Quick Notes - A2 Organic Chemistry
Advanced Organic Chemistry
Acid Anhydrides
- Acid anhydrides are made from two carboxylic acids joined together in a condensation reaction.
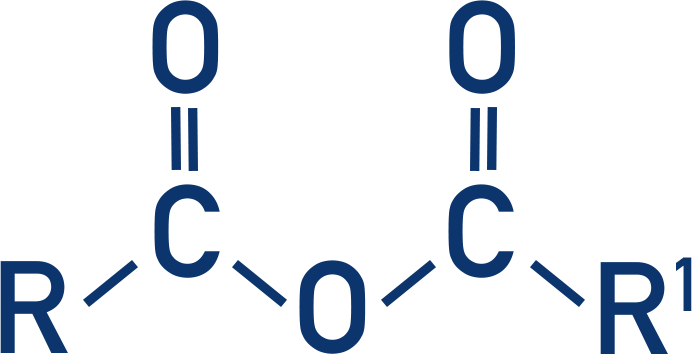
- Acid anhydrides react in similar ways to acyl chlorides, but they are less reactive, making them safer to use.
- Acid Anhydride + Alcohol → Ester + Carboxylic Acid
Acyl Chlorides
- Acyl chlorides are highly reactive and easily react with nucleophiles. They have the functional group:
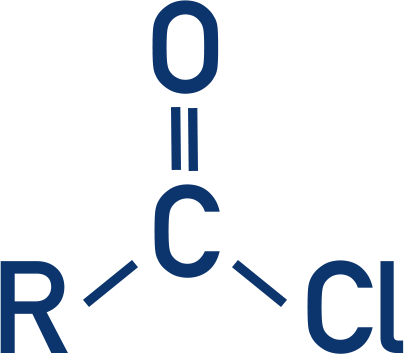
- They are prepared by reacting a carboxylic acid with phosphorus pentachloride (PCl5) in anhydrous conditions (acyl chlorides react easily with water).
- Acyl chlorides react with nucleophiles bonded to a hydrogen atom in addition-elimination reactions:

- Key reactions:
- Acyl Chloride + Water → Carboxylic Acid (+ HCl)
- Acyl Chloride + Alcohol → Ester (+ HCl)
- Acyl Chloride + Ammonia → Primary Amide (+ Ammonium Chloride)
- Acyl Chloride + Primary Amine → Secondary Amide (+ Alkyl Ammonium Chloride)
Carboxylic Acids
- A carboxylic acid group can lose a proton to become a negatively charged carboxylate ion.

- Carboxylic acids can be formed by the oxidation of a primary alcohol with acidified potassium dichromate, under reflux conditions.
- Carboxylic acids can be reduced to primary alcohols using hydride ions (from lithium tetrahydridoaluminate (LiAlH4)).
- The reduction must be in dry ether as LiAlH4 reacts violently with water molecules.
- Negatively charged carboxylate ions can bond ionically with positive ions, forming carboxylate salts.
Carbonyls - Formation and Reduction
- A carbonyl group is a carbon atom double bonded to an oxygen atom (C=O).
- Aldehydes have a carbonyl group at the end of a carbon chain; ketones have a carbonyl group in the middle of a carbon chain.
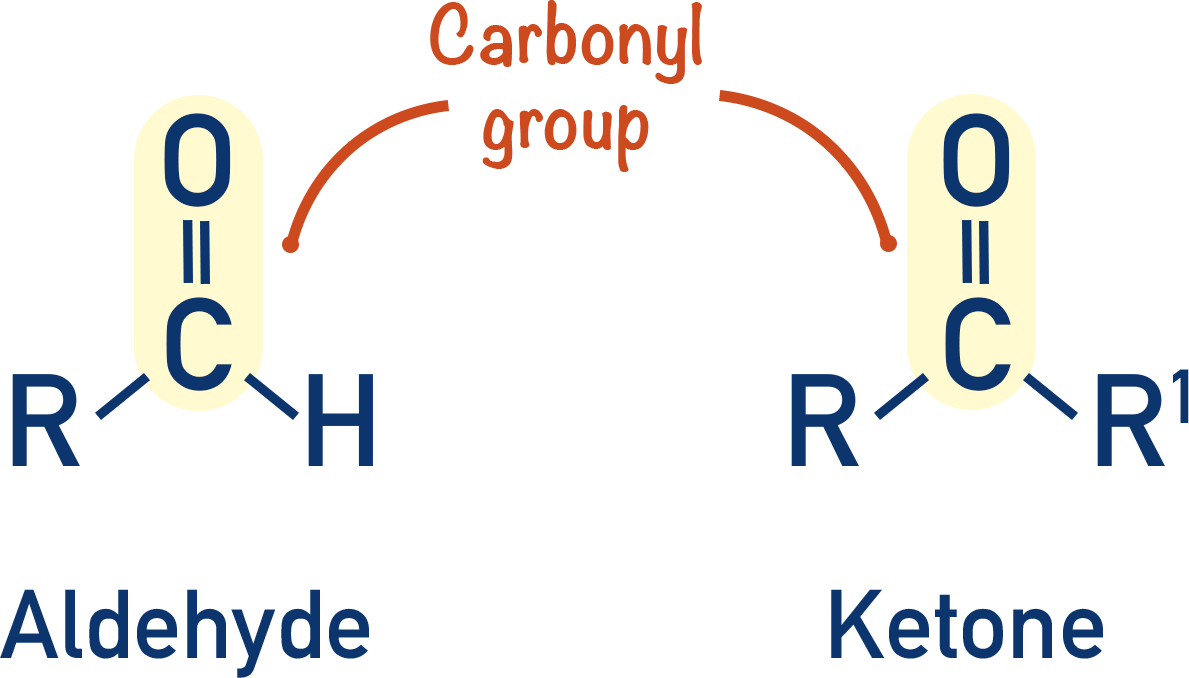
- Aldehydes can be formed from the oxidation of primary alcohols; ketones can be formed from the oxidation of secondary alcohols.
- The partially positively charged carbon atom in an aldehyde and a ketone react with nucleophiles in addition reactions.
- Aldehydes can be reduced with hydride ions (H-) to primary alcohols.
- Ketones can be reduced with hydride ions to secondary alcohols.
- A common source of hydride ions for reduction is the compound lithium tetrahydridoaluminate (LiAlH4).
Identifying Carbonyls
- Aldehydes can be identified by using Tollens’ reagent (silver precipitate formed) or Fehling’s solution (red precipitate formed).
- For both tests, the aldehyde reduces a metal ion (Ag+ in Tollens’; Cu2+ in Fehling’s).
- Aldehydes and ketones can both be identified using Brady’s reagent, producing an orange precipitate.
- A methyl carbonyl group can be identified using a (warm) mixture containing iodine and sodium hydroxide, forming a yellow solid.
