Video Tutorial Heterogeneous Catalysis
Quick Notes Heterogeneous Catalysis
- In heterogeneous catalysis, the catalyst is in a different phase to the reactants.
- With solid-based catalysts and gaseous or aqueous reactants, the catalysis process occurs in stages:
- reactants diffuse onto catalyst surface
- reactants are adsorbed onto the catalyst
- the reaction starts and an intermediate is formed
- the reaction completes and products are made
- products desorb (leave) the surface of the catalyst
Full Notes Heterogeneous Catalysis
There are two types of catalysis: heterogeneous and homogeneous.
Heterogeneous catalysis is where the catalyst is in a different phase to the reactants. For example, in a car exhaust the catalysts in a catalytic converter are solid metals, but the reactants are in gaseous states.

In the above reaction, in the presence of a catalyst two reactants (A and B) react to produce C.
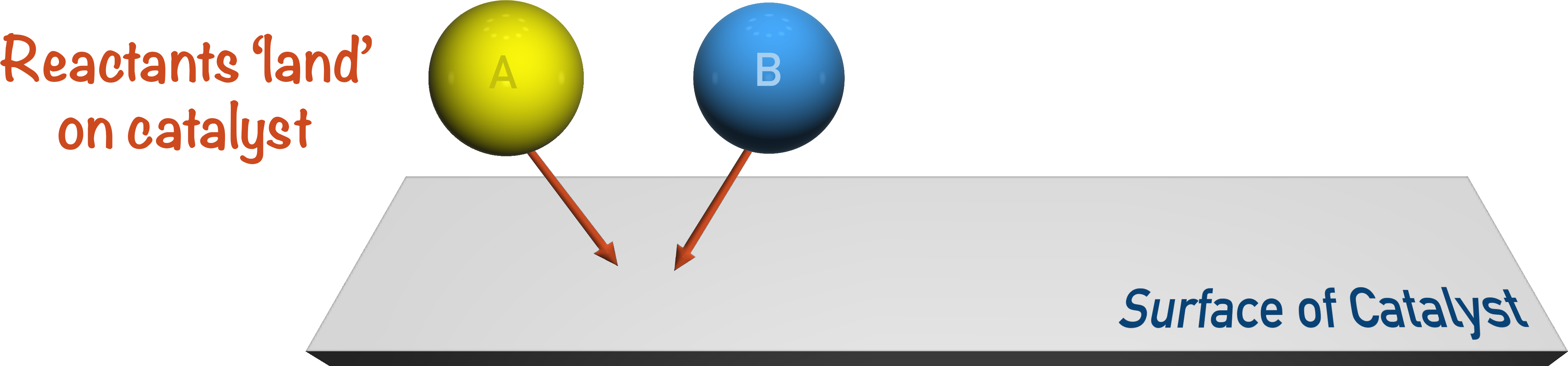
For solid-based catalysts and gaseous reactants: the reactants diffuse (through other gases) to the catalyst and ‘land’ on the surface.
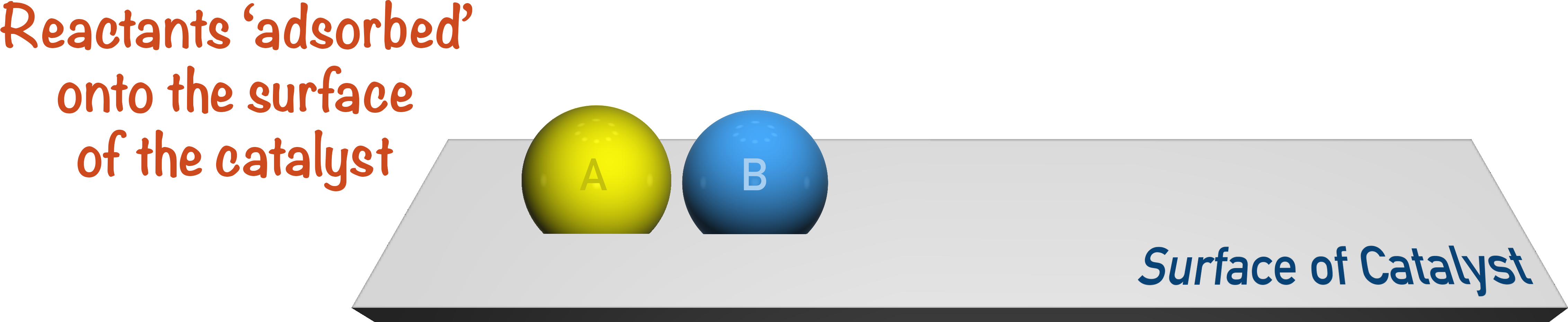
The reactants then get adsorbed onto the surface of the catalyst.
