Video Tutorial Metal Aqua Ions - Precipitation Reactions
Quick Notes Metal Aqua Ions - Precipitation Reactions
- Metal aqua complex ions can behave as (very) weak acids in solution.
- High polarisation of the O-H bond in water ligands enables a proton to be removed by a water molecule, and the ligand becomes a negatively charged OH group.
- Equilibriums are established for the removal of a proton from the aqua complex ion.
- Adding hydroxide ions forces the position of equilibrium to favour the release of protons by the complex ions.
- If the number of OH groups in the aqua complex becomes the same as the positive charge of the metal ion, the complex ion becomes a solid precipitate, as it can no longer be dissolved in solution.
- The greater the positive charge of the metal ion, the more acidic the complex ion.
Full Notes Metal Aqua Ions - Precipitation Reactions
The complexes formed with metal ions and water are called aqua-ions. When a solution containing metal aqua-ions is mixed with an aqueous alkali, a solid precipitate is formed. These reactions are called precipitation reactions, and the solid formed is the precipitate of the metal hydroxide. Due to the nature of transition metals, many of the precipitates formed are different colours, see Transition Elements, Colour.
![metal aqua ion complex with hydroxide ions to form precipitate [Cu(H2O)6]2+(aq) forms Cu(OH)2(H2O)4(s)](images/A2Inorganic/complexionchemistry/complexionsaquareactions1.png)
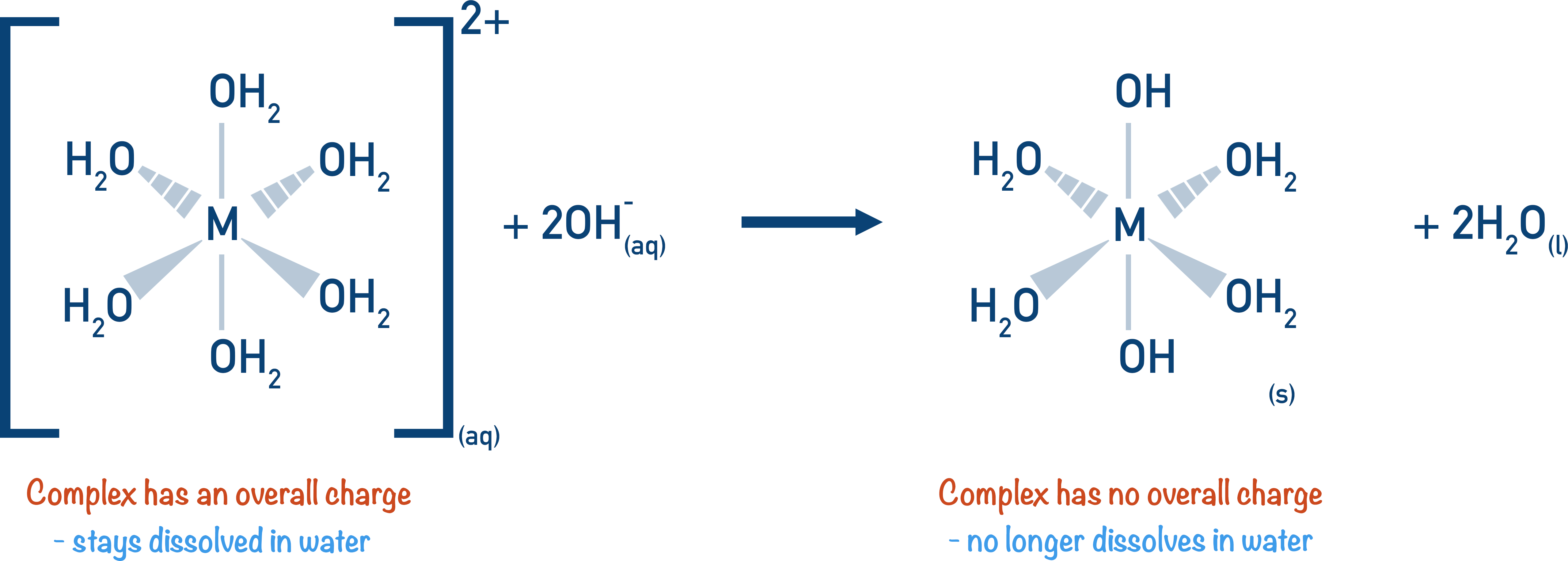
Very simply, the metal aqua-ion is charged, which is why it is not a solid – water is a polar solvent and will readily dissolve charged (and polar) species. When the metal aqua-ion reacts with an alkali it is no longer a charged complex, therefore it will not dissolve in water. This is seen as lots of small solids forming in the solution – what we call the ‘precipitate’.