Carbonyls - Nucleophilic Addition
- Hydrogen cyanide reacts with carbonyl groups in nucleophilic addition reactions to form hydroxynitriles.
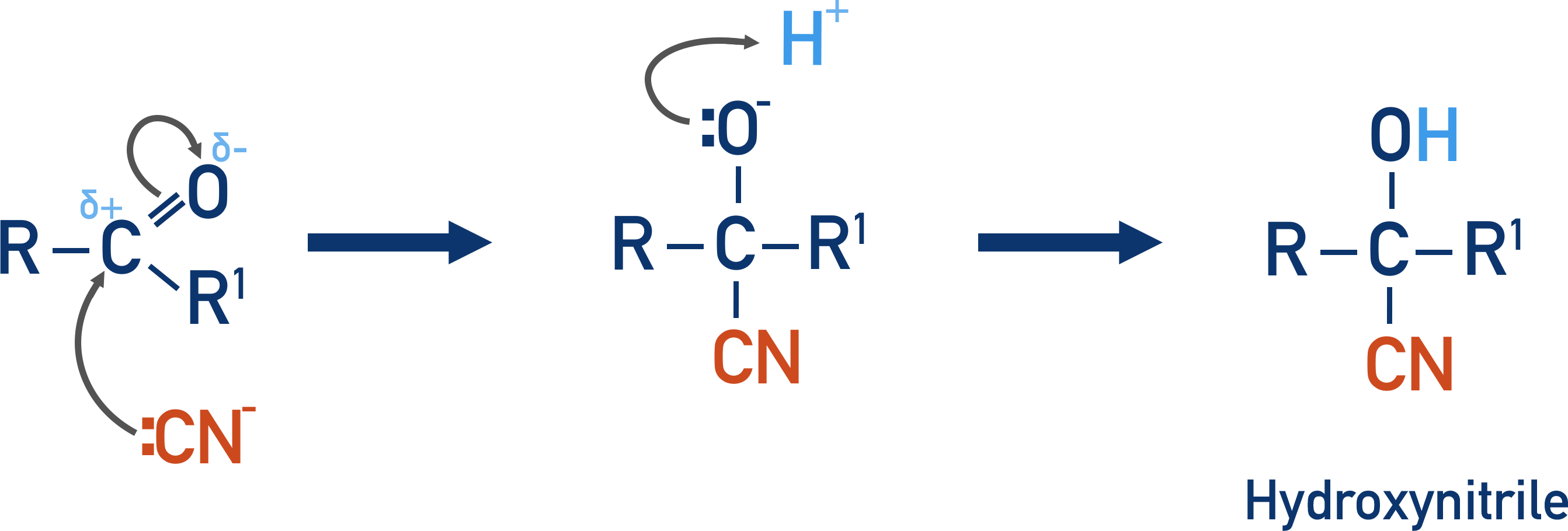
- The length of the carbon chain in a carbonyl molecule can be extended using this reaction.
- Hydrogen cyanide is highly reactive (and toxic); it can be prepared ‘in situ’ using a metal cyanide (KCN or NaCN) and dilute sulfuric acid (H2SO4).
Esters
- Esters are made from a carboxylic acid and an alcohol held together by an ester link.
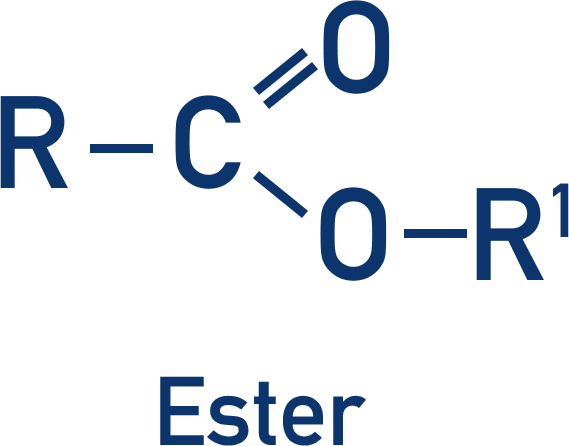
- An ester is formed in an esterification reaction by reacting a carboxylic acid and alcohol together, in the presence of concentrated sulfuric acid (under reflux conditions).
- Esterification is a condensation reaction because water is released as a product.
- Ester links can be broken in hydrolysis reactions (addition of water).
- Hydrolysis of an ester in acidic conditions produces a carboxylic acid and alcohol.
- Hydrolysis of an ester in alkaline conditions produces a carboxylate ion and alcohol.
- Fats and oils are natural examples of esters, called tri-glycerides, made from glycerol (propane-1,2,3-triol) and long chain fatty acids (carboxylic acids with long carbon chains).
Naming Esters
- Esters are named based on the length of the carbon chains in both the carboxylic acid and alcohol that are used to make the ester.
- The general form is alkyl-carboxylate.

- The first part of the name comes from the carbon chain in the alcohol (-alkyl).
- The second part of the name is the same as the carboxylate ion of the carboxylic acid (-carboxylate).
Optical Isomerism
- Carbon can form four covalent bonds. If a carbon atom is bonded to four different groups there are two possible ways of arranging the groups, creating two possible isomers (with the same molecular formula, but differently arranged atoms).
