Video Tutorial Hess’s Law
Quick Notes Hess' Law
- Hess’ Law states that the energy change that occurs when a substance is changed into a product is the same, regardless of the route taken.
- There is often more than one way to go from a reactant to a product, meaning there is more than one ‘route’ to convert a substance to a particular product.
- Hess cycles are ways of drawing out the energy changes that occur when a substance is turned into a different substance via more than one route.
- By using values for known enthalpy changes, unknown enthalpy changes can be found. This can be useful for enthalpy changes that can't be measured directly in experiments.
Full Notes Hess' Law
When a substance reacts to form a new product, an enthalpy change, ΔH, occurs (see Enthalpy Change).
Very often in chemistry there is more than one route (way) to turn a set of reactants into given products. But the enthalpy change that occurs is the same regardless of how we get to our end product. This is called Hess's Law.
Hess’ Law:The enthalpy change for a reaction is always the same, regardless of the route taken.
Imagine having to get to the top of a mountain, you can either try to go straight up the mountain or walk around a path that winds up the mountain. With either route, the height you climbed would be exactly the same!
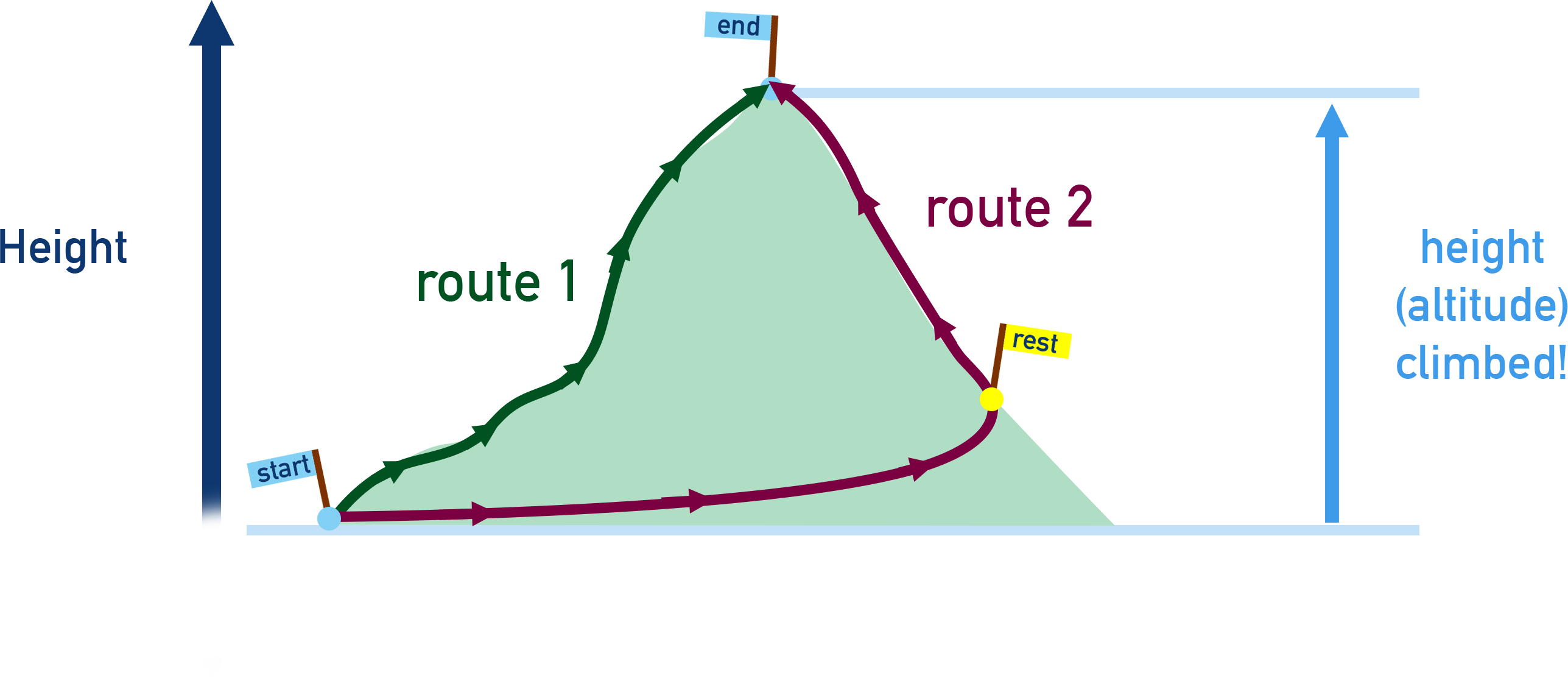
If you ‘stop’ halfway up the mountain before using a different path again to get to the top, the height you climbed would still be the same as if you had gone straight up in one go. This is the same for enthalpy changes in reactions; it doesn’t matter HOW you get from a reactant to a product, the total enthalpy change will always be the same.
Imagine a reaction where A + B + C get turned into D + E.
