Video Tutorial d-orbital splitting
Quick Notes d-orbital Splitting
- There are five d-shaped orbitals that each occupy a different area in space around the nucleus of an atom.
- Some d-orbitals are repelled more by approaching ligands than others, making them higher energy (and the other d-orbitals lower energy).
- The d-orbitals are split into two groups: high energy and low energy, with a specific energy gap between them.
Full Notes d-orbital Splitting
All five d-orbitals have complicated shapes that occupy different areas around a nucleus.
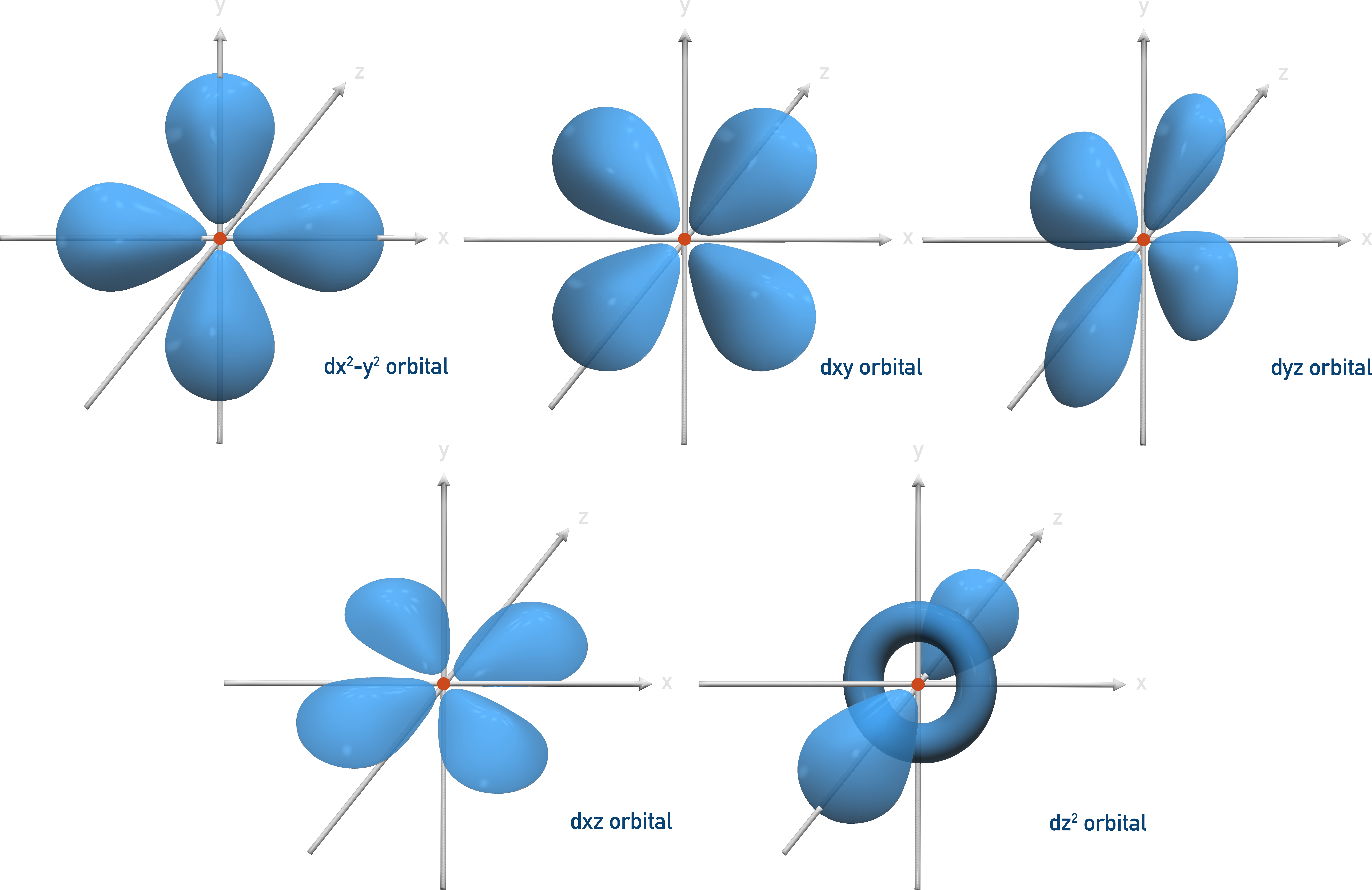
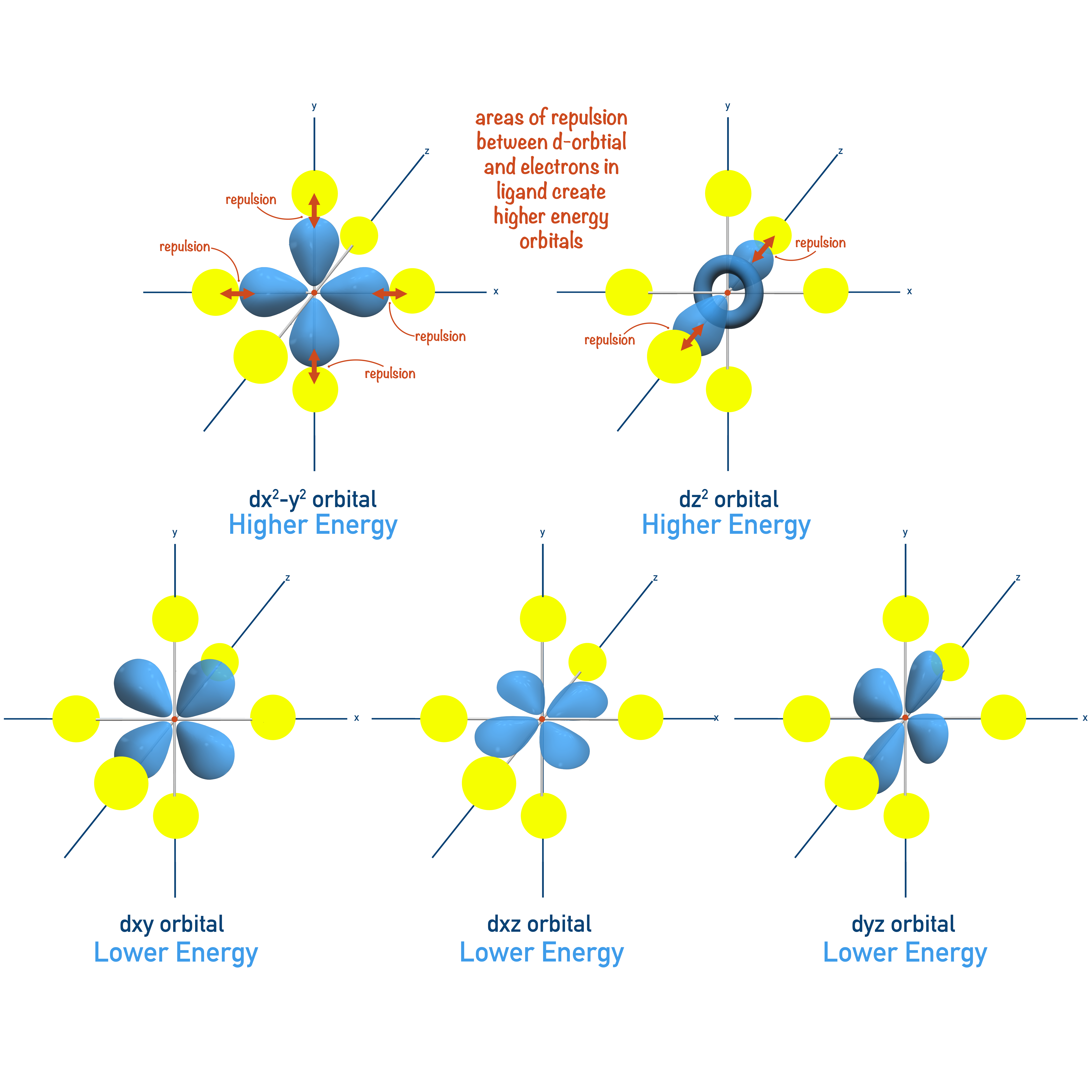
When ligands move towards a metal ion, some of the metal ions’ d-orbitals are closer to the ligand than others. Ligands have a lone pair of electrons, which repel the electrons in d-orbitals that are closest to them. Electrons in d-orbitals further away from the ligand are repelled less. Repulsion leads to high energy. This makes the d-orbitals repelled by the ligands a higher energy, and the other d-orbitals a lower energy.