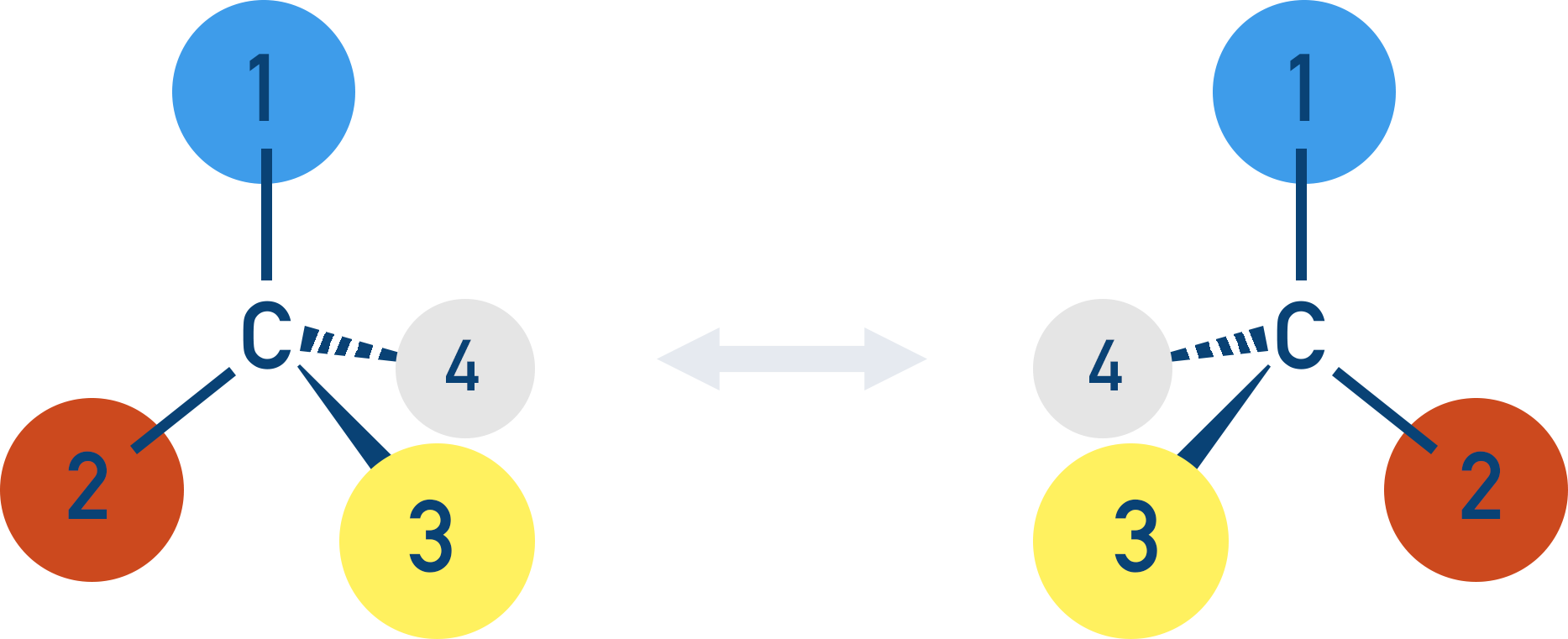
- Optical isomers (called enantiomers) are mirror images of each other that rotate plane-polarised light in opposite directions. They exhibit a form of stereoisomerism (isomers with the same structural formula, but arranged in a different way).
- If equal amounts of both isomers are present in a mixture, there is no overall rotation of plane-polarised light – the mixture is racemic.
Benzene and Aromatic Chemistry
Structure of Benzene
- Benzene is cyclic hydrocarbon made of six carbon atoms and six hydrogen atoms (C6H6).
- The Kekule structure shows bonding in benzene as alternating carbon double bonds.
- Bond length data and enthalpies of hydrogenation show this to be incorrect.
- Each carbon atom has an un-bonded electron in a p-orbital. The p-orbitals merge to create two rings of electron density, spreading the electrons throughout the carbon ring (delocalised electrons).

- Due to the rings of delocalised electrons, benzene is easily attacked by electrophiles.
- Compounds that contain cyclic carbon rings with delocalised electron bonding (π bonding) are called aromatic compounds.
- The study of these compounds is called aromatic chemistry.
Benzene Reactions
- Electrophiles (electron deficient species) are attracted to the ring of delocalised electrons in benzene; nucleophiles are repelled by the delocalised electrons.
- Benzene easily reacts with strong electrophiles.
- Electrophiles react with benzene in electrophilic substitution reactions, forming a bond with a carbon atom in the ring and forcing the hydrogen atom already bonded to the carbon atom to be lost.
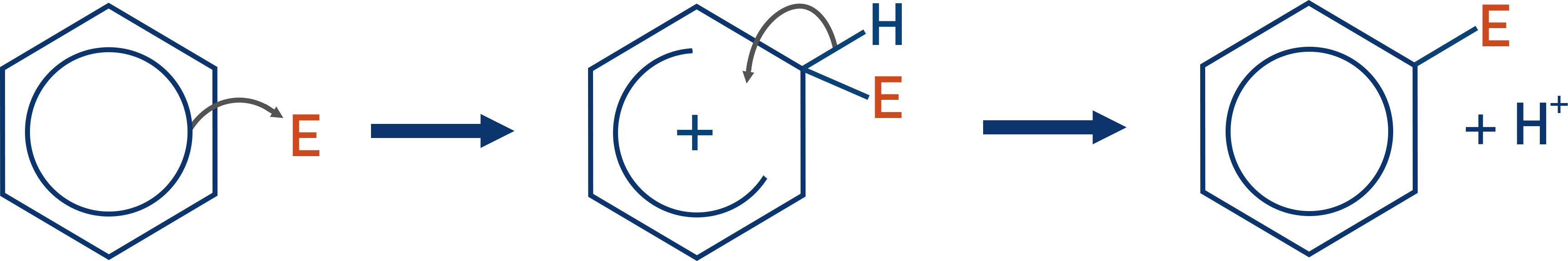
Nitration of Benzene
- The nitration of a benzene is an electrophilic substitution reaction, with a nitronium ion, NO+2, acting as the electrophile.
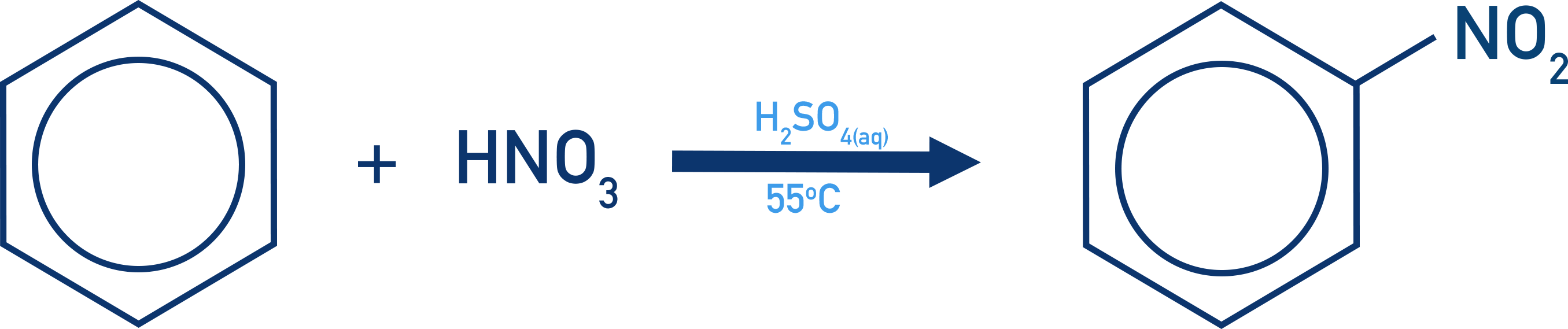
- The nitronium ion is produced by reacting concentrated nitric acid with concentrated sulfuric acid.
