Video Tutorial Phenol Reactions
Quick Notes Reactions of Phenol
- The hydroxyl group (OH) in phenol ‘activates’ the benzene ring, making it more reactive with electrophiles.
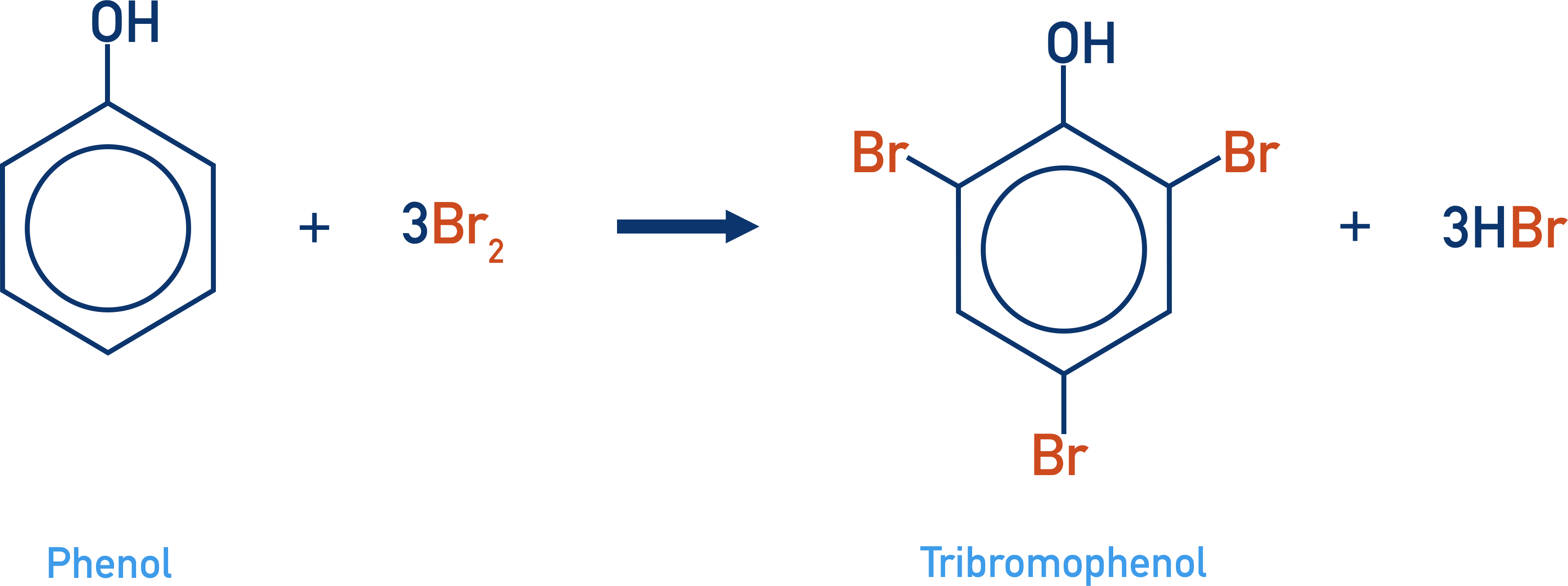
- Phenol is able to react directly with bromine (unlike benzene) in multiple substitution reactions to produce 2,4,6-tribromophenol.
