Video Tutorial Electron Orbitals
Quick Notes Acylation of Benzene
- Acylation of benzene involves the substitution of an acyl group onto a benzene ring:
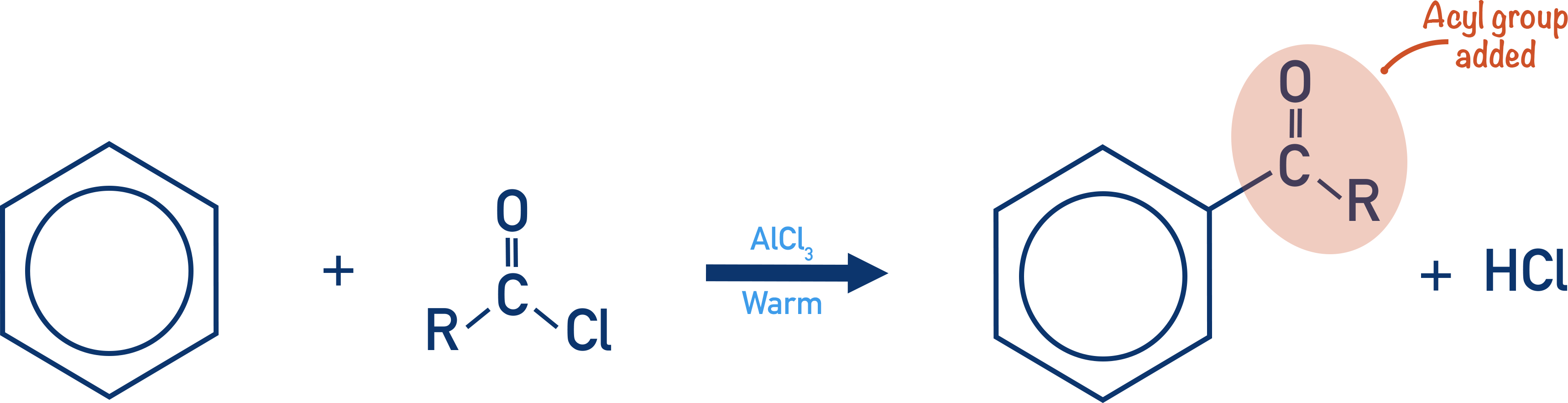
- Acyl chlorides are reacted with a halogen carrier (AlCl3) to form an acyl ion, which acts as a strong electrophile and reacts with the benzene ring in an electrophilic substitution reaction.
- The reaction requires warm conditions and HCl is formed as a product.
Full Notes Acylation of Benzene
Acylation of benzene refers to the substitution of an acyl group (RCO-) onto a benzene ring.

In order to be substituted onto the ring, the acyl group must be an electrophile. Acyl chlorides provide an easy way to form an acyl group with a positive charge on the carbon atom. As with the alkylation of