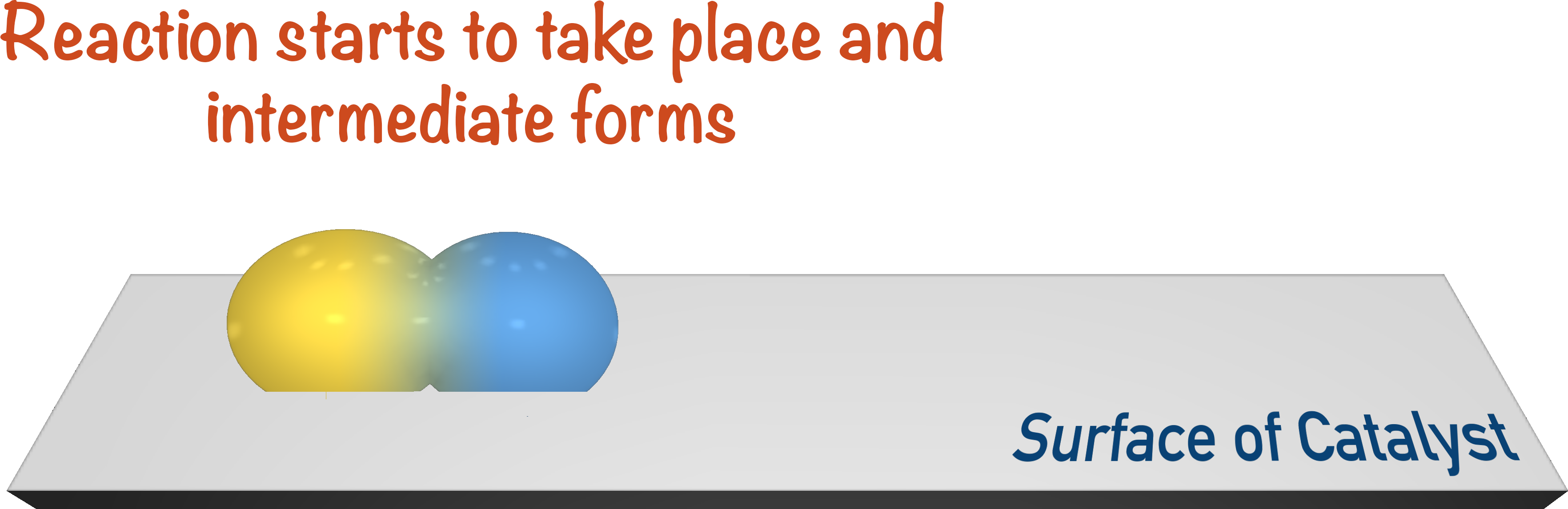
After adsorption has taken place, bonds within the reactants are weakened to the point where the reaction can start to take place, and an intermediate is formed.

After the reaction has completed, the product remains adsorbed on the surface of the catalyst.

The product will desorb from the surface of the catalyst and will diffuse away.
