Quick Notes Group 2 Metal Reactions
- Reactivity of the group 2 metals increases going down the group.
- Outer electrons are held less tightly due to increased inner electron shielding, so they are easier to lose, making the metals more reactive.
- Group 2 metals usually form metal hydroxides when they react with water.
- Beryllium is not reactive enough to form a hydroxide.
- Magnesium does react with water but only slowly.
- The solubility of group 2 metal hydroxides increases down the group.
- The pH of the metal hydroxide solutions increases down the group.
- The solutions become more alkaline as more hydroxide ions are released into the solutions (as the solubility of the group 2 metal hydroxide increases).
- The solubility of group 2 metal sulfates decreases down the group, with barium sulfate being completely insoluble.
Full Notes Group 2 Metal Reactions
As you go down group 2, the metal atoms lose their outer electrons more readily, forming positive ions more easily. This is because of increased inner electron shielding causing a weaker attraction between the nucleus and outer electrons. As you go down the group, the metals become more likely to react – their reactivity increases.
Reactions with Water
The trend in reactivity down group 2 can be observed with the reactions of group 2 metals with water.
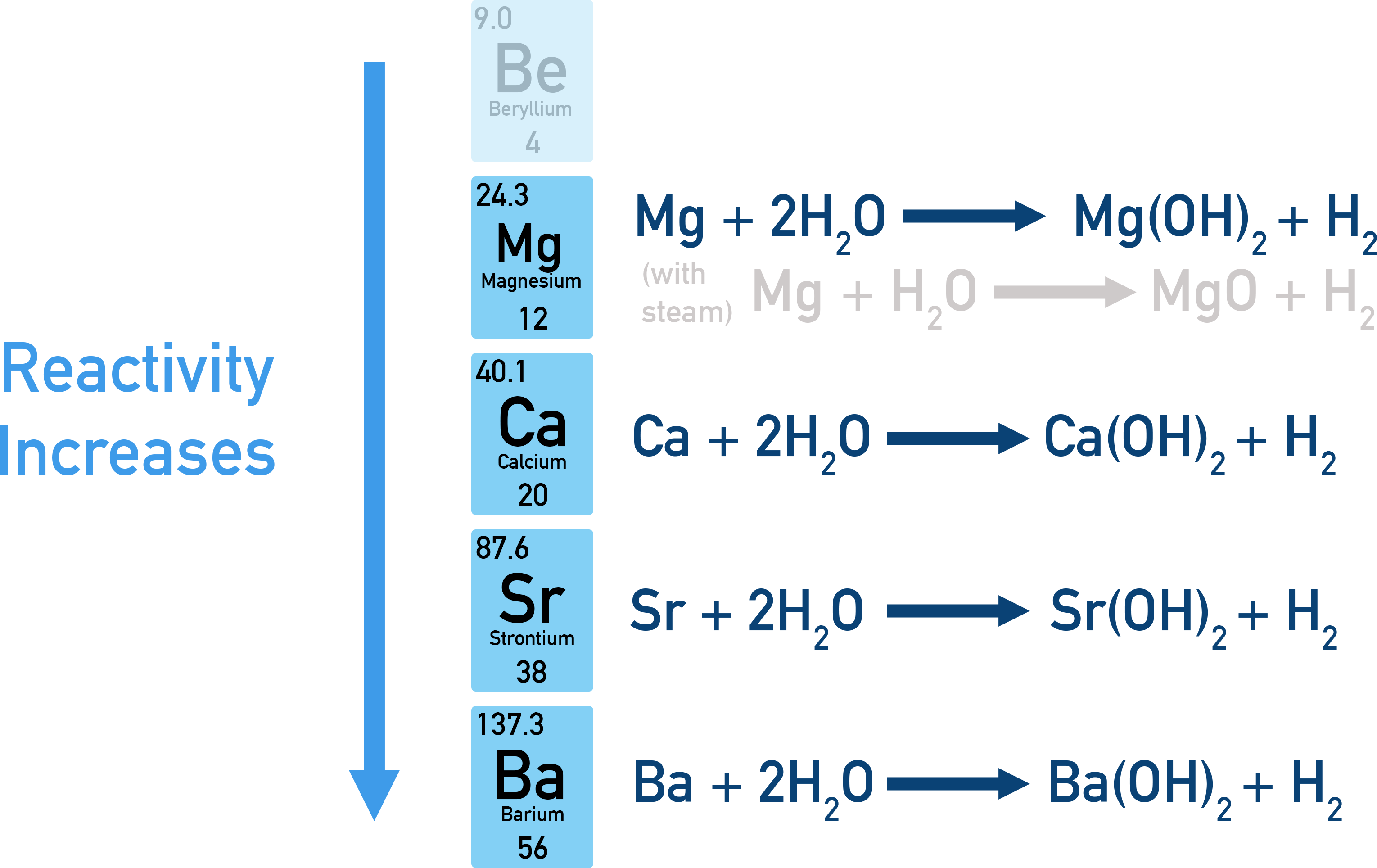
Mg(s) + 2H2O(l) → Mg(OH)2(s) + H2(g)
Magnesium with water is a very slow reaction, producing Mg(OH)2(aq) and hydrogen gas. However, reacting magnesium with steam forms magnesium oxide, in a much faster reaction.
Mg(s) + H2O(g) → MgO(s) + H2(g)
Below magnesium, the group 2 metals react vigorously with water, each producing metal hydroxides.
Ca(s) + 2H2O(l) → Ca(OH)2(aq) + H2(g)
Solubility of Hydroxides
The metal hydroxides produced from reacting group 2 metals with water (see above), dissolve differently in solution. How well a substance dissolves in water is described as its solubility. A substance with a high solubility will readily dissolve in water, a substance with a low solubility will not dissolve easily in water.
For group 2 hydroxides, solubility in water increases as you go down the group.
