporay induced dipole-dipole forces would be weaker than in hydrogen chloride.
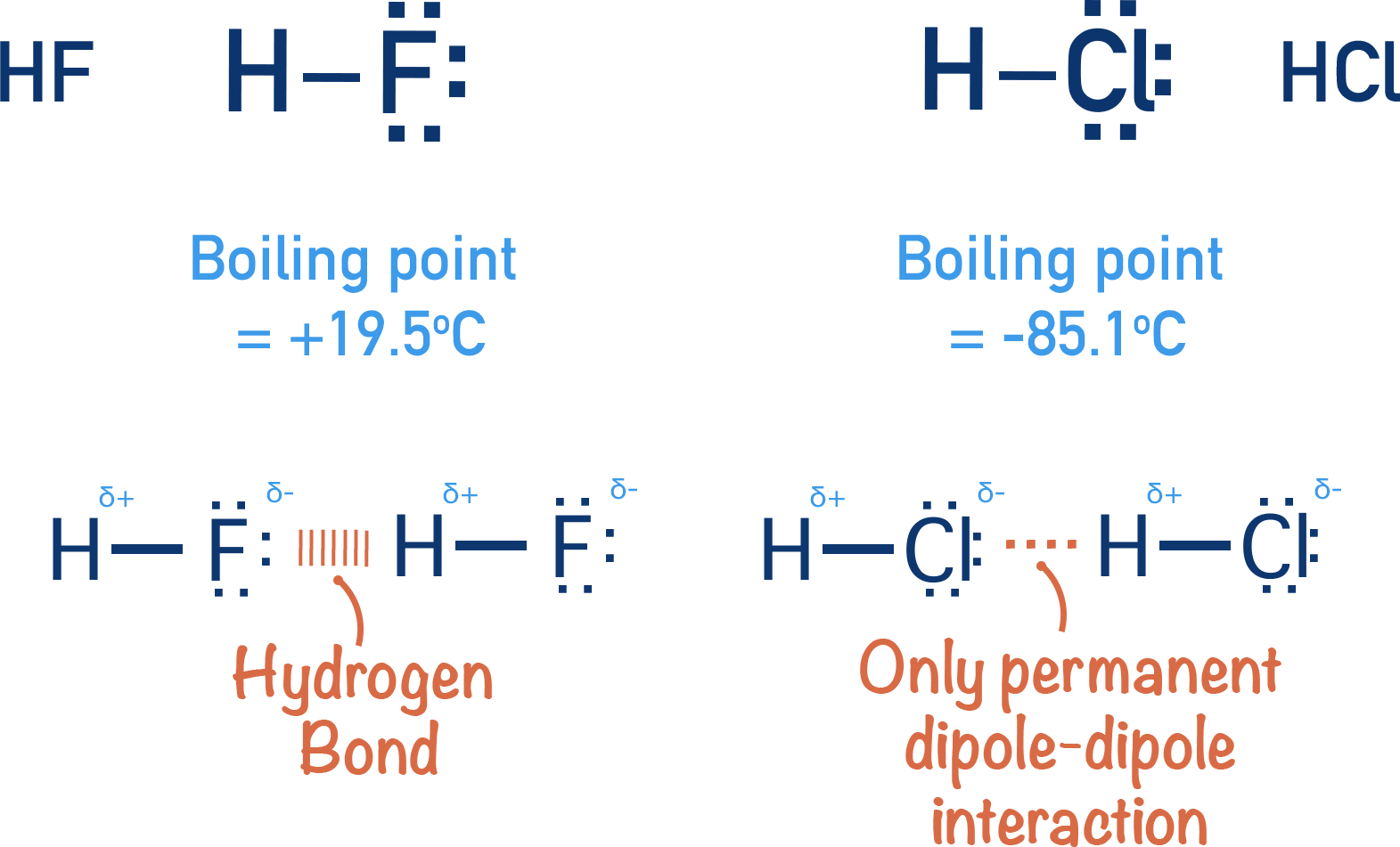
These observations can all be explained by the presence of hydrogen bonding between water and hydrogen fluoride molecules.
We’ve launched our new site! 🎉
Course-specific notes with built-in search!
AP • A-Level (AQA • CIE • Edexcel • OCR) • IB • NCERT 11 + 12
over 750+ new pages and 3,500 images.
Visit the new homepage
