Video Tutorial Transition Metals and Colour
Quick Notes Transition Metals and Colour
- The d-orbitals in a transition metal ion can be split into high energy and low energy orbitals. This creates an energy gap between the two sets of orbitals.
- Electrons from lower energy d-orbitals can be excited to higher energy d-orbitals by absorbing energy.
- White light contains waves of electromagnetic radiation that have different energies and colours.
- If the energy required to excite an electron can be found in white light, then specific wavelengths of light are absorbed by the transition metal ion and removed from the white light.
- The complimentary colour of the absorbed wavelengths is now observed, and the transition metal ion has a ‘colour’.
Full Notes Transition Metals and Colour
All five d-orbitals have complicated shapes that occupy different areas around a nucleus. When electrons from ligands move towards a metal ion, some of the d-orbitals are closer to the ligand than others, so they experience more repulsion, making them higher in energy compared to the other d-orbitals.
Note, for background on ligands see Complex Ions .
The d-orbitals energies are split: some orbitals are high energy, some are low energy. See d-orbital Splitting.
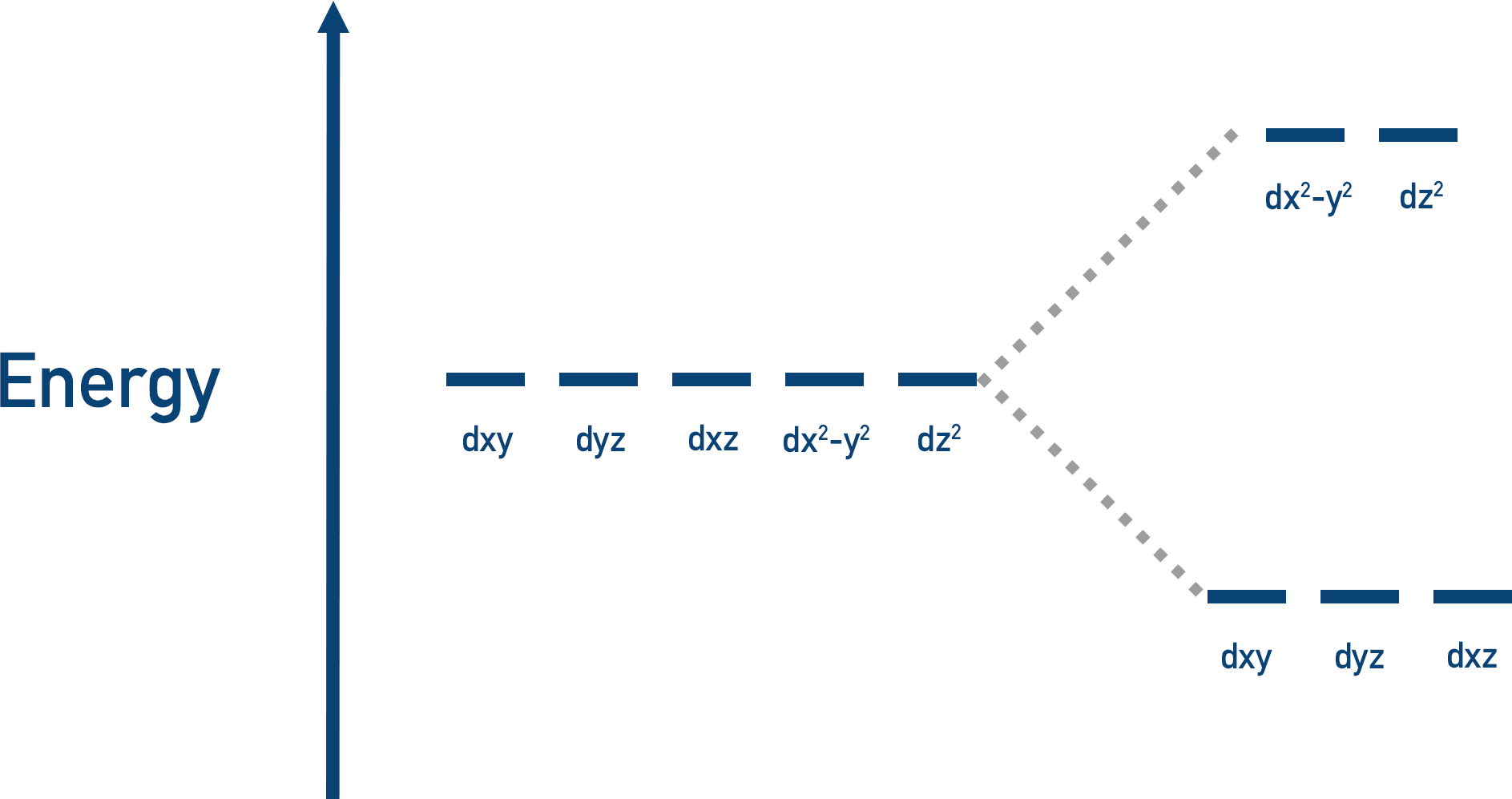
Transition elements form ions that have partially filled d-subshells, which means there will always be a d-orbital with a space for an electron.