arenes (see Alkylation Reactions), a halogen carrier is used to break the bond between the carbon atom and the chlorine atom in an acyl chloride.
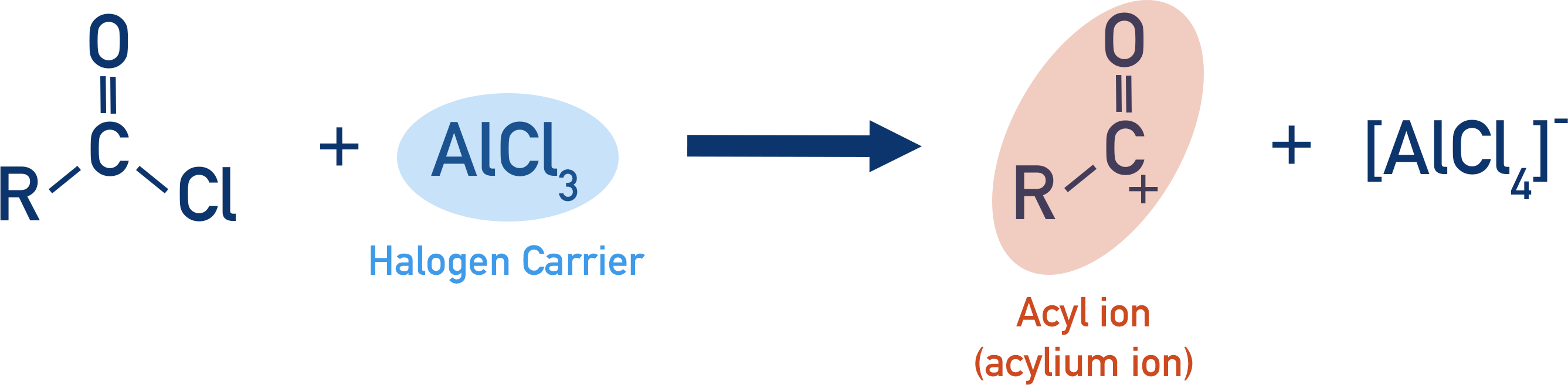
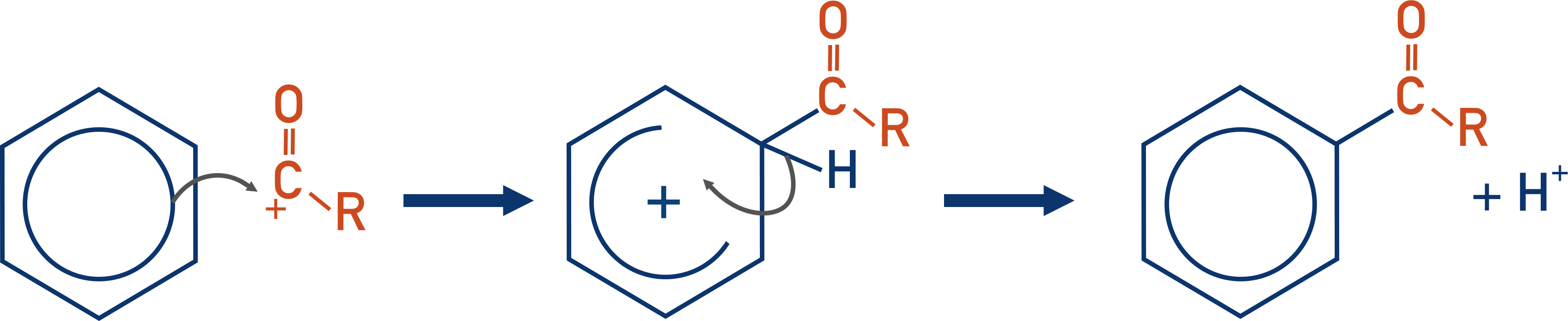
The electrophile formed can then react with benzene in a standard electrophilic substitution reaction. See Benzene Reactions.
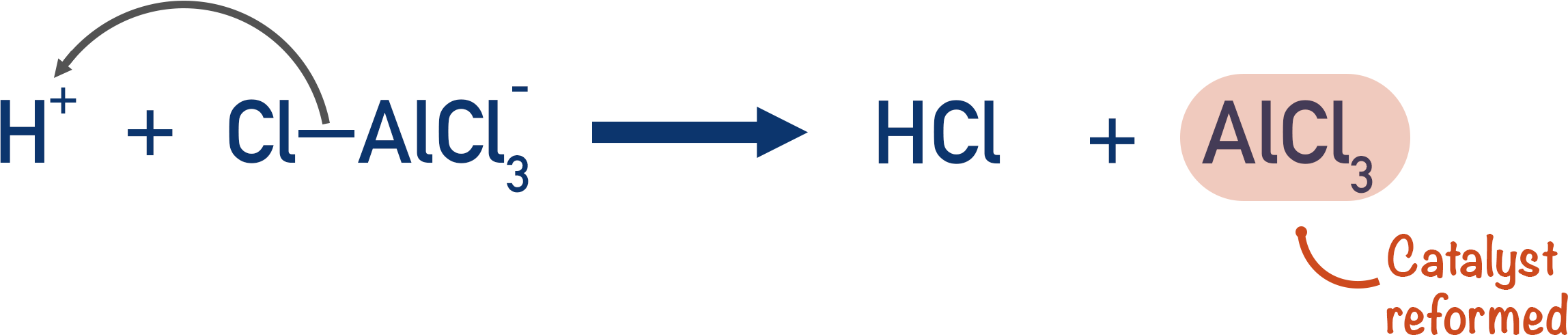
HCl is formed at the end of the reaction. The tetra ion formed from the halogen carrier reacts with the hydrogen ion (H+) released during the reaction, forming HCl and the original halogen carrier again.
We’ve launched our new site! 🎉
Course-specific notes with built-in search!
AP • A-Level (AQA • CIE • Edexcel • OCR) • IB • NCERT 11 + 12
over 750+ new pages and 3,500 images.
Visit the new homepage