Note, in more advanced study of chemistry ‘low energy’ is referred to as ground state and ‘high energy’ as excited state, but these terms aren’t commonly used in A-level studies.
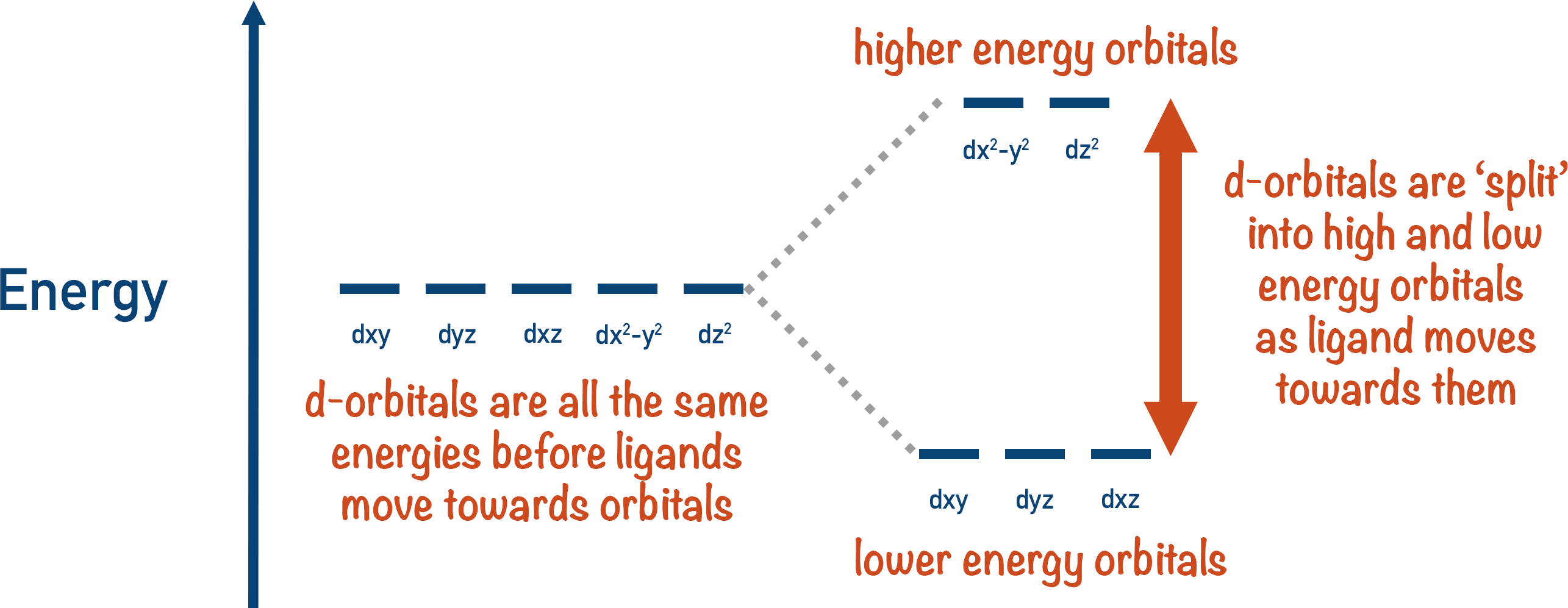
The d-orbitals energies are split – some are high energy, some are low energy.
The difference in energies between the orbitals creates two sets of orbitals: high energy and low energy. The energy gap between the high and low orbitals is unique for every metal ion, although it can be changed if the ligands around the metal ion are changed, as different ligands will repel the orbitals differently.
We’ve launched our new site! 🎉
Course-specific notes with built-in search!
AP • A-Level (AQA • CIE • Edexcel • OCR) • IB • NCERT 11 + 12
over 750+ new pages and 3,500 images.
Visit the new homepage