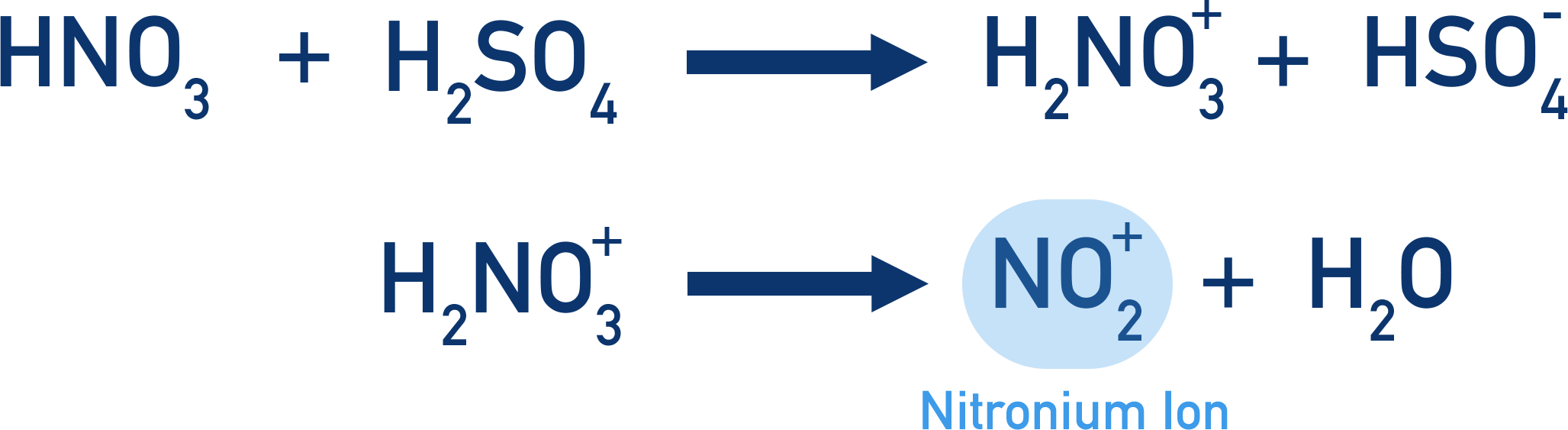
- The reaction is carried out at 55oC and produces a nitroarene.
Acylation of Benzene
- Acylation of benzene involves the substitution of an acyl group onto a benzene ring:
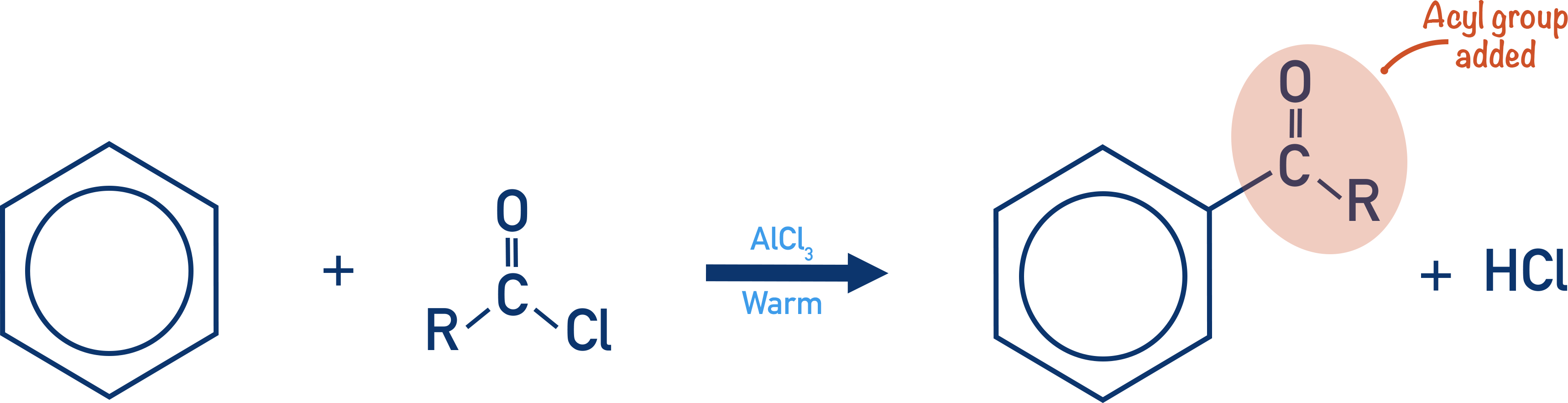
- Acyl chlorides are reacted with a halogen carrier (AlCl3) to form an acyl ion, which acts as a strong electrophile and reacts with the benzene ring in an electrophilic substitution reaction.
- The reaction requires warm conditions and HCl is formed as a product.
Alkylation of Benzene
- Alkylation of benzene involves the substitution of an alkyl group onto a benzene ring.
- Alkyl groups are carbon chains that bond onto another carbon chain (e.g. methyl).
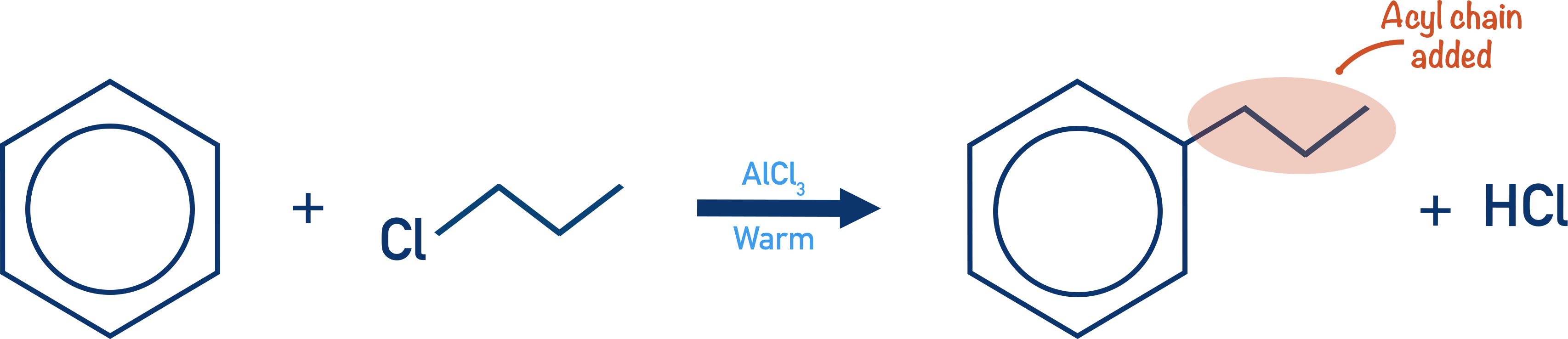
- Halogenoalkanes are reacted with a halogen carrier to produce an R group with a positive charge, which can act as the required electrophile.
- The reaction requires warm conditions and HCl is formed as a product.