Magnesium hydroxide, Mg(OH)2 is only sparingly soluble in water, whereas strontium hydroxide, Sr(OH)2 and barium hydroxide, Ba(OH)2 are highly soluble in water.

Metal hydroxides are ionic compounds that, when dissolved in water, ‘split’ apart into their negative ions (anions) and positive ions (cations), which make up the substance.
For example, Ba(OH)2(s) in water dissolves to release Ba2+(aq) and 2OH-(aq).When a metal hydroxide dissolves in water, an alkaline solution is formed as it contains OH- ions. Alkaline solutions are solutions that contain OH- ions, and they have a pH of above 7.
The group 2 hydroxides dissolve more readily as you go down the group, causing the solutions formed to become more alkaline, as there are more OH- ions released by the same mass of hydroxide dissolving.
The ability of group 2 hydroxides to form alkaline solutions can be very useful.
Lime (Ca(OH)2) is widely used in agriculture; it’s applied to fields to help raise the pH of acidic soil, making growing conditions more favourable for some crops.
Magnesium hydroxide, Mg(OH)2, is commonly used as indigestion tablets to help neutralise excess stomach acid.
Group 2 Sulfates
Group 2 metal sulfates are ionic compounds, made from a group 2 metal ion (M2+) and a sulphate ion (SO42-).
The solubility of group 2 metal sulfates decreases as you go down the group. MgSO4 is highly soluble, but BaSO4 is insoluble.

The insolubility of barium sulfate can be very helpful in chemistry; it forms the basis of a test to identify sulphate ions in solution.
Sulfate Ion Test
A barium salt solution (commonly BaCl2(aq)) is added to a solution (with dilute nitric acid) that may contain sulfate ions. If sulfate ions are present then insoluble BaSO4 will be formed, which can be identified as a white precipitate.
Barium sulfate is also used in radiography in medicine. Being a metal, barium shows up well in an x-ray, the only problem is it’s highly toxic when ingested. However, barium sulfate is insoluble, meaning the barium is ‘trapped’ in the sulfate compound and cannot do any damage to a patient’s body. A patient can ingest barium sulfate, and when x-rayed we can see where the barium sulfate has travelled to in the patient’s body. This can help identify digestive disorders and irregularities.