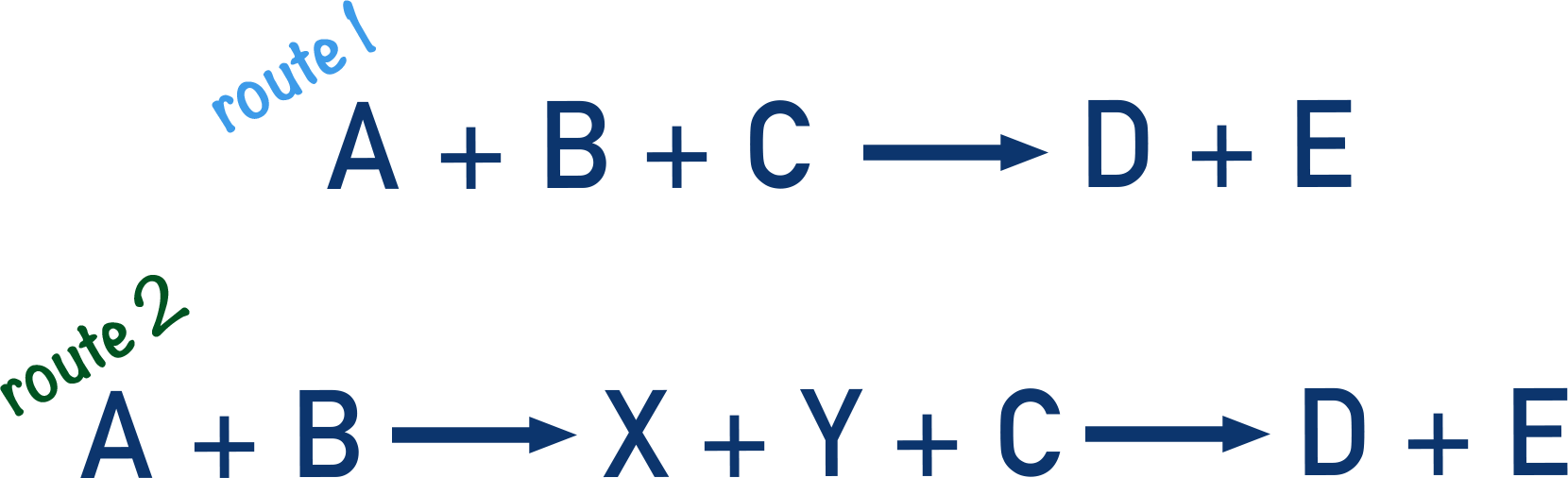
The reaction can occur by a direct route: A + B + C → D + E or can occur by another route in two stages: A + B → X + Y and then X + Y + C → D + E.
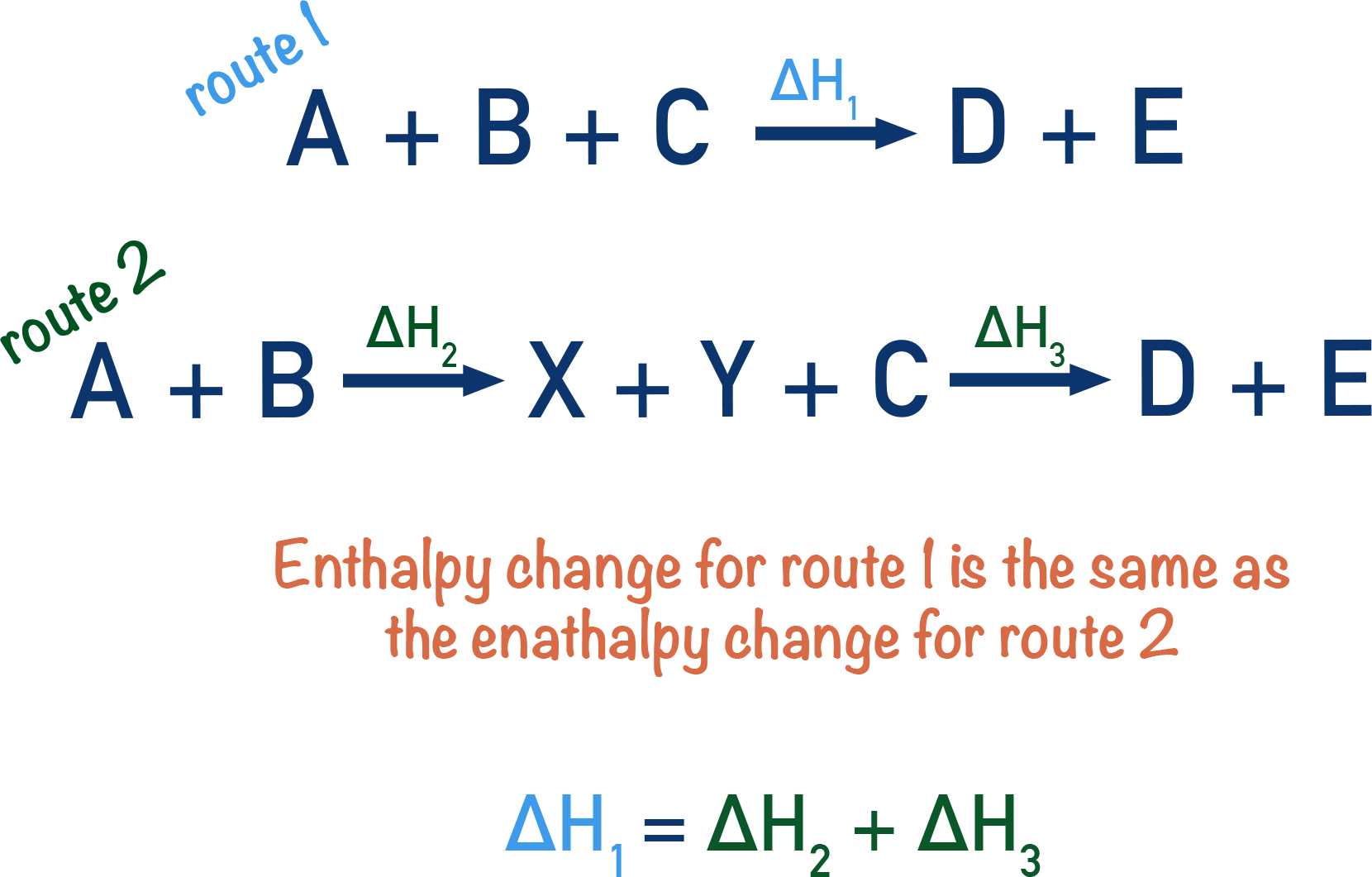
The enthalpy change for both routes will be the same.