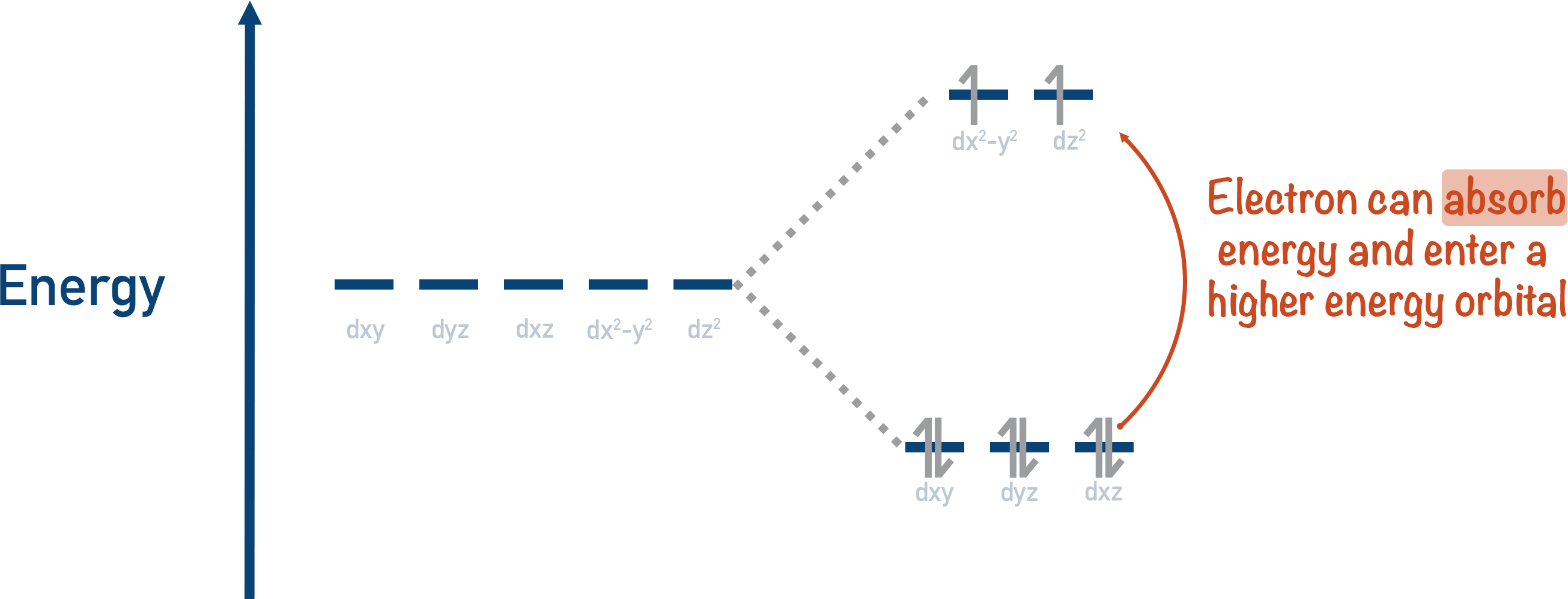
Lower energy orbitals are always filled first – the higher energy levels are only filled once the lower orbitals are full. So, it’s the higher energy orbitals that have a space for an electron.