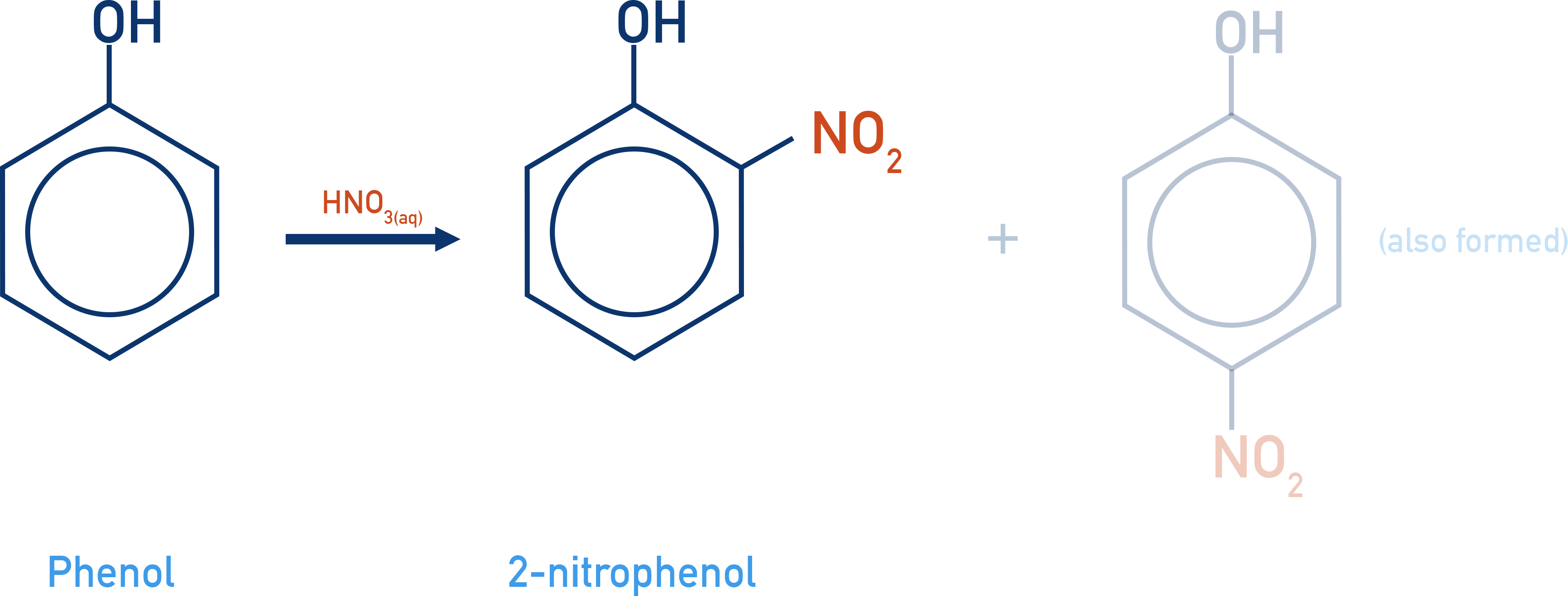
- Nitration of phenol is easier than the nitration of benzene. Only nitric acid (HNO3) at room temperature is required (for nitration of benzene: nitric acid, sulfuric acid and temperature of 55oC).
- The OH group in phenol has a directing effect, meaning substitutions are most likely to occur on the second, fourth and sixth carbon (carbon one is occupied by the OH group).
Full Notes Reactions of Phenol
Phenol can react directly with bromine, whereas benzene is unable to.

The hydroxide group is described as ‘activating’ because of the extra electron density it gives the delocalised electron ring, making the molecule more reactive with electrophiles.
Bromination