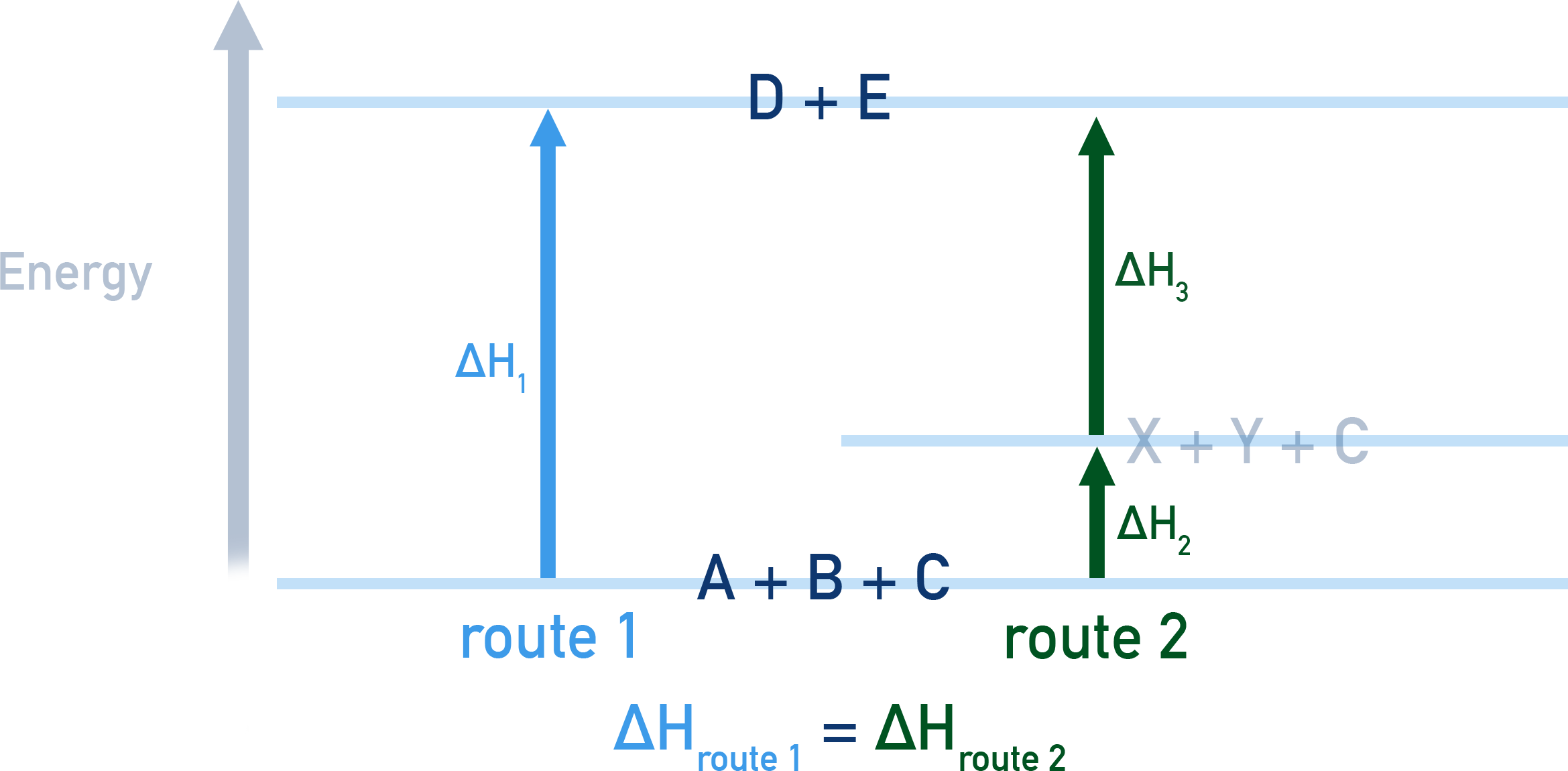
The fact that one of the routes (route 2 in this imaginary example) happens in two stages doesn't matter - their combined enthalpy changes will be be the same as for the other route (route 1).