Why and how this happens is a little more complex.
The metal ion in the aqua-ion complex is positively charged and surrounded by water molecules (ligands). The positive charge of the metal ion polarises the co-ordinate bonds between the metal ion and the water molecules.
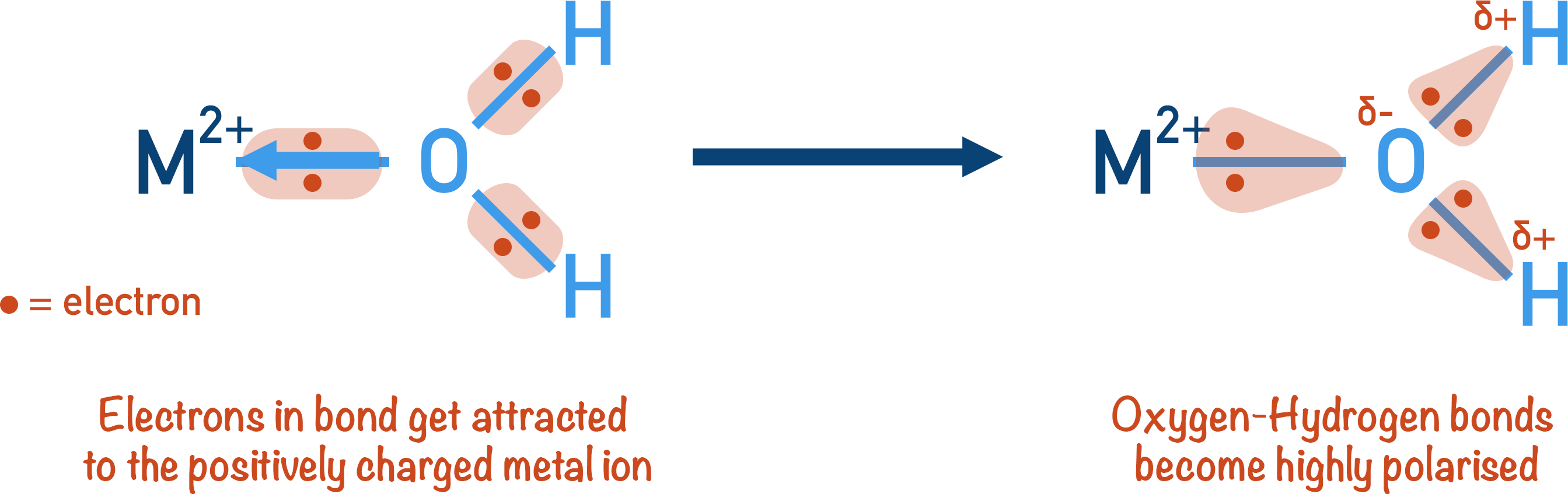
This polarisation pulls electrons away from the oxygen atom in the water molecule. Oxygen is highly electronegative, so it does not want to lose any electron density. To stop this from happening, electrons in the oxygen and hydrogen bonds are pulled towards the oxygen atom more strongly than usual. This creates a very polar bond between the oxygen atom and hydrogen atoms. The more polarised a bond is, the easier it is to break. The O-H bonds in the water molecule are now so weak, a free water molecule can ‘pull’ a proton away from the H2O ligand.