The increased electron density in the ring within phenol (see Phenol) enables the ring to polarise a bromine molecule enough to form an electrophile.
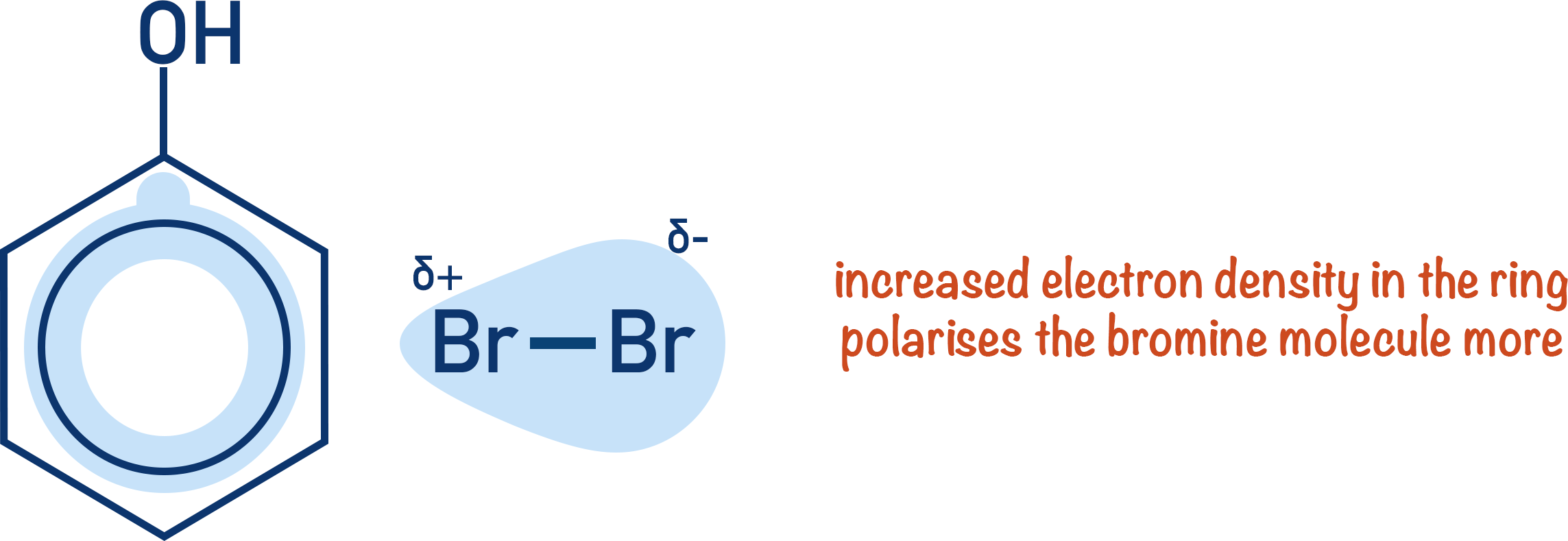
Benzene does not have enough electron density in the ring for this to happen. This is why benzene needs a halogen carrier to react with bromine but phenol does not.
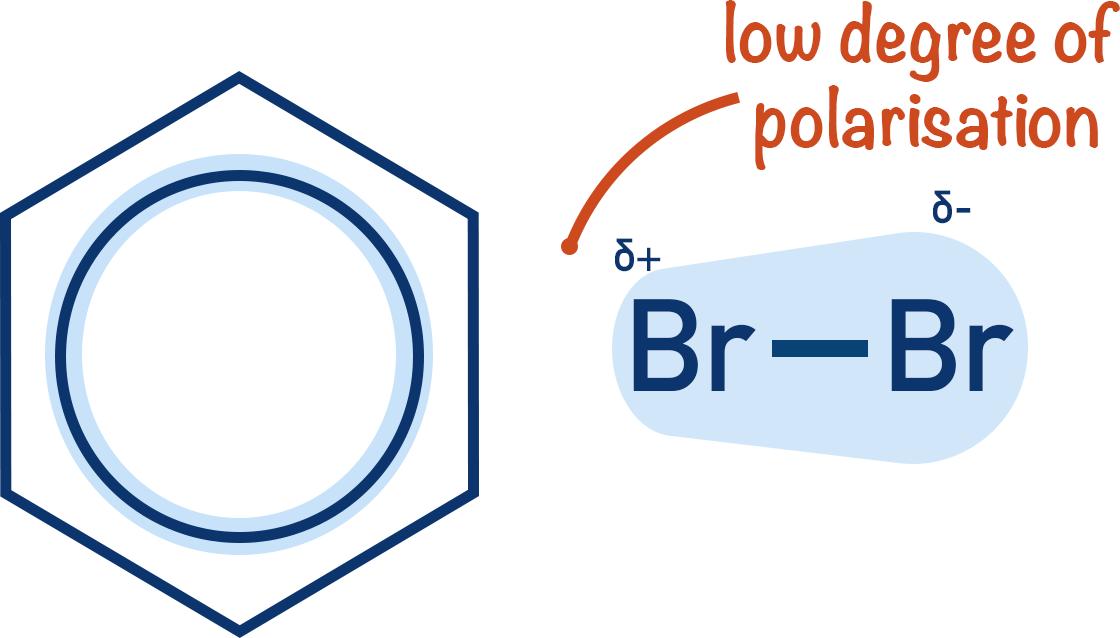
A lone pair of electrons on the oxygen atom in phenol is donated or ‘merged’ into the pi-bonding system in the benzene ring. The electron density inside the benzene ring increases as a result; this increased electron density enables phenol to react with electrophiles more easily than benzene can.
Phenol also undergoes multiple substitutions, forming 2,4,6-tribromophenol.

