Quick Notes Electromotive Force, EMF
- In an electrochemical cell, charge flows between two electrodes because of a potential difference between them.
- Once both potentials are the same, no charge will flow.
- To maintain a constant flow of charge, the potential difference between the electrodes needs to be maintained – this requires energy.
- The amount of energy needed to maintain a certain potential difference is called the electromotive force (EMF).
- The electromotive force in electrochemical cells comes from the energy released by redox reactions occurring in each half-cell, keeping the potentials different.
- As reactants are used up, the reactions slow down meaning the electromotive force drops, resulting in less potential difference between electrodes, so a smaller current is produced. This is why batteries lose power and ‘go flat’.
Full Notes Electromotive Force, EMF
EMF is electromotive force and it’s commonly misunderstood by students as it appears to be the same as the potential difference of an electrochemical!
This is background information that you are unlikely to be examined on, but I recommend you understand the difference between EMF and potential difference to avoid any confusion if either one is referred to in an exam question.
If there is a difference in potential between two connected surfaces, charge will ‘flow’ from one to the other, producing an electrical current.
Touching a statically charged surface, for example, causes charged particles to move from the charged surface to the person touching the surface – this rapid movement creates a small electrical current that can be seen (and felt) as a spark. It’s the difference in potential between the person and the surface that enables this to happen. The surface has a build-up of charged particles on it, so it has a different electrical potential to the person.
Once the potentials of both surfaces are the same, no further charge will move. This is why a spark only happens once. If we want to maintain a potential difference between two connected surfaces, energy is required to generate and maintain it. This energy is called an electromotive force.
It’s a little bit like having two containers connected by a hose at the bottom. If one container is full of water, water will go through the hose into the other container.
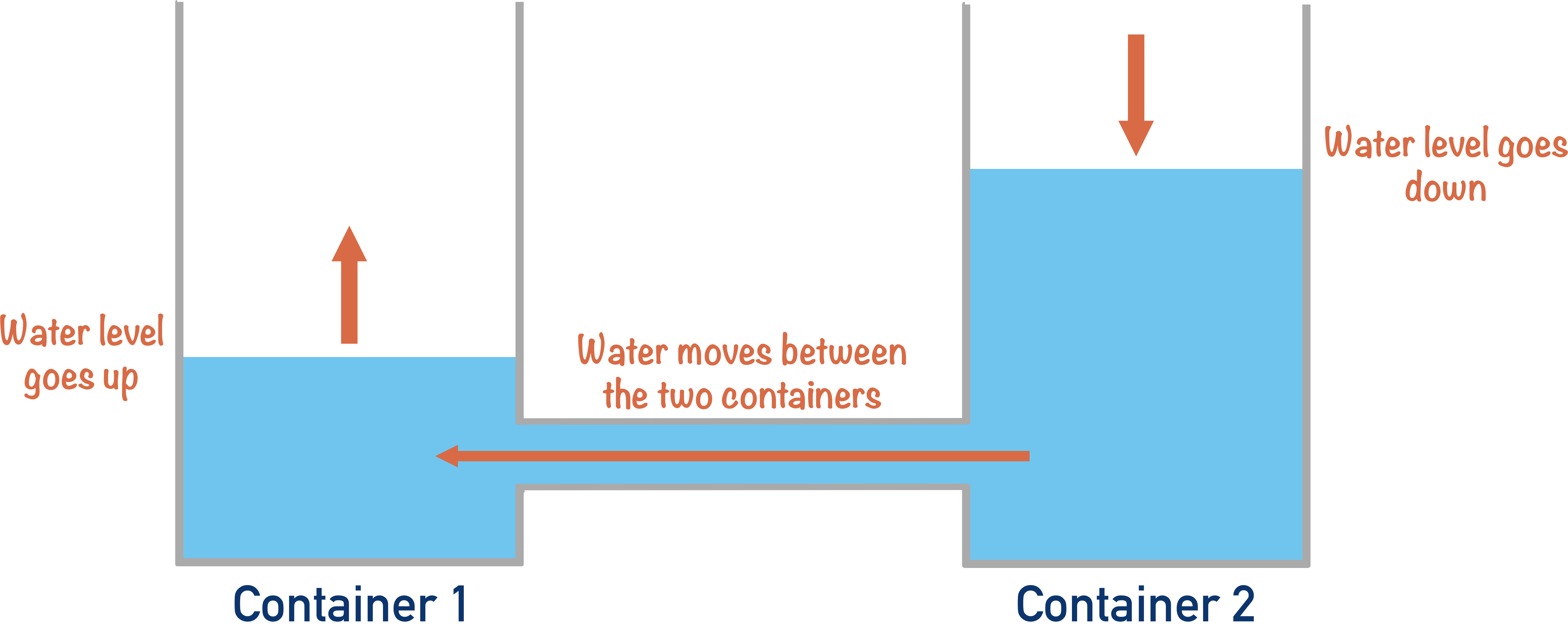
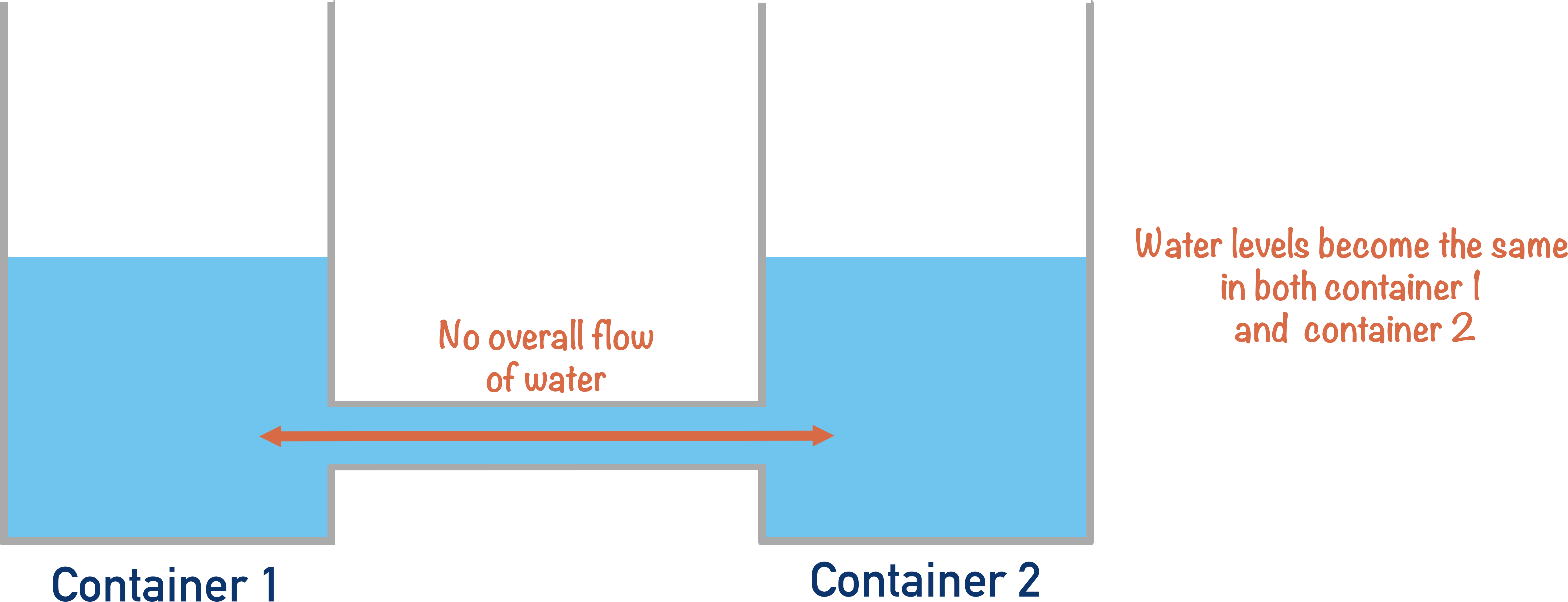
Water will flow until they both contain the same amount of water.