If we know all the enthalpy changes for individual stages within a reaction process, we can calculate the overall enthalpy change. We can also rearrange the enthalpy changes to calculate enthalpy changes for a specific stage. This can be done using Hess Cycles.
Hess Cycles
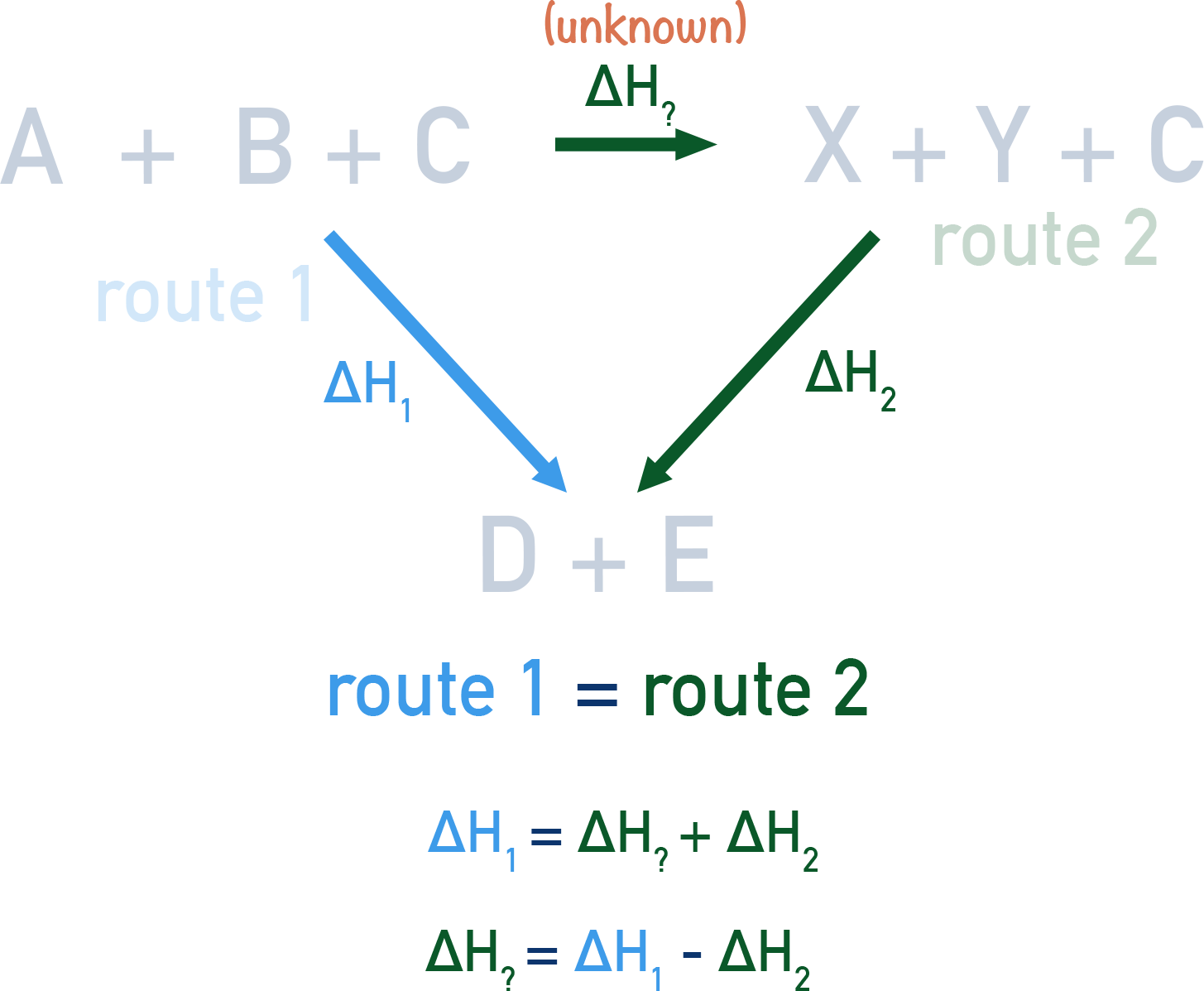
By drawing out two routes to form a product from reactants, we can find the enthalpy change for one of the routes if the other enthalpy change is known. A simple triangle can be constructed and the missing enthalpy change found.
Using the above example of A + B + C → D + E, we can show both possible routes 'at the same time'.