Benzene only undergoes a single substitution, forming bromobenzene.
Nitration of Phenol
Just like with benzene, phenol can react with a nitronium ion (NO+2), and a nitro group is added onto the carbon ring.
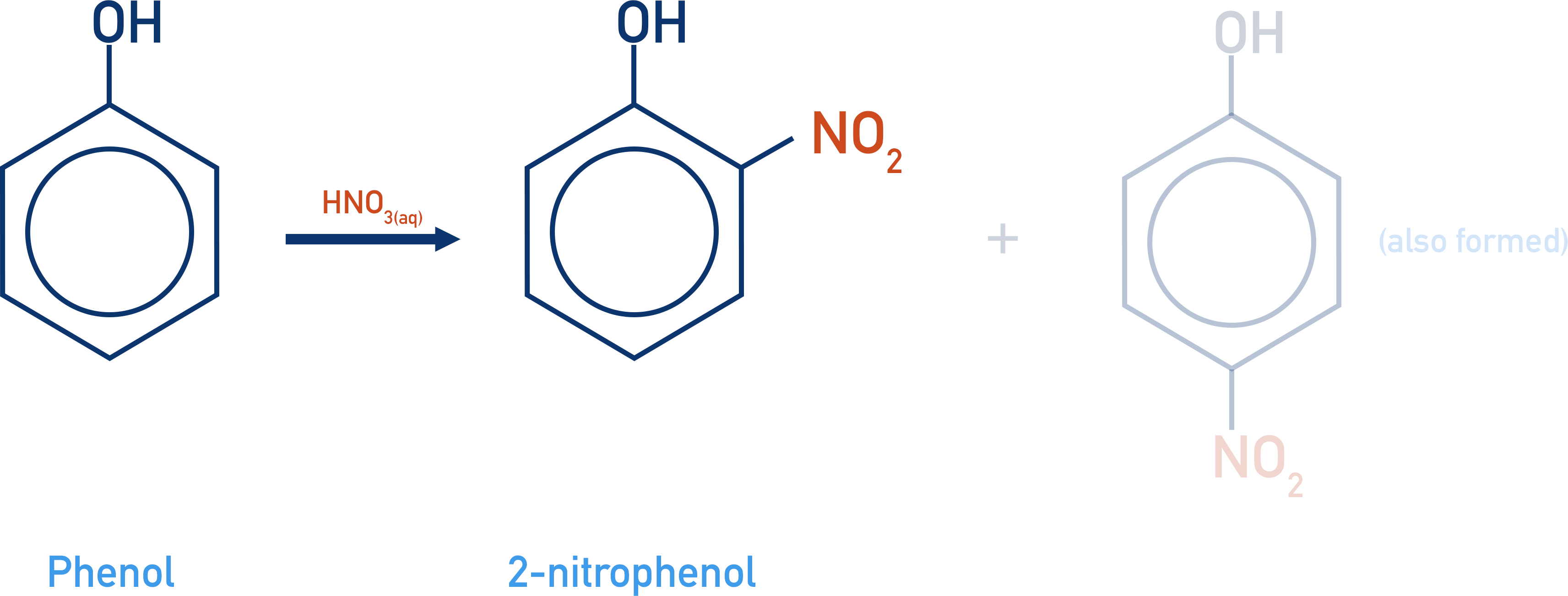
As seen with bromination (see above), phenol is more reactive with electrophiles than benzene. So, the nitration of phenol happens more easily than the nitration of benzene. Room temperature and dilute nitric acid are the only reactants and conditions required (with the nitration of benzene, higher temperatures (55oC) and concentrated sulfuric acid are also necessary).
Directing Effect of Phenol
The positions that an electrophile substitutes to in phenol are as a result of electrons from the oxygen atom being moved around the ring. This detail is not needed for A-level Chemistry, but it does explain why the above occurs.
If we draw out the Kekulé structure of the phenyl group (benzene ring) in phenol, it’s easier to see how the electrons from the negatively charged oxygen cause the positions of double bonds to move around the ring.