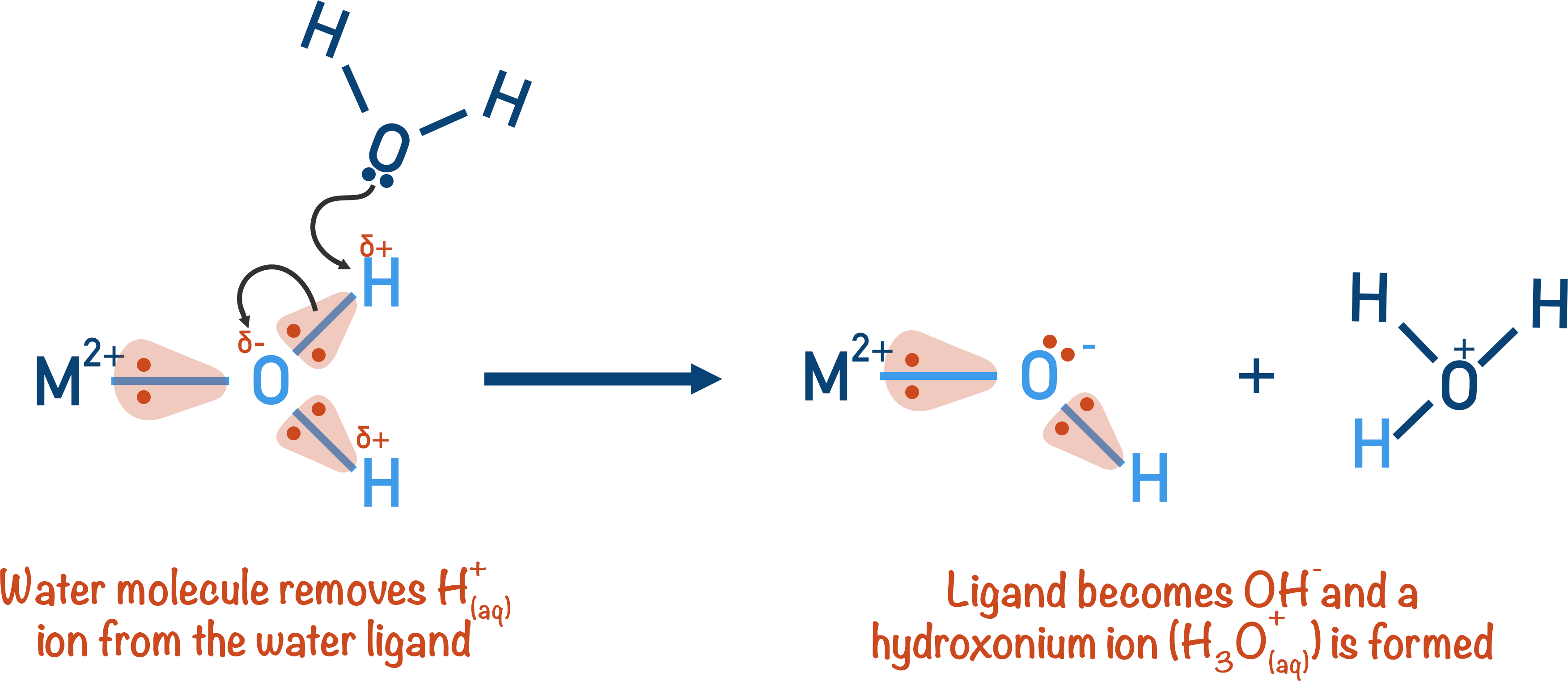
The free water molecule comes near the polarised bond and the lone pair of electrons from the oxygen atom will be attracted to the (very) partially positive hydrogen atom in the ligand and remove it, creating hydroxonium ions (H3O+) – these are shown in equations as H+(aq)ions.
The water molecule that was bonded to the metal ion has now lost a proton and becomes an OH- group. The overall positive charge on the metal aqua-ion decreases by one as a result.