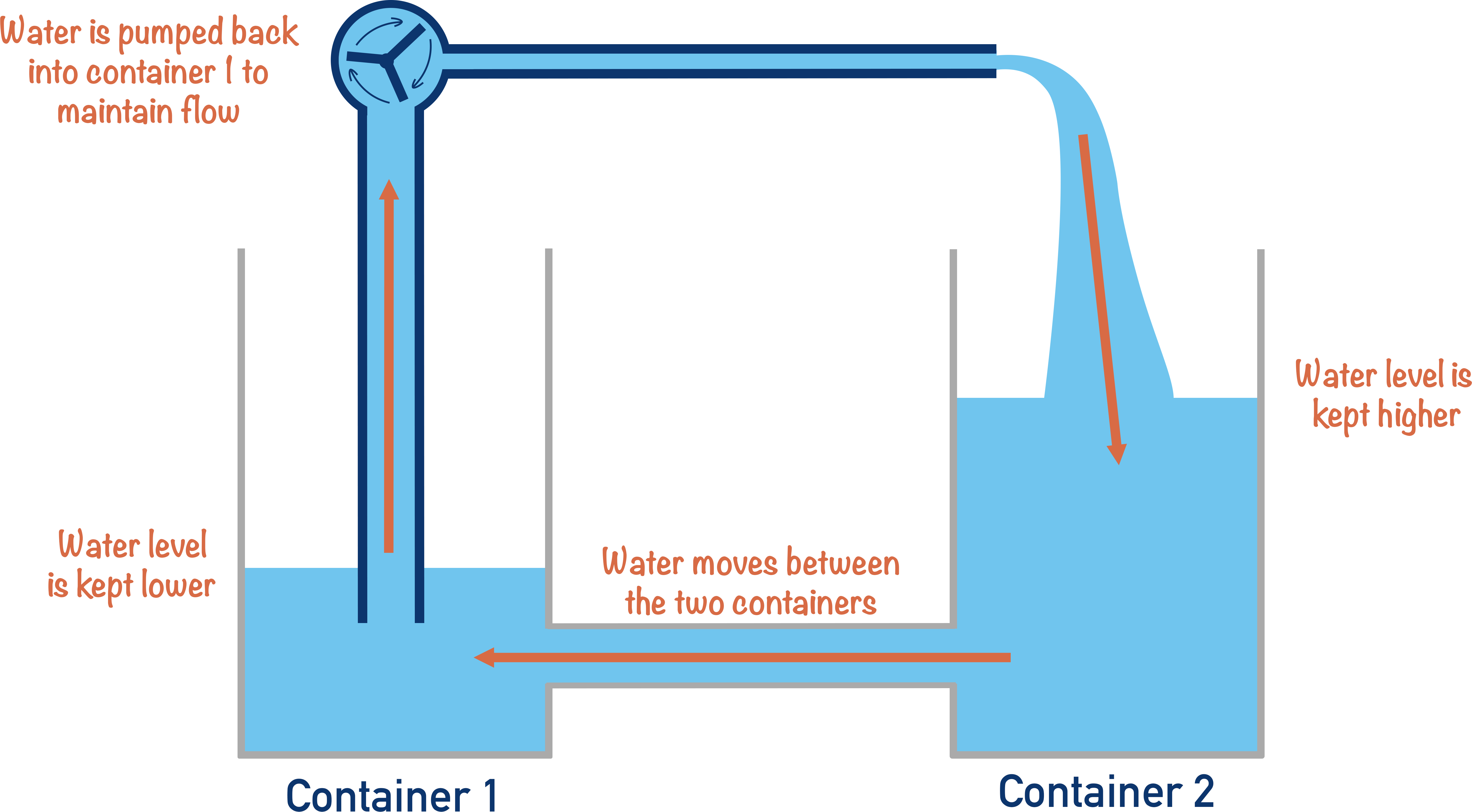
If we want to keep water moving between them, water must be taken out of the second container and added back into the first container – this maintains a ‘difference’ in the water levels and ensures water keeps flowing.
Energy is needed to move the water back from container two to container one. If instead of water we were trying to move charged particles back from one surface to another, the energy needed is called electromotive force, and would maintain a potential difference between the two surfaces.
The value for the potential difference and the electromotive force of an electrochemical cell is the same. The electromotive force comes from the redox reactions occurring in the half-cells, and the resulting potential difference is the same as the electromotive force produced by the redox reactions.
They are technically different concepts, but don’t let yourself get confused. If one exam question asks you to calculate the ‘EMF’ and another asks for the ‘potential difference’ of an electrochemical cell – they should be the same values and both would have the units of volts.