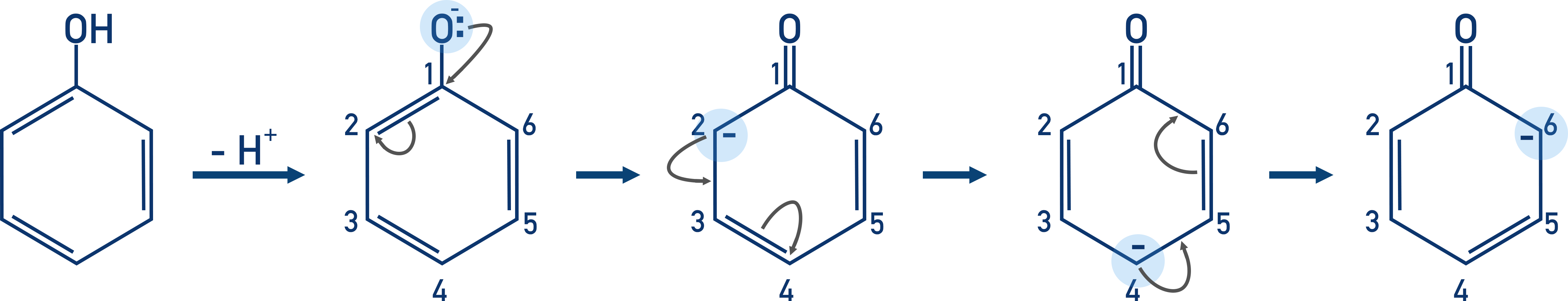
The phenol dissociates in solution and releases a H+ ion from the hydroxide group.
The electrons added to the ring from the oxygen atom repel the electrons in the double bond nearest the carbon attached to the oxygen. The repulsion moves the electrons in the bond to the next carbon, making it negatively charged.
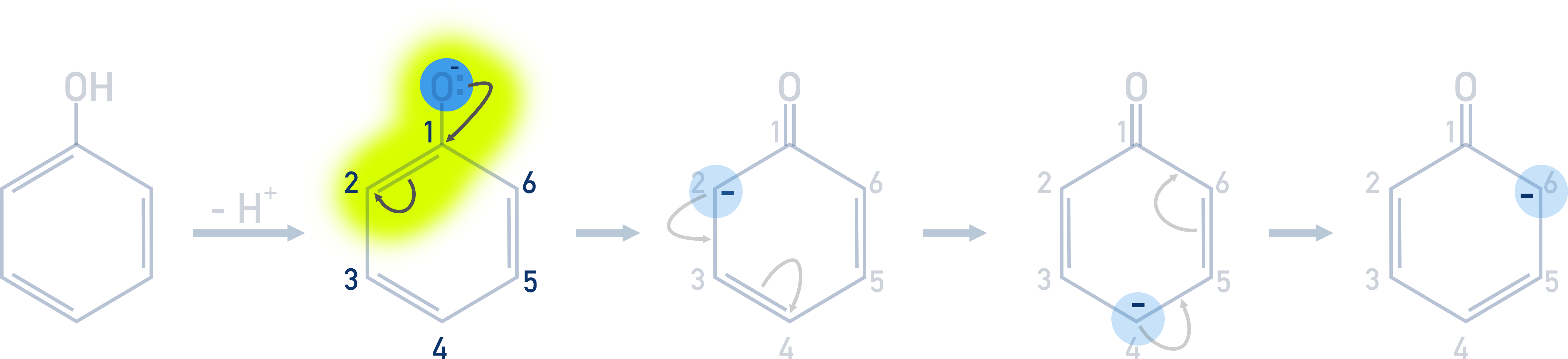
This negatively charged carbon is more likely to bond with an electrophile than any other carbon atoms in the ring, this is why the electrophile bonds to the 2nd carbon. Making it 2-bromophenol in the case of bromination.