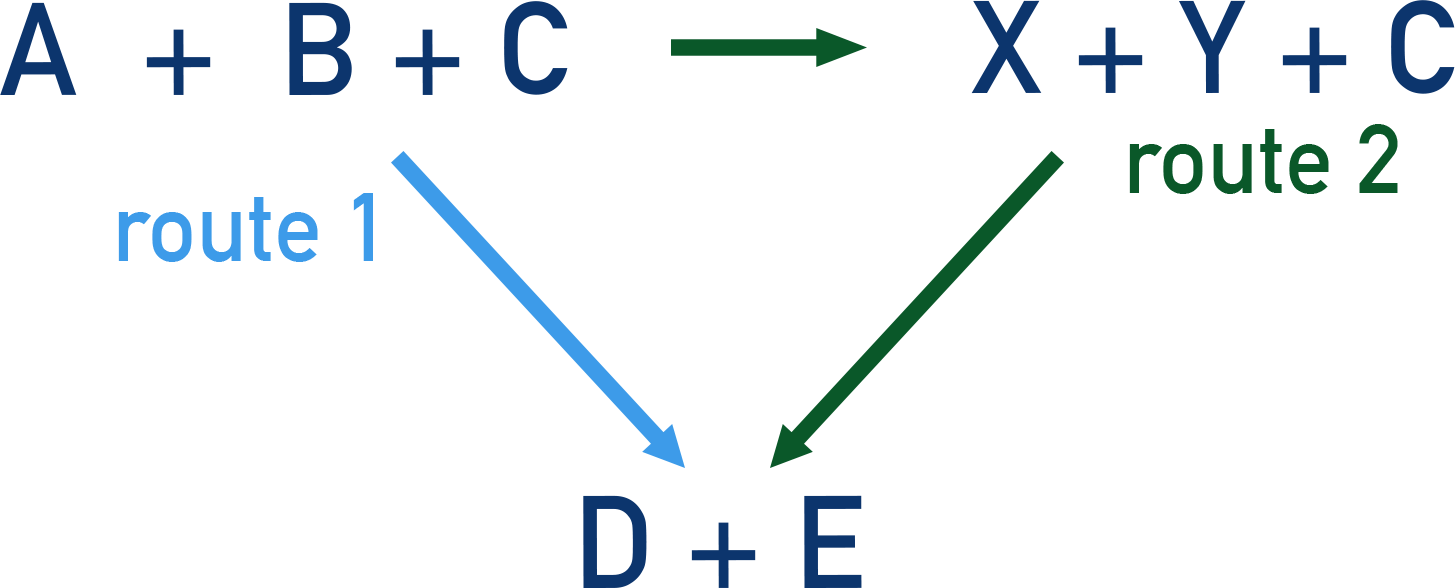
Hess's Law tells us that the overall enthalpy change for both routes will be the same, meaning that we can find an unknown enthalpy change for part of a route using the other enthalpy changes.

Hess cycles can be very useful for finding enthalpy changes for reactions that can't be measured experimentally, such as enthalpies of formation.
For example...
The enthalpy of formation of propane (C3H8) can be found by using a Hess cycle and combustion data. This enthalpy change can't be measured directly by experiment, only by using a Hess cycle.
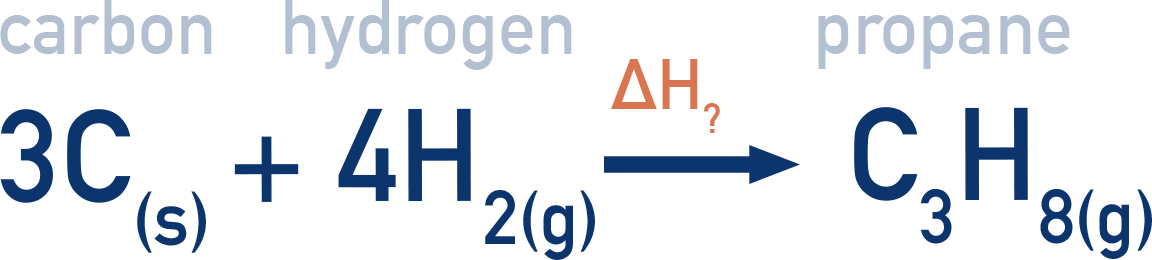
To make one mole of propane (C3H8), three moles of carbon (C) and four moles of hydrogen (H2) are needed.
Three moles of carbon and four moles of hydrogen can be combusted to form three moles of carbon dioxide and four moles of water. One mole of propane can be combusted to also form three moles of carbon dioxide and four moles of water.
This means we can construct a Hess cycle showing both the combustion of carbon and hydrogen and the combustion of propane as well as the formation of propane.