Bromination of Benzene
- The bromination of benzene involves the electrophilic substitution of a bromine atom onto a benzene ring:
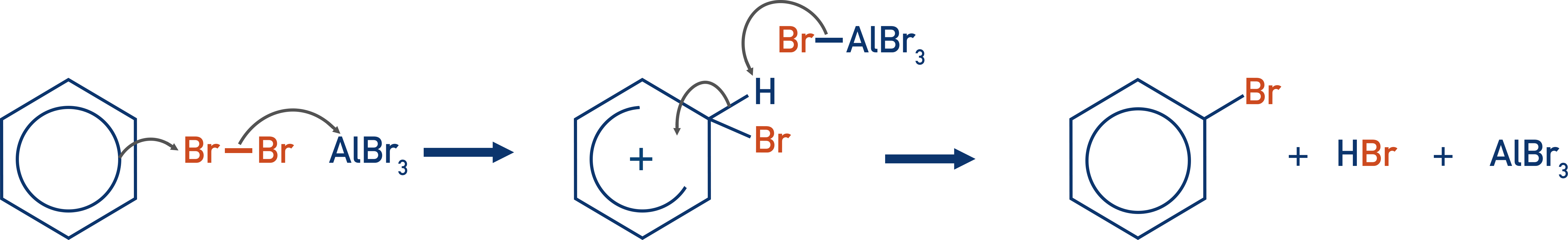
- A halogen carrier is needed (usually AlBr3) to create an electrophile, as the delocalised electrons in the benzene are unable to polarise the bromine molecule sufficiently (unlike with bromine and alkenes).
- Benzene reacts with bromine in electrophilic substitution reactions.
- A catalyst is needed.
- Alkenes and cyclohexene react with bromine in electrophilic addition reactions.
- No catalyst is needed.
- Benzene reacts with bromine in electrophilic substitution reactions.
Phenol
Phenol
- Phenol is an organic molecule that has a benzene ring with a hydroxyl group (OH) attached.
- The lone pair of electrons on the oxygen atom merge with the pi-bonding system in benzene.
- The OH group can release a proton because the negative charge that forms on the oxygen atom can be spread through the pi-bonding system in benzene.
- Phenol can act as a weak acid.
- Alcohols are not weak acids as a negative charge on the oxygen atom is not stabilised.
Reactions of Phenol
- The hydroxyl group (OH) in phenol ‘activates’ the benzene ring, making it more reactive with electrophiles.
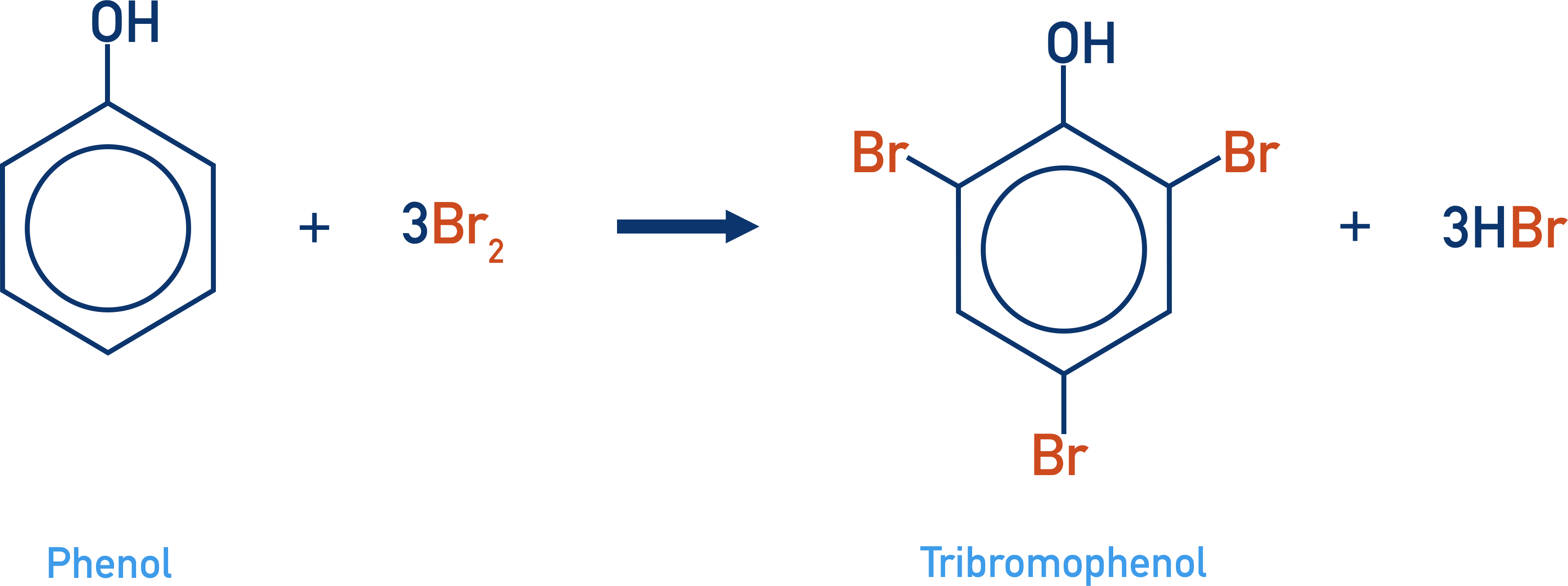
- Phenol is able to react directly with bromine (unlike benzene) in multiple substitution reactions to produce 2,4,6-tribromophenol.
- Nitration of phenol is easier than the nitration of benzene. Only nitric acid (HNO3) at room temperature is required (for nitration of benzene: nitric acid, sulfuric acid and temperature of 55oC).
- The OH group in phenol has a directing effect, meaning substitutions are most likely to occur on the second, fourth and sixth carbon (carbon one is occupied by the OH group).
Amines and Amides
Amines
- Amines contain a carbon chain bonded to a nitrogen atom that is bonded to either hydrogen or another carbon chain (alkyl).
- Amines can be primary, secondary or tertiary (quarternary amines exist as positively charged ions).