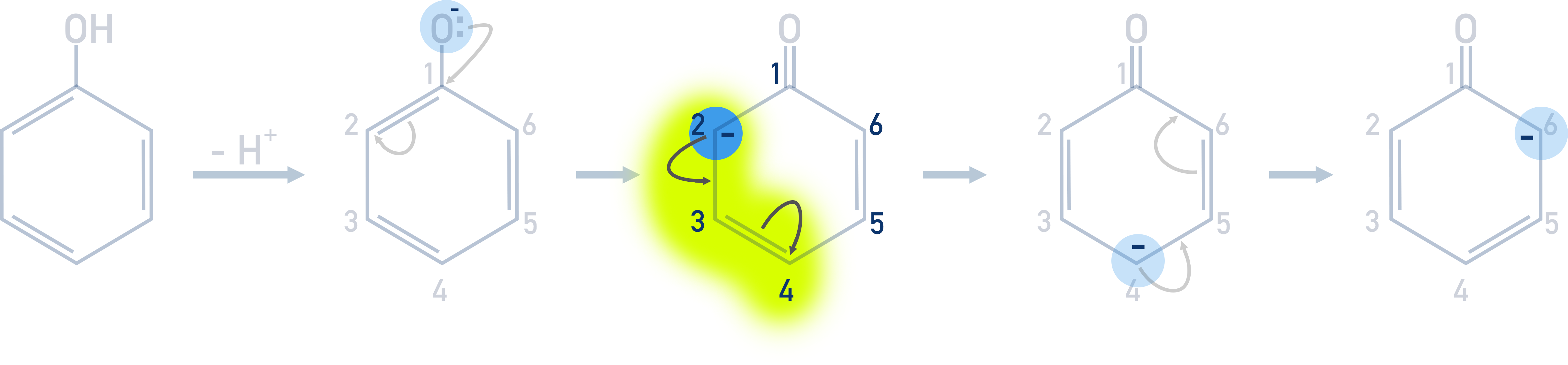
The negative charge does not just remain on the 2nd carbon though – it causes repulsion with the next closest double bond, forcing those electrons onto the second furthest carbon (4th carbon), making the 4th carbon negatively charged. This 4th carbon is now the most likely to react with an electrophile, so the next electrophilic substitution takes place on the 4th carbon.
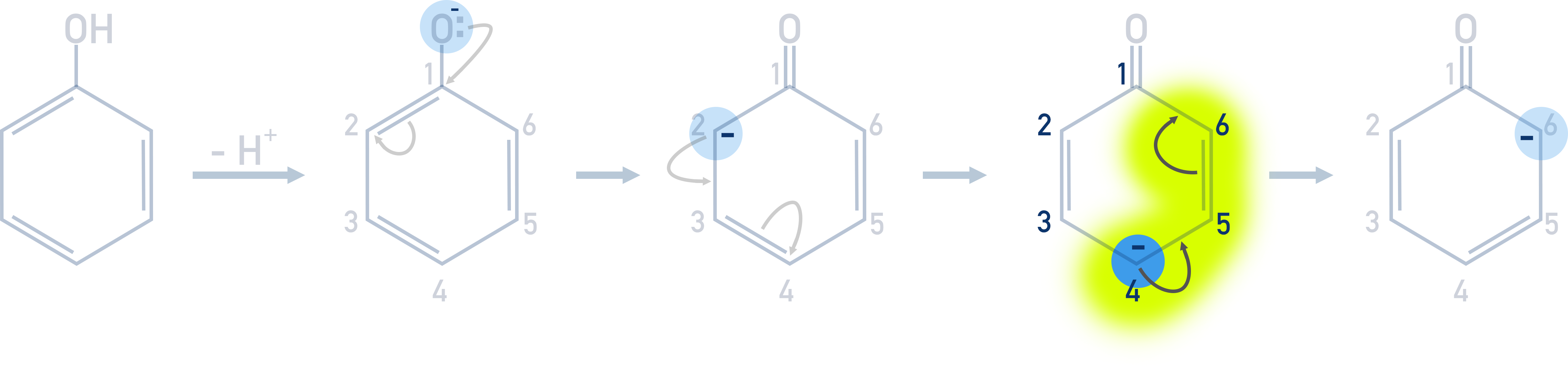
This same process also occurs with the 6th carbon.
The movement of charge around a molecule may look very strange (and for A-level students – you do not need to know this), but it is just a way to show how the charge can be spread around the molecule. The possible structures that result from the movement of delocalised electrons around a molecule are called resonance structures.
We’ve launched our new site! 🎉
Course-specific notes with built-in search!
AP • A-Level (AQA • CIE • Edexcel • OCR) • IB • NCERT 11 + 12
over 750+ new pages and 3,500 images.
Visit the new homepage