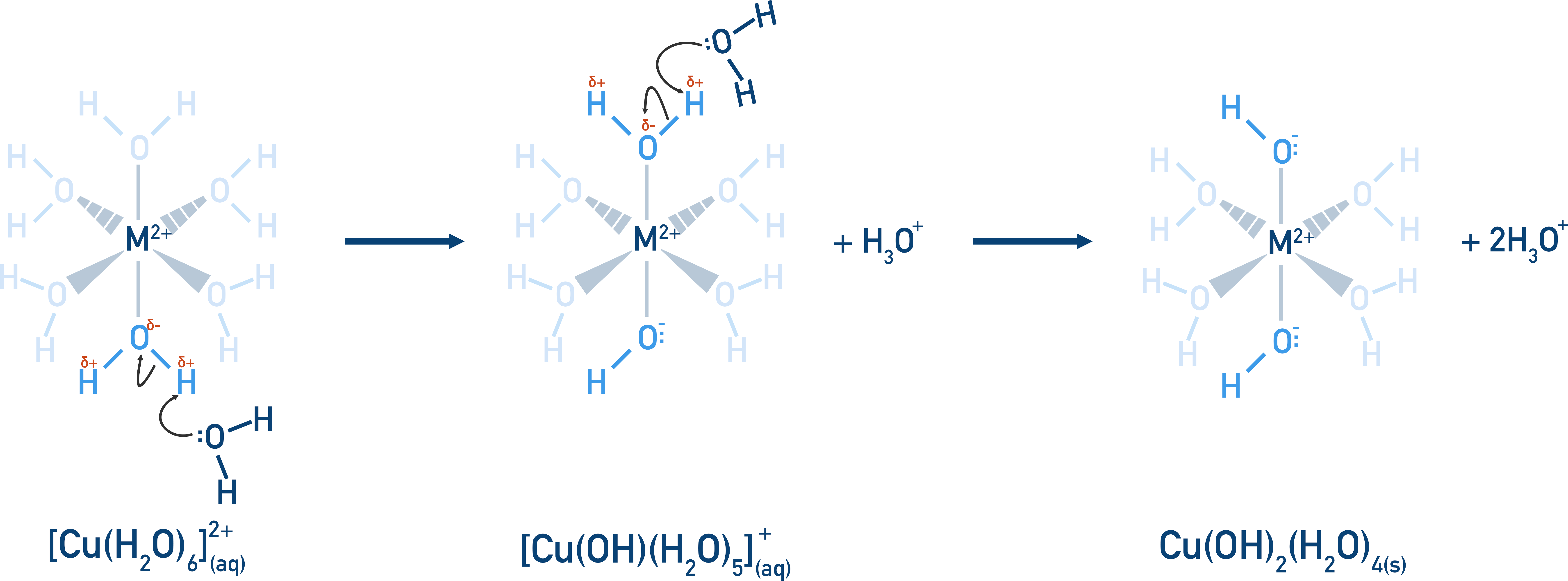
There is still an overall positive charge however, and the positive metal ion is still able to polarise another co-ordinate bond with a water ligand, enabling the same process to occur.
When the number of OH- groups attached to the metal ion is the same as the positive charge of the metal ion, the metal aqua ion no longer has an overall charge and cannot stay dissolved in water. It forms a solid precipitate.
Each of the above steps is in equilibrium, however, and the positions of equilibrium favour the reverse reactions, meaning in reality the precipitate only forms if forced to do so.
![acid equilibrium metal aqua ion [Cu(H2O)6]2+](images/A2Inorganic/complexionchemistry/complexionsaquareactions6.png)
By adding hydroxide ions, OH-(aq), the H+(aq)ions in solution react with the hydroxide ions and the position of equilibrium now moves to oppose this change in H+(aq)ion concentration (by producing more H+(aq)ions).
