Video Tutorial Permanent Dipole-Dipole
Quick Notes Permanent Dipole-Dipole
- Permanent dipole-dipole interactions exist between polar molecules.
- A partially negatively charged area of one molecule is attracted to a partially positively charged area of another molecule.
- The attractions are permanent and are stronger than temporary, induced dipole-dipole forces.
- The greater the polarity of a bond or molecule, the stronger the permanent dipole-dipole interactions.
Full Notes Permanent Dipole-Dipole
The theory behind permanent dipole-dipole interactions is very similar to temporary, induced dipole-dipole interactions. There is an unequal distribution of electrons within a molecule, leading to partially positive and partially negative regions of charge. These regions of charge interact with neighbouring molecules, and forces of attraction can arise.
Molecules can be polar because electrons within a covalent bond are not evenly shared between the bonding atoms. This results in one atom experiencing a greater share of the electrons and becoming partially negatively charged; the other atom becomes partially positively charged as it has a lesser share of electrons. Such a bond is described as polar (it has two ‘poles’ – positive and negative).
The ability of an atom to attract electrons towards itself in a covalent bond is called its electronegativity (see Electronegativity). The greater the electronegativity of a covalently bonded atom, the ‘greedier’ it is, so the more likely it is to pull electrons towards itself.
The important thing to remember here is that electronegativity is relative. It’s the difference in electronegativity between two atoms that leads to a bond becoming polar.
If you have an atom that is not very electronegative (hydrogen, for example) and an atom that is very electronegative (fluorine, for example), then there will be a very polar bond. Because the fluorine is much more electronegative than hydrogen, it will pull the electrons within the bond towards itself.

But, if you have two atoms that have similar electronegativities (hydrogen and carbon, for example) there will not be a polar bond as neither atom is significantly better at pulling the electrons towards itself than the other.
There is nearly always an uneven distribution of electrons within a covalent bond if the bonding atoms are not the same, but the difference is often so small that at A-level we do not consider the C-H bond to be polar.
If there is a polar bond within a molecule then the charges of the polar bond will align themselves with the opposite charges on the polar bond of a neighbouring molecule.
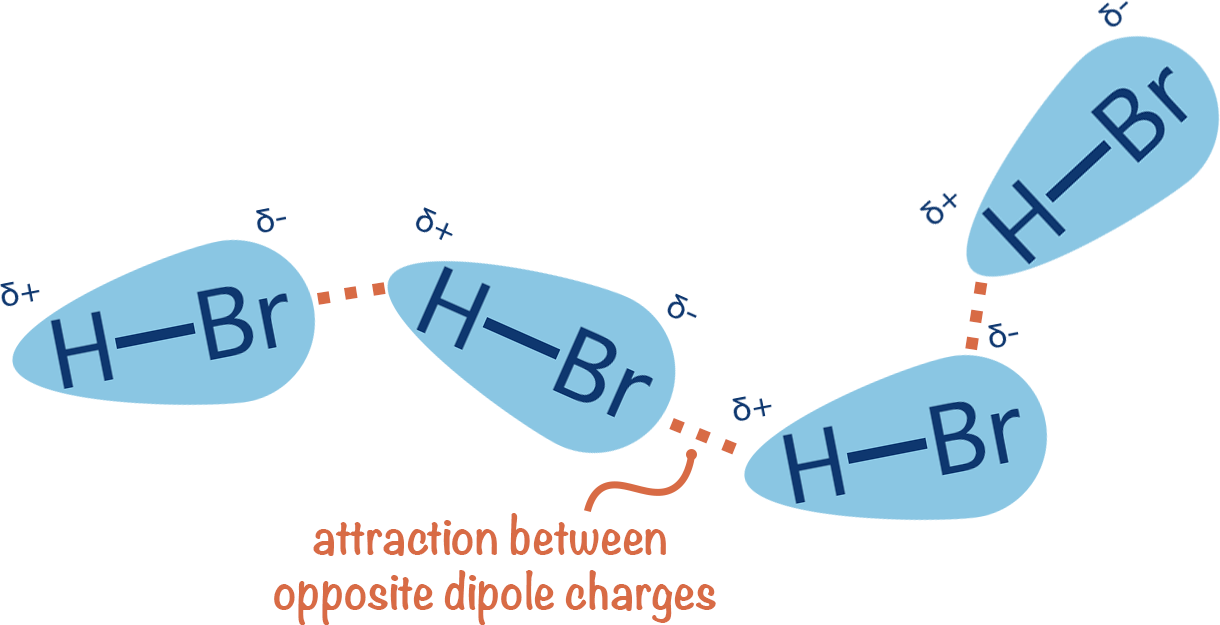
Opposite charges attract and this electrostatic attraction allows forces to arise between the polar bonds. These forces are known as permanent, dipole-dipole interactions.
The more polar a molecule or bond is, the stronger the dipole-dipole forces are because the forces of attraction are greater.
We’ve launched our new site! 🎉
Course-specific notes with built-in search!
AP • A-Level (AQA • CIE • Edexcel • OCR) • IB • NCERT 11 + 12
over 750+ new pages and 3,500 images.
Visit the new homepage