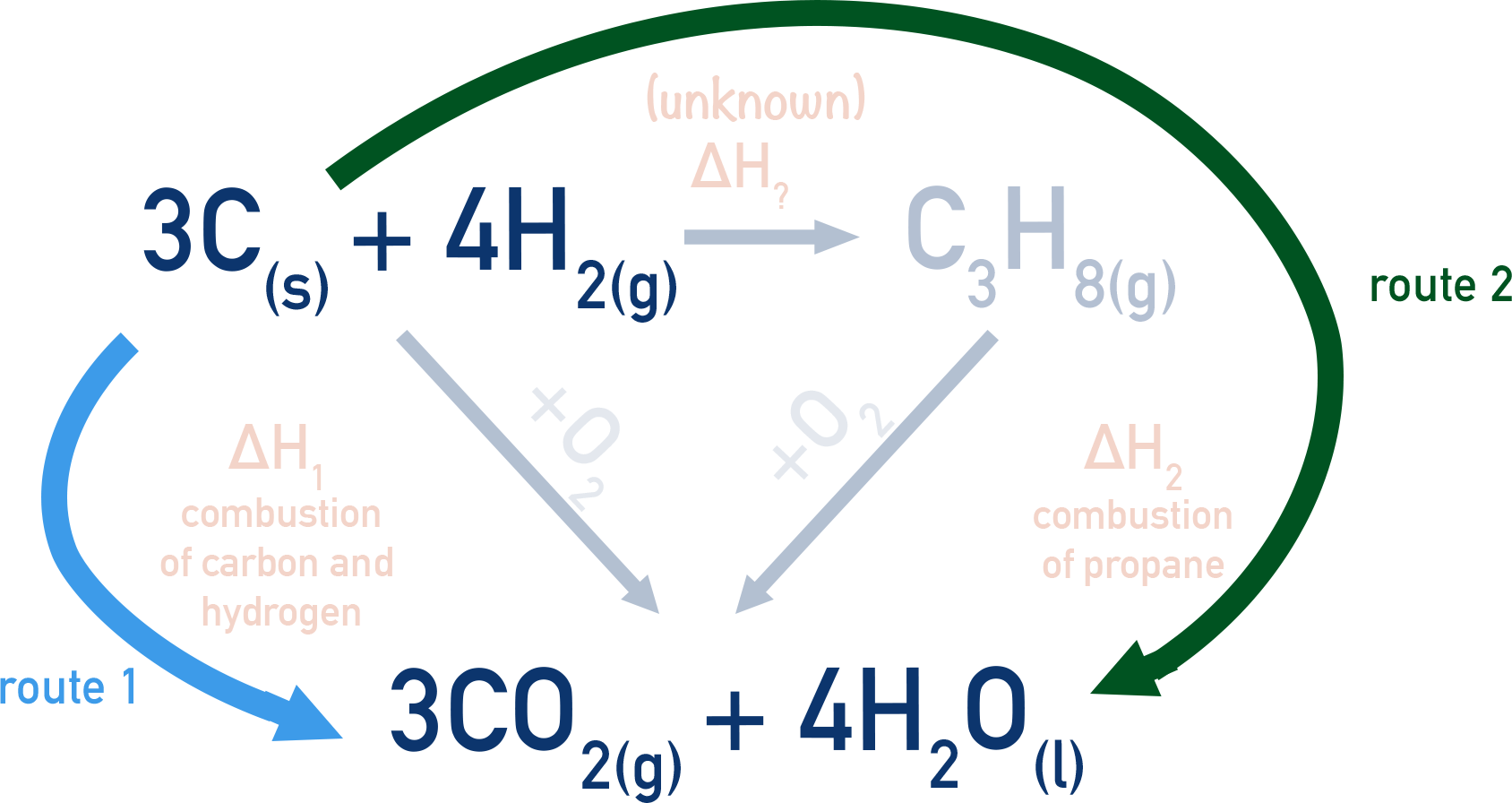
There are two routes in the cycle - the first route is to combust three moles of carbon and four moles of hydrogen directly. The second route is to convert three moles of carbon and four moles of hydrogen into propane, then combust the propane.
Hess’s law tells us the overall enthalpy change for both routes will be the same.
We can use enthalpies of combustion data to find the enthalpy changes for combustion reactions in the cycle.
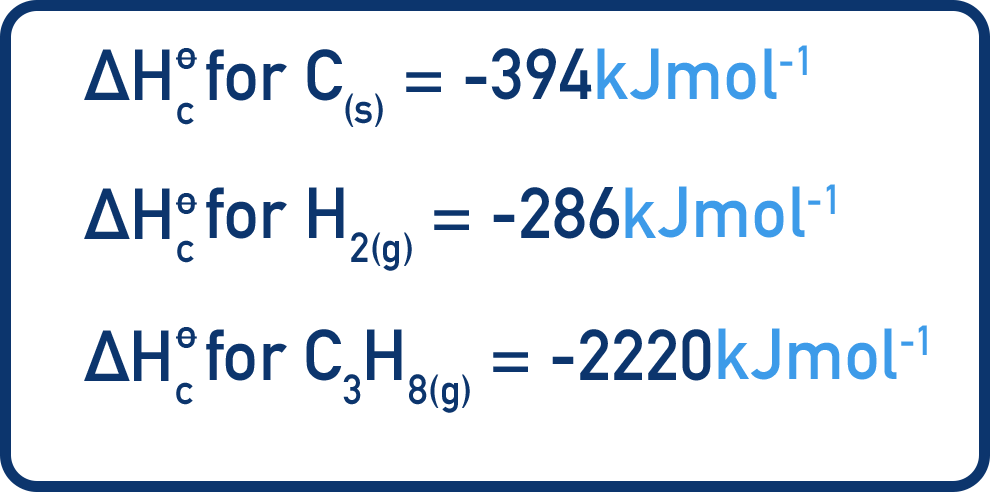
For route 1 (ΔH1), there are three moles of carbon and four moles of hydrogen combusting. This means ΔH1 = -2326 kJmol-1.