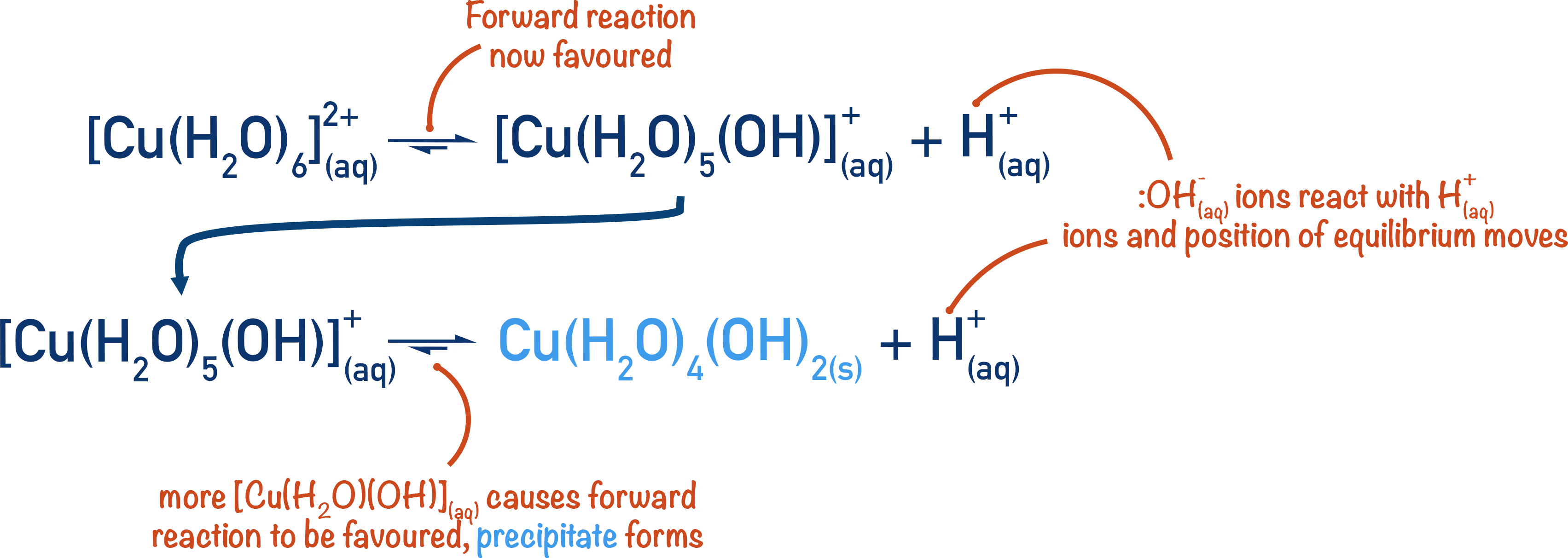
By releasing more H+(aq)ions, more of the [M(OH)(H2O)5]+ complex ion is formed. This then forces the position of equilibrium for the second reaction to favour the forward reaction, producing the solid metal hydroxide in sufficient amounts to be seen as a precipitate.
Metal Aqua-ions as Weak Acids
The polarisation of an O-H bond in the water ligands means the metal aqua-ion can give away a H⁺ ion in solution (as above). This is why metal aqua-ions can behave as weak acids, forming an equilibrium in solution. The equilibrium formed favours the reverse reaction, meaning very little H+ is actually released in solution, making the metal aqua-ions very weak acids.
The greater the charge on the metal ion, the more acidic the metal aqua-ion complex. This is because the O-H bond is polarised more – making the bond weaker and easier to break. As a result, metal aqua-ion complexes with a metal ion that has a 2+ charge are less acidic than metal aqua-ion complexes with a metal ion that has a 3+ charge.
We’ve launched our new site! 🎉
Course-specific notes with built-in search!
AP • A-Level (AQA • CIE • Edexcel • OCR) • IB • NCERT 11 + 12
over 750+ new pages and 3,500 images.
Visit the new homepage