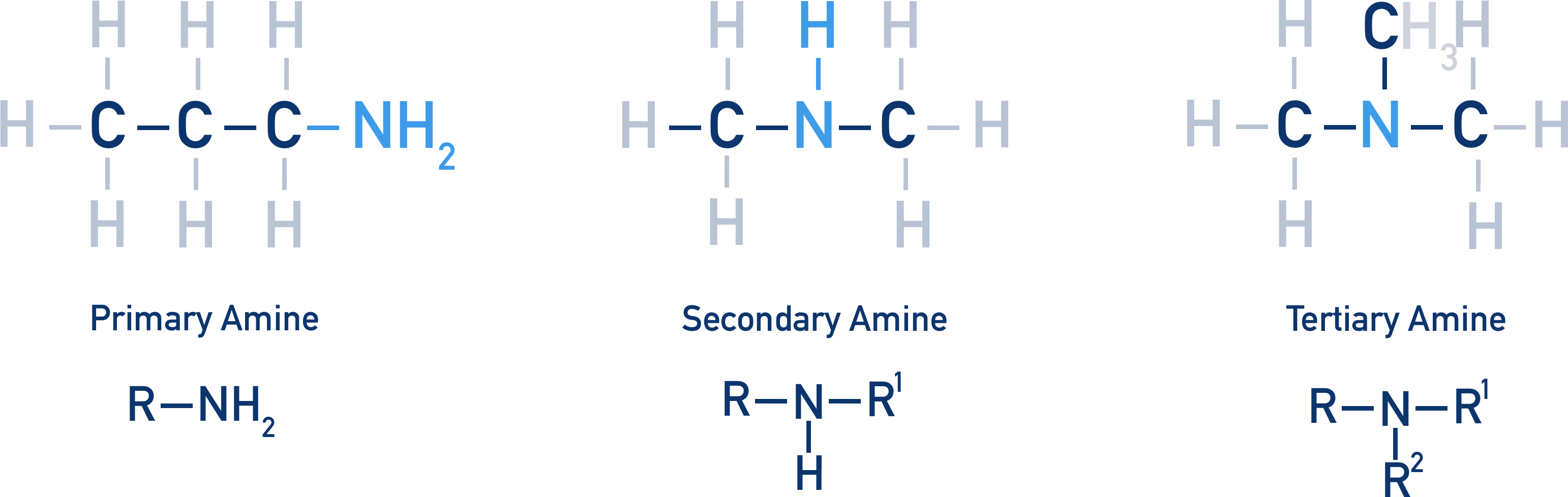
- A lone pair of electrons on the nitrogen atom enables amines to act as bases (and nucleophiles).
- Primary amines are less basic than secondary amines, and secondary amines are less basic than teritiary amines.
- This is due to the positive inductive effect of alkyl chains.
- Aromatic amines are weak bases as the benzene ring is electron withdrawing, making the lone pair of electrons on the nitrogen less available to accept a proton.
- Primary amines are less basic than secondary amines, and secondary amines are less basic than teritiary amines.
Producing Primary Amines
- Primary amines are formed by the reaction of a halogenoalkane with ammonia.
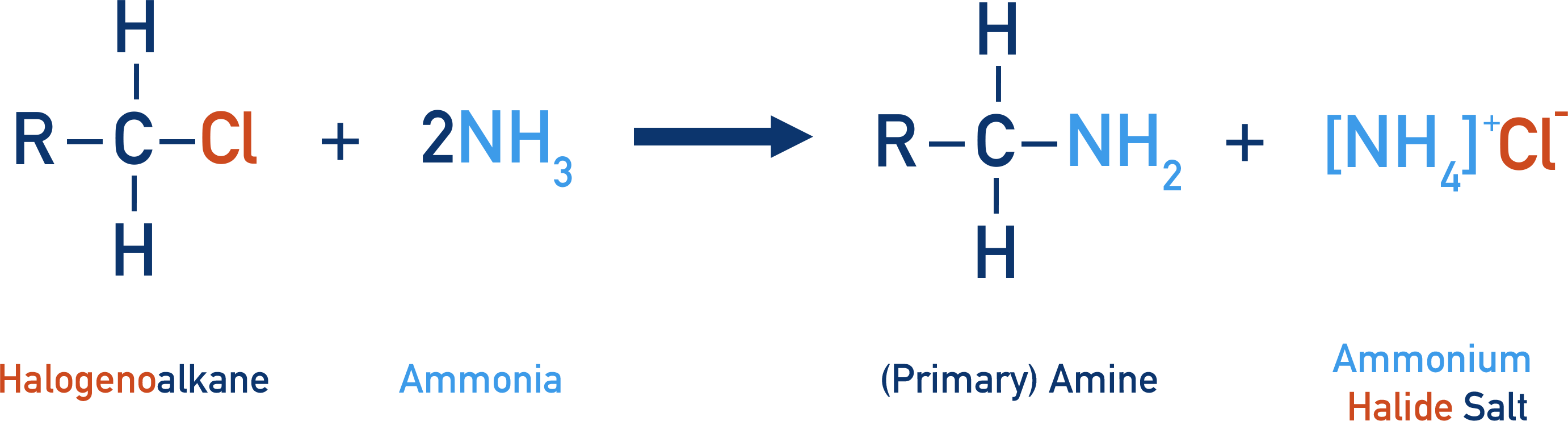
- Ammonia acts as a nucleophile and is substituted for the halogen, making the mechanism nucleophilic substitution.
- Ammonium ions are formed at the end of the reaction because the ammonia (not the halide ions) removes a proton from the positively charged nitrogen in the intermediate.
Producing Secondary and Tertiary Amines
- If a halogenoalkane is reacted with an excess of ammonia, a primary amine is formed.
- When insufficient ammonia is present to undergo substitution with all the halogenoalkane molecules, the primary amines that did form undergo further substitution and can become secondary, tertiary and quaternary amines.
- Once four substitutions have happened, the nitrogen has four bonds and forms a positively charged ammonium ion. This quaternary ammonium ion is attracted to negative halide ions, forming a quaternary ammonium salt.
Producing Aromatic Amines
- Aromatic amines are formed by the reduction of a nitroamine.