If just the right amount of energy is given to an electron in a lower energy orbital, it can become excited and go into a partially filled high energy orbital. The amount of energy required for this to occur is very specific, and it’s different for every metal ion. The electron absorbs energy to go to a higher energy orbital.
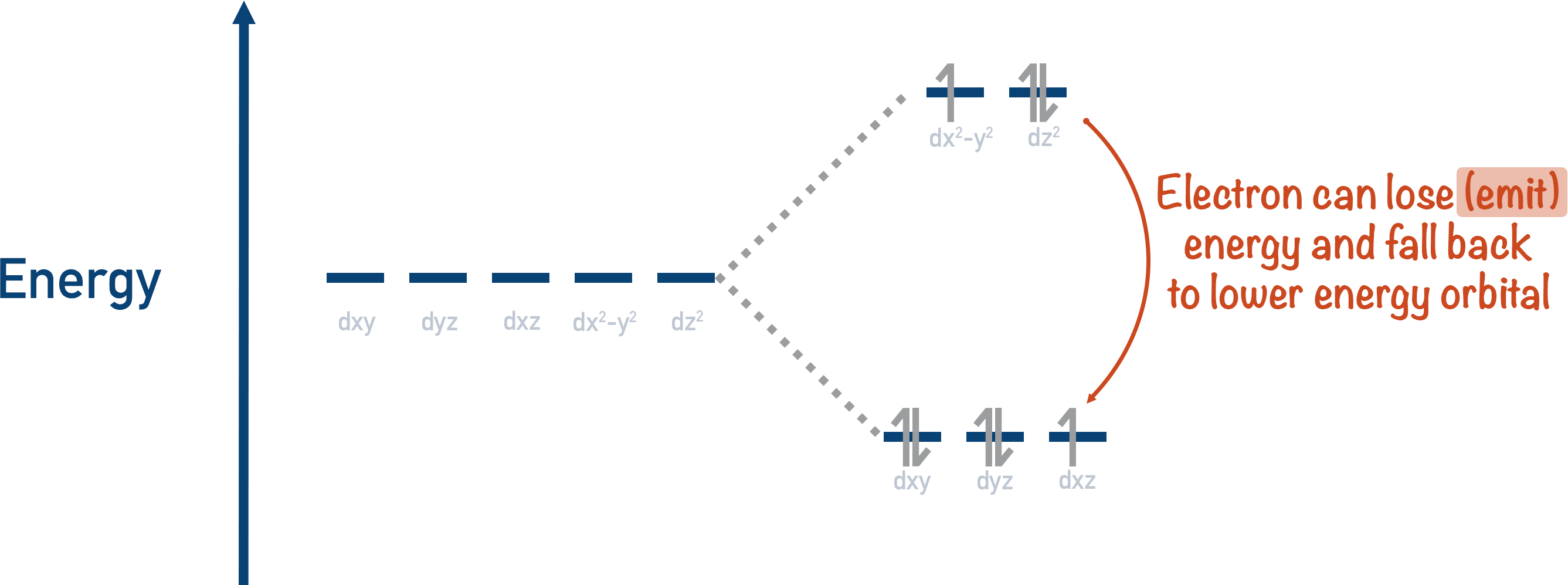
Excited electrons can also lose energy and go back down to a lower energy orbital. The energy lost by the electron is ‘emitted’, usually as light of a particular wavelength. This is how organic molecules can produce light (such as in fireflies, for example). A chemical reaction excites the electrons to a higher energy d-orbital, then as the electrons fall to a lower energy orbital, energy is released as light.
Light is part of the electromagnetic spectrum – a large range of waves that transmit energy from one place to another. The sun emits electromagnetic radiation, some of which is visible to us as light. It’s very important to recognise that visible light is just particular wavelengths of electromagnetic radiation. The range of wavelengths that humans can see is called the visible region of the electromagnetic spectrum.
White light contains all wavelengths in the visible region and has no observable colour. If any wavelength is removed then it’s no longer white light, so a colour is observed. Different colours of light have different wavelengths and are different energies. Violet light is the highest energy; red light is the lowest energy.