Route 2 is made up of the combustion of propane (ΔH2 = -2220 kJmol-1) and the formation of propane.
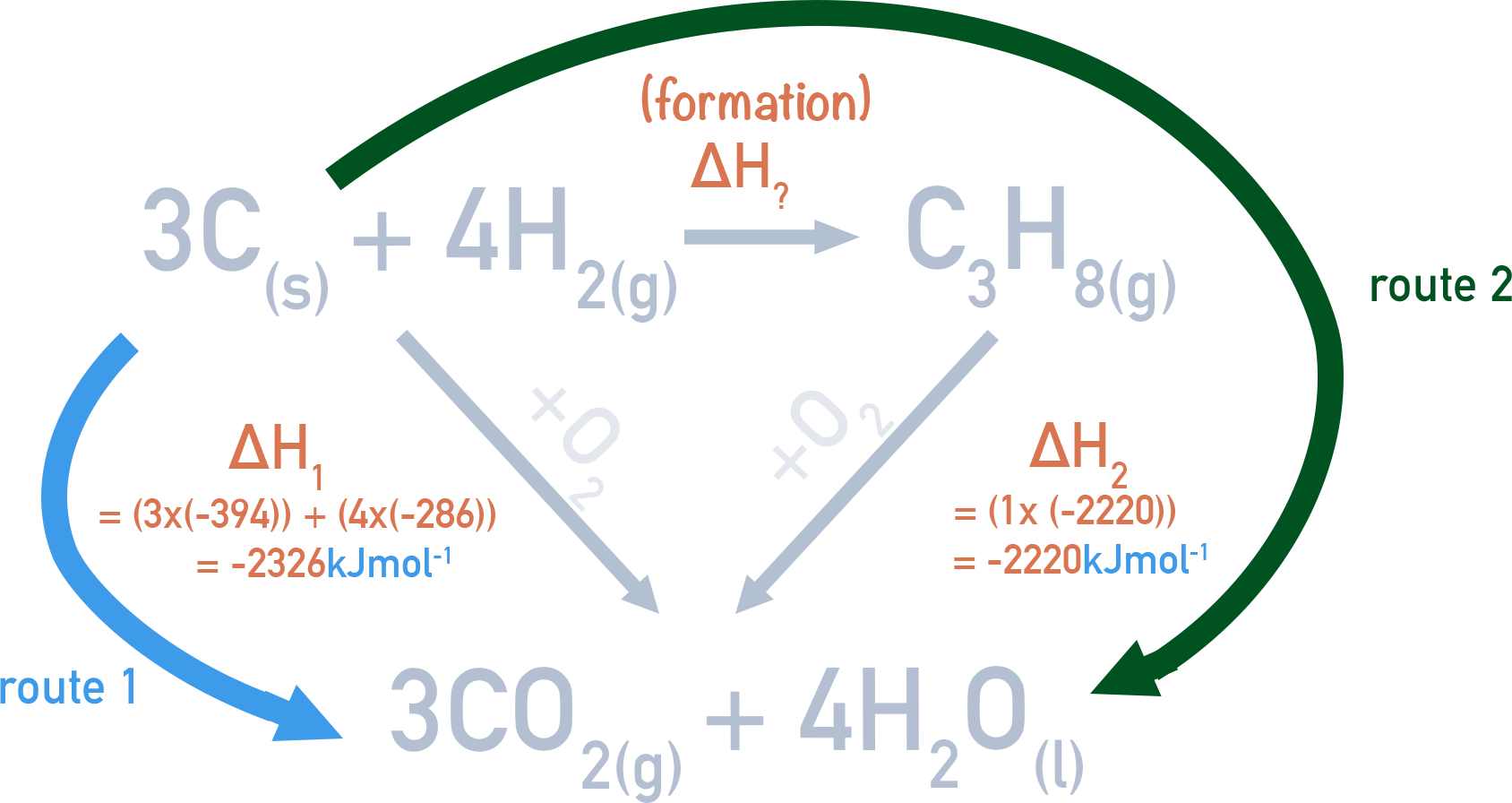
Hess’s law tells us the overall enthalpy change for both routes will be the same.