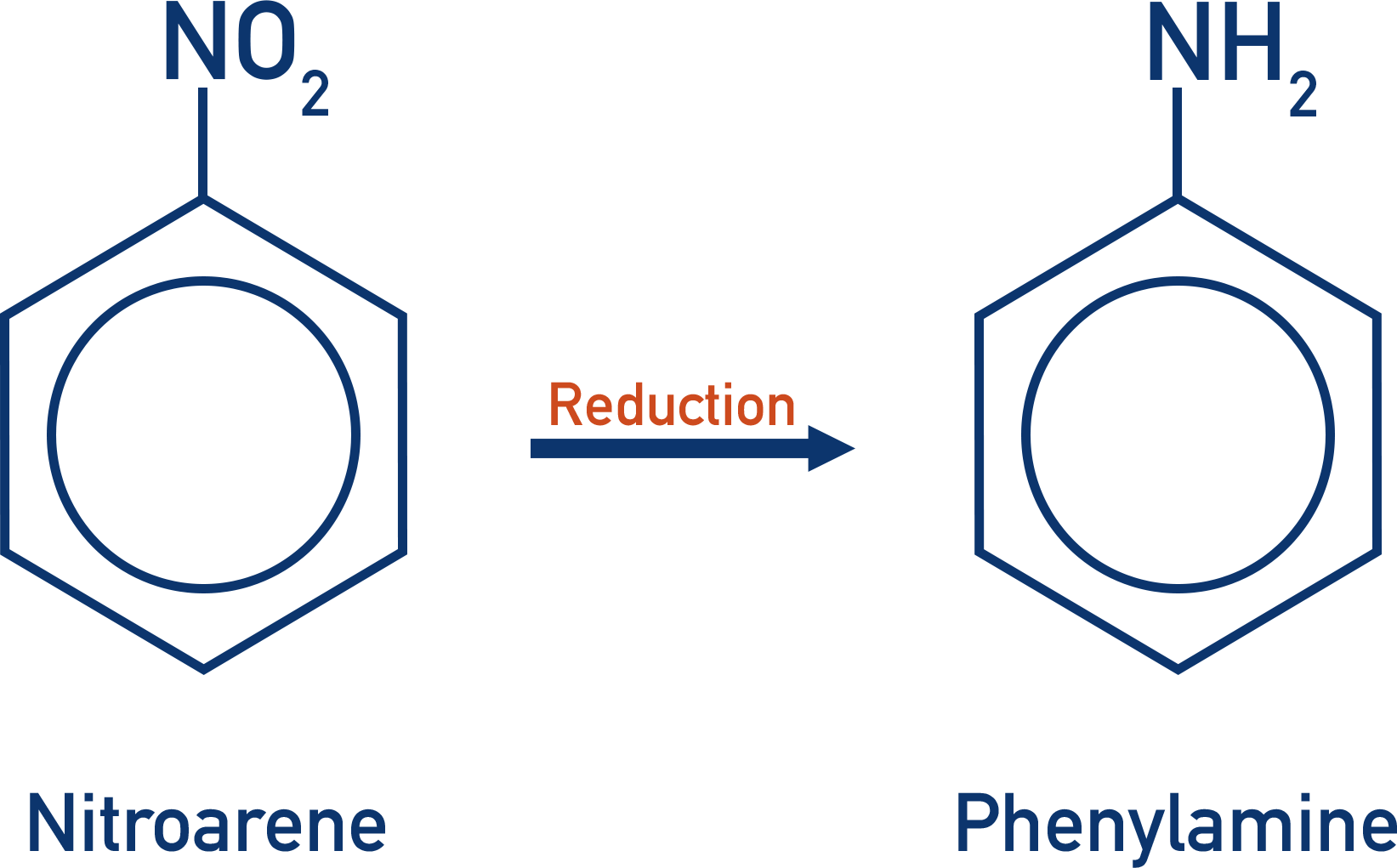
- A tin catalyst and concentrated hydrochloric acid are used under reflux conditions (reflux conditions are essential to stop gaseous vapors of hydrochloric acid being released).
- Acidic conditions result in an ammonium ion being formed, which is converted into an amine group by adding sodium hydroxide.
Amides
- Amides are carboxylic acid derivatives that have a carbonyl group with an amine attached to the carbon from the carbonyl. They are generally unreactive.
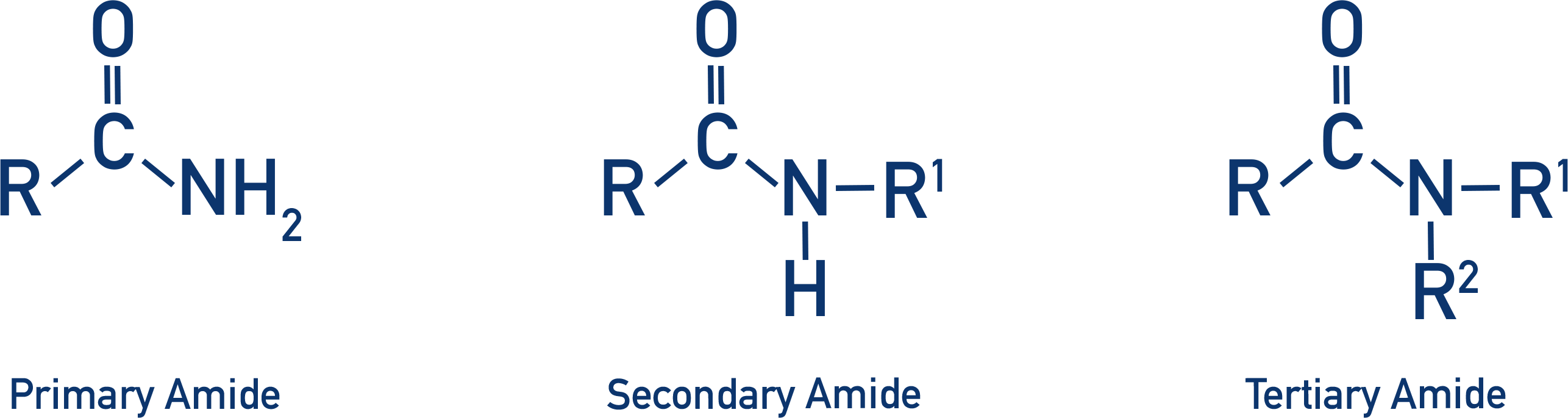
- Amides can be primary, secondary and tertiary.
- (Primary) amides are formed from acyl chlorides and ammonia in a nucleophilic addition-elimination reaction, producing an ammonium chloride salt.
- Secondary amides are formed from acyl chlorides and a primary amine.
- Tertiary amides are formed from acyl chlorides and a secondary amine.
Dyes
Organic Dyes
- Organic dyes are compounds bonded to materials to give the material a specific colour.
- Within organic dyes, electrons are excited from low energy to high energy by absorbing energy from the visible part of the electromagnetic spectrum.
- The part of the dye where the excitation of electrons occurs is called the chromophore.
- Most organic molecules absorb higher energy waves (ultraviolet light – not visible) to excite electrons.
- Increased levels of electron delocalisation decrease the energy required to excite electrons and enable visible light to be absorbed by the molecule, forming a chromophore.
- Wavelengths of light not absorbed by the chromophore are scattered by the dye and observed as the complimentary colour to the colour of light absorbed by the dye.
Preparing Azo Dyes
- The chromophore in an organic dye requires a high level of electron delocalisation.
- Azo dyes are compounds formed from a diazonium salt and an aromatic compound (usually phenol).