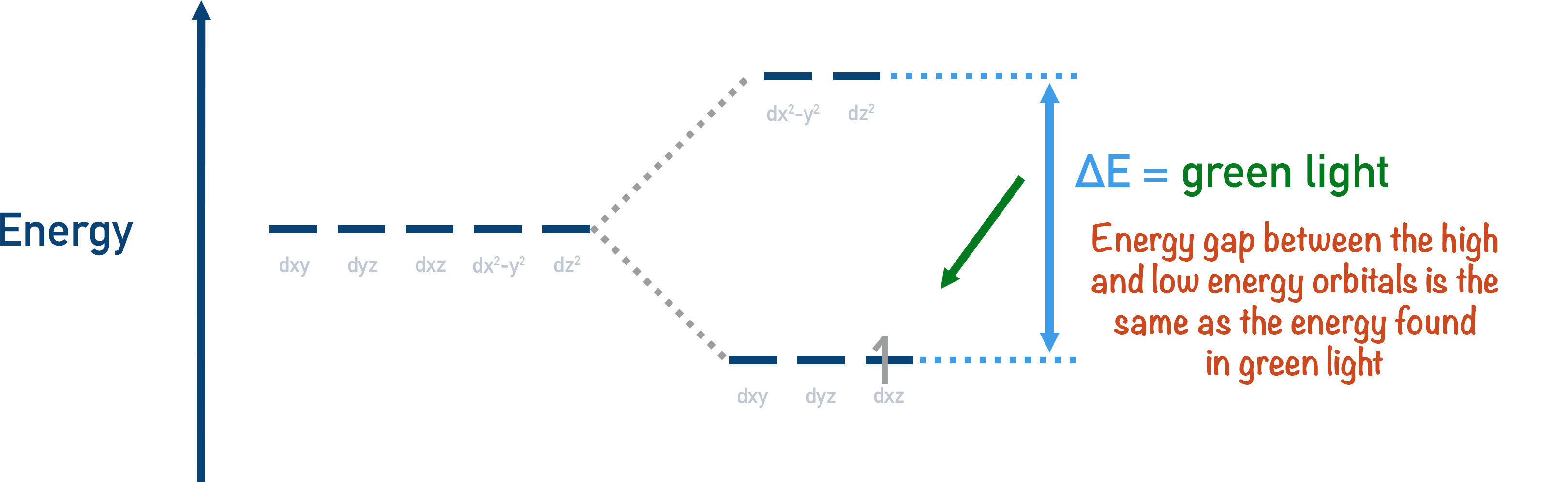
With most transition metal ions, the energy gaps between high and low energy orbitals fall within the visible region of the electromagnetic spectrum. This allows ‘light waves’ to excite electrons from low energy d-orbitals to high energy d-orbitals.
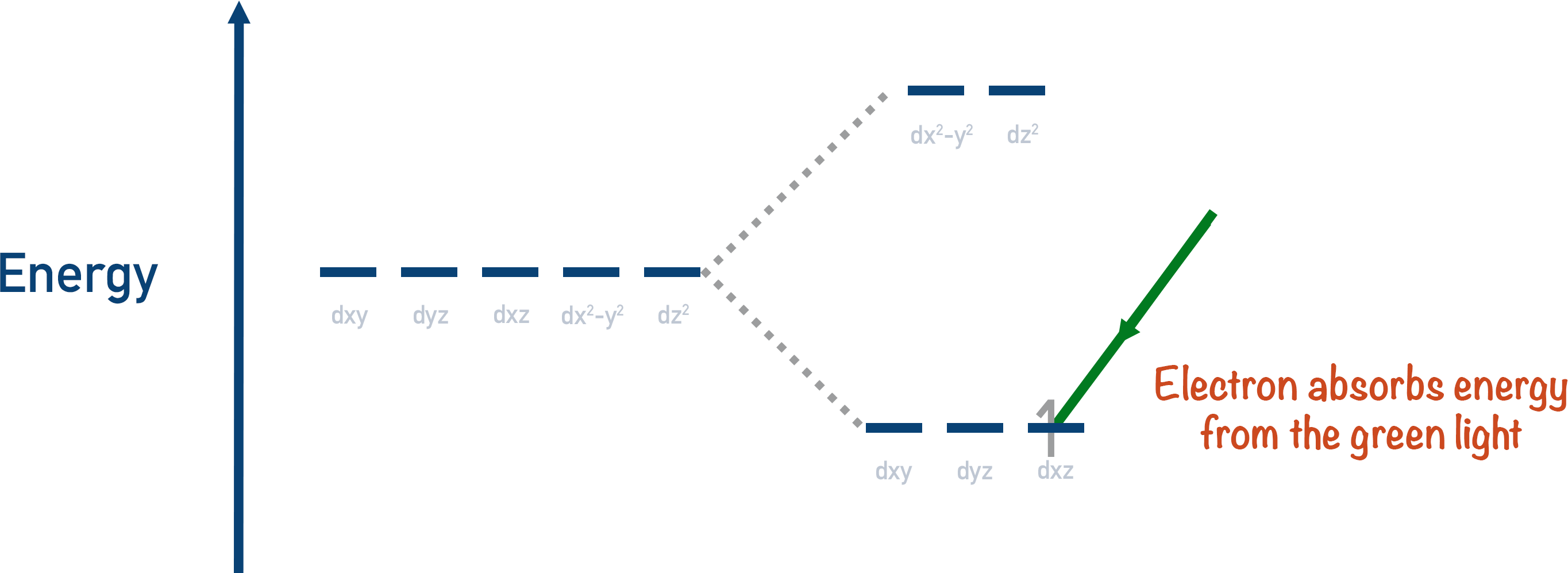