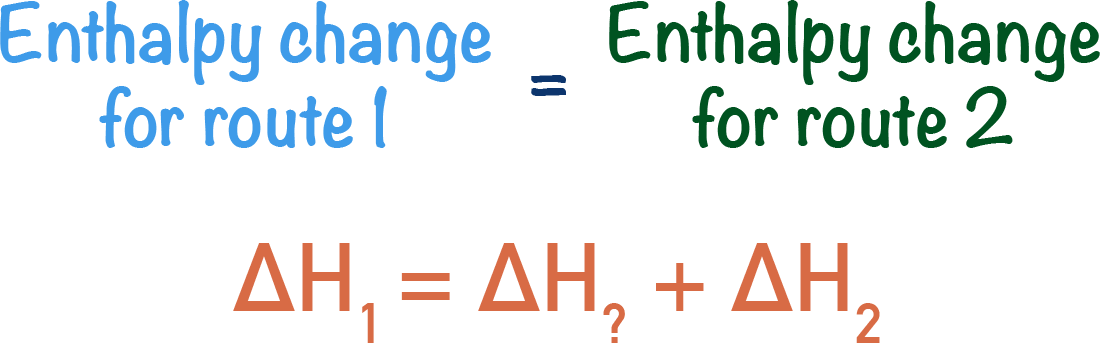
Now, we just need to re-arrange the equation shown to find our unknown enthalpy change for the formation of propane.
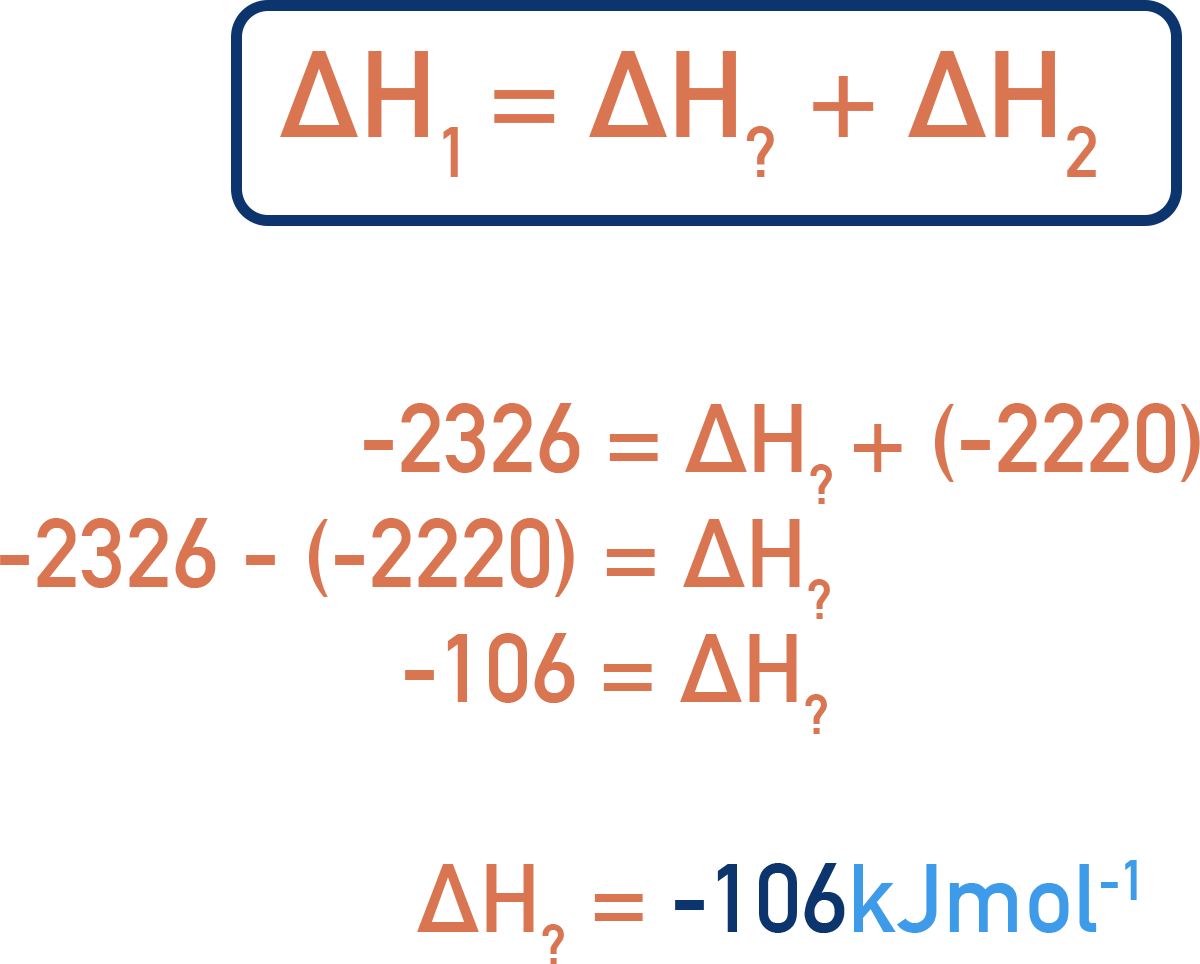
We’ve launched our new site! 🎉
Course-specific notes with built-in search!
AP • A-Level (AQA • CIE • Edexcel • OCR) • IB • NCERT 11 + 12
over 750+ new pages and 3,500 images.
Visit the new homepage