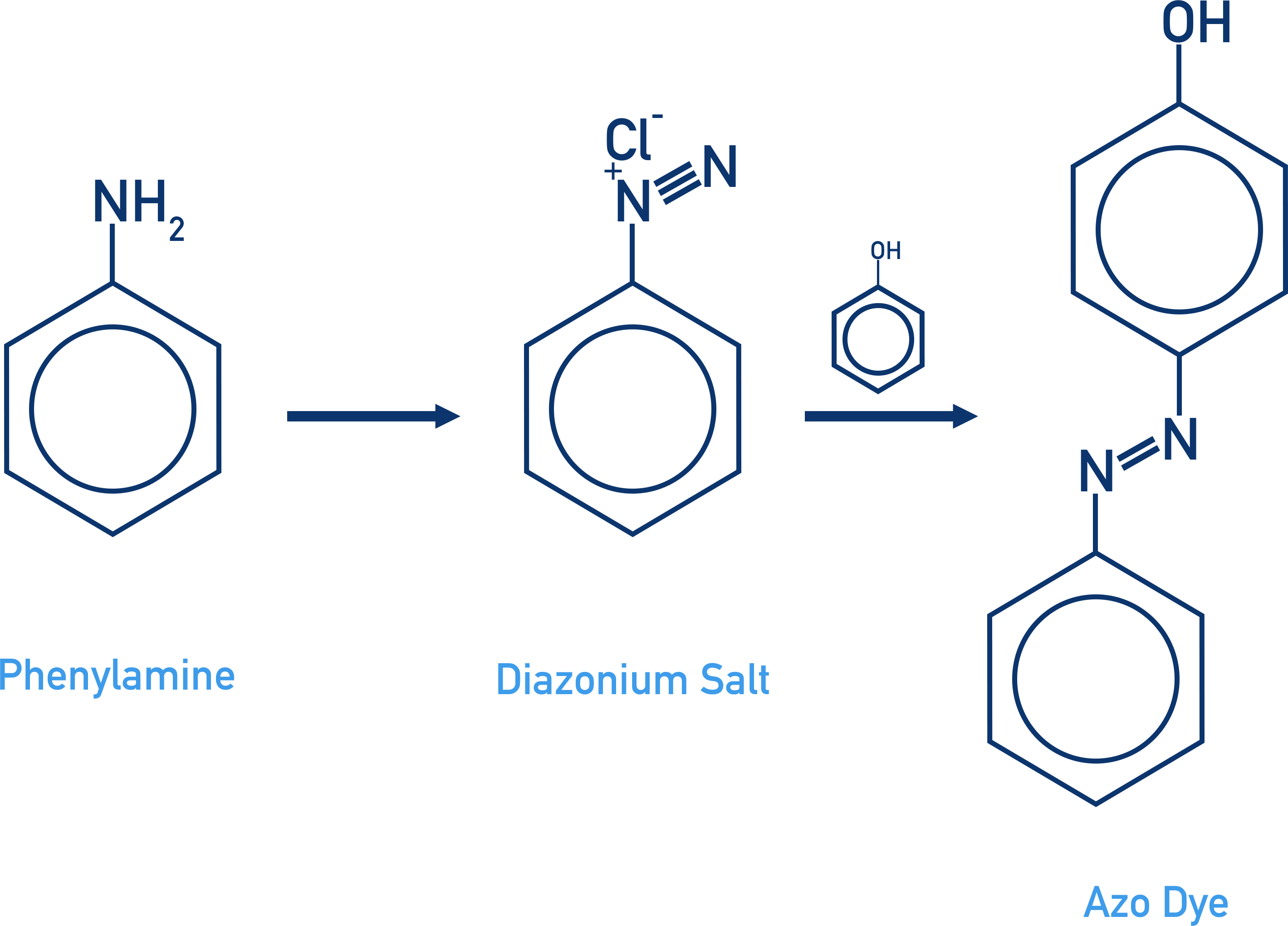
- The formation of azo dyes happens in two stages:
- A diazonium salt is formed from the reaction of phenylamine with nitrous acid and hydrochloric acid.
- The diazonium salt is coupled with the phenol by reacting them together with sodium hydroxide.
- Diazonium salts are highly unstable, and to stop them decomposing during the reaction low temperatures are used (5oC).
Polymerisation
Addition Polymerisation
- Polymers are long chain molecules made of repeating units bonded together.
- Molecules that form repeating units are called monomers.
- Alkenes can undergo addition polymerisation to create polyalkenes.
- The double carbon bond can be opened to enable each carbon from the double bond to form new bonds with another repeating unit.
Condensation Polymerisation
- Condensation polymers are formed when monomers bond together and one water molecule is released per bond.
- To break apart a condensation polymer (into its monomers), a water molecule is needed and a hydrolysis (breaking of bond with water) reaction happens.
- Polyamides are formed from the reaction of a di-carboxylic acid with a di-amine. The links between each monomer are called a peptide links.
- Polyesters are formed from the reaction of a di-carboxylic acid with a di-ol in the presence of an acid.
Amino Acids and Proteins
- Amino acids are a collection of different organic molecules that all contain an amine group and a carboxylic acid group.
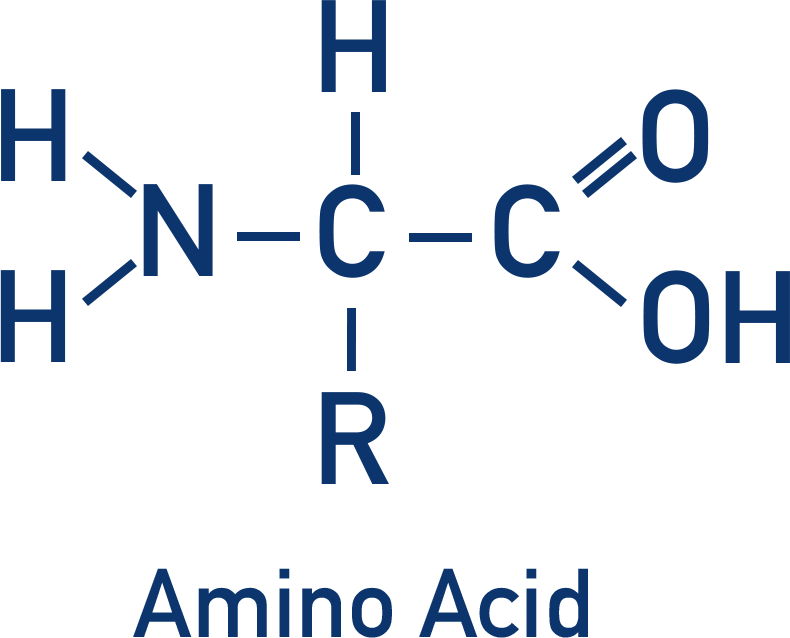
- Amino acids join together to form proteins.
- The amine group in an amino acid can accept a proton (basic) to become a positive ion.
- The carboxylic acid group in an amino acid can donate a proton (acidic) to become a negative ion.
- Different amino acids lose or gain protons at different pHs.
- A zwitterion is an amino acid that has lost a proton from its carboxylic acid group but gained a proton at its amine group – forming negatively and positively charged groups, but no overall charge.
- Zwitterions are formed at an amino acid’s isoelectric point.
Properties of Polymers
- Longer chain polymers have higher melting points than shorter chain polymers.
- The larger the contact surface between polymers, the greater the number of intermolecular forces that can form between them, resulting in a higher melting point.
- Branched chain polymers have lower melting points than straight chained polymers as there is less contact surface between them, resulting in fewer intermolecular forces.
- Addition polymers are not biodegradable.
- Condensation polymers are biodegradable and are broken down in hydrolysis reactions.