When this happens, the transition metal ion absorbs the wavelength of light that matches the energy gap between its d-orbitals, and this colour of light is removed from the white light.
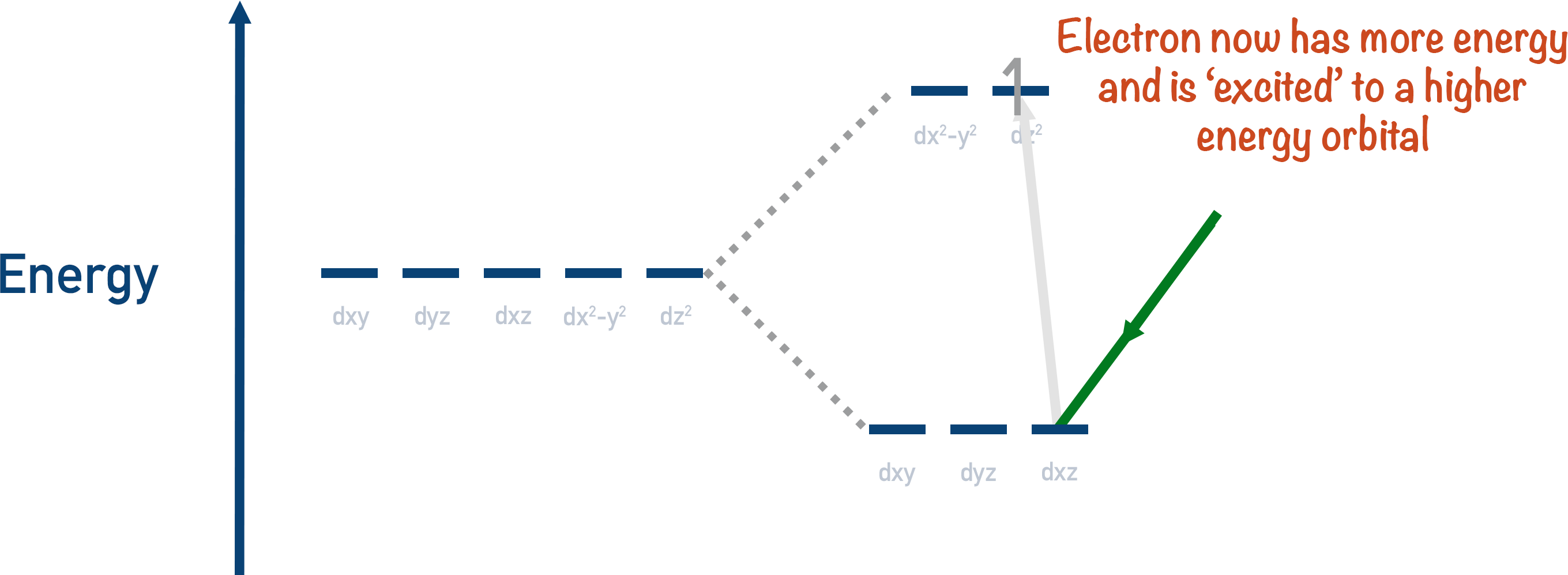
Removing a colour from white light means the light now has an overall colour and can be observed.
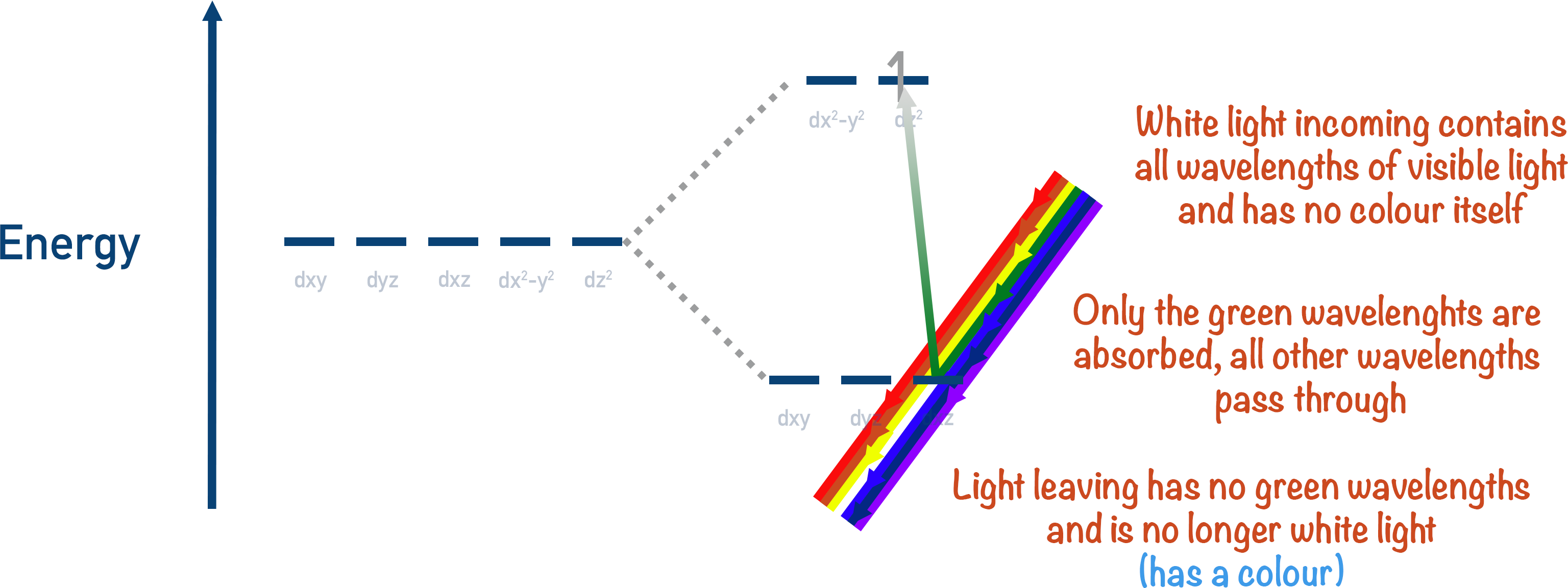
The colour observed when a particular colour is removed from white light is called its complimentary colour. For example, if green light is absorbed by a transition metal ion then all the other colours in white light are reflected (or transmitted), and the colour observed is a combination of these waves. In this case, magenta would be the colour seen.
We’ve launched our new site! 🎉
Course-specific notes with built-in search!
AP • A-Level (AQA • CIE • Edexcel • OCR) • IB • NCERT 11 + 12
over 750+ new pages and 3,500 images.
Visit the new homepage