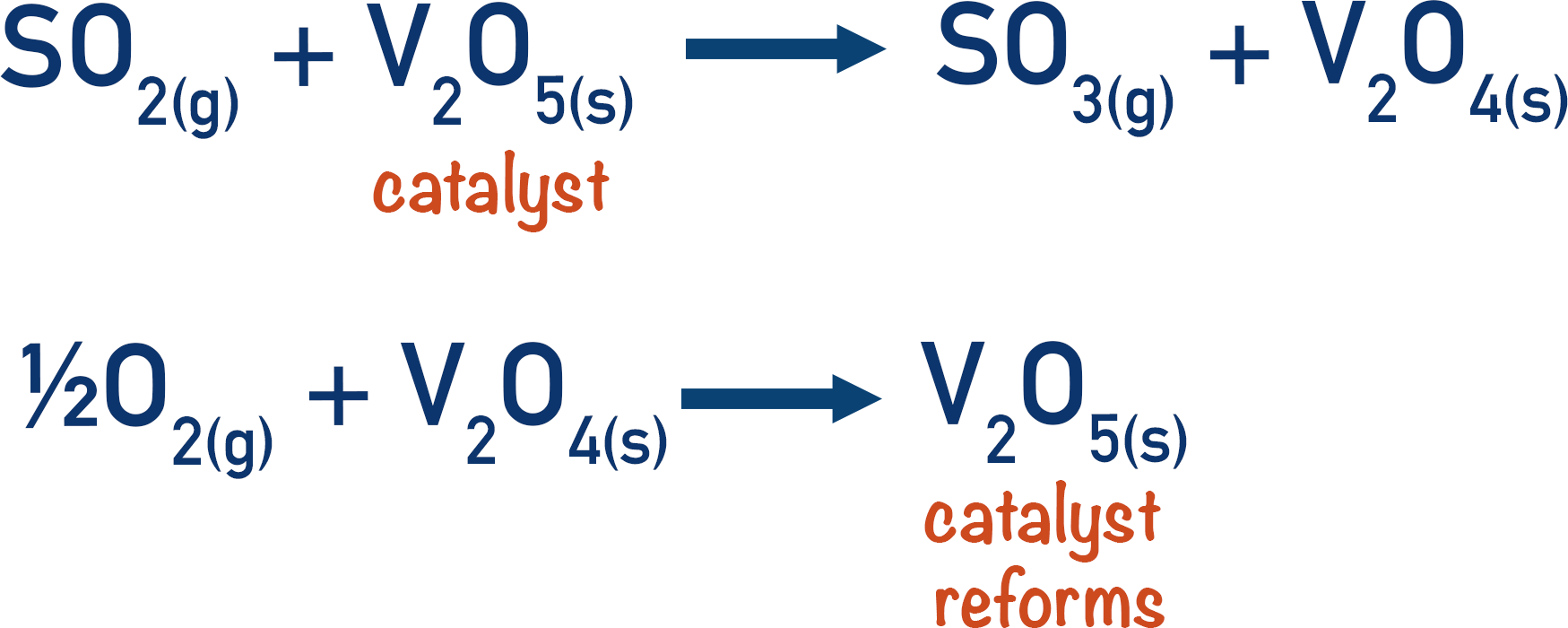
The Contact Process
The contact process is a frequently used example of heterogeneous catalysis, where vanadium (V) oxide is used as a catalyst in the production of sulfur trioxide from sulfur dioxide. (Sulfur trioxide is a reactant used to produce sulfuric acid.)
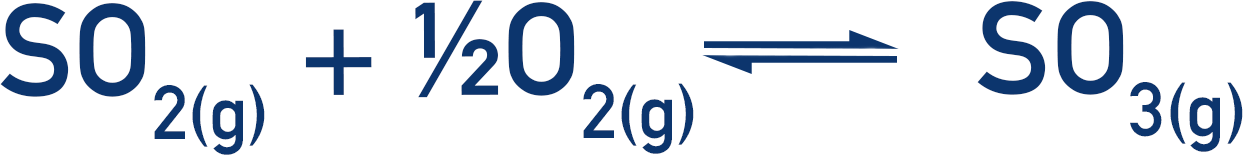
The vanadium (V) catalyst is used to produce the sulfur trioxide, then it’s reformed when reacted with oxygen.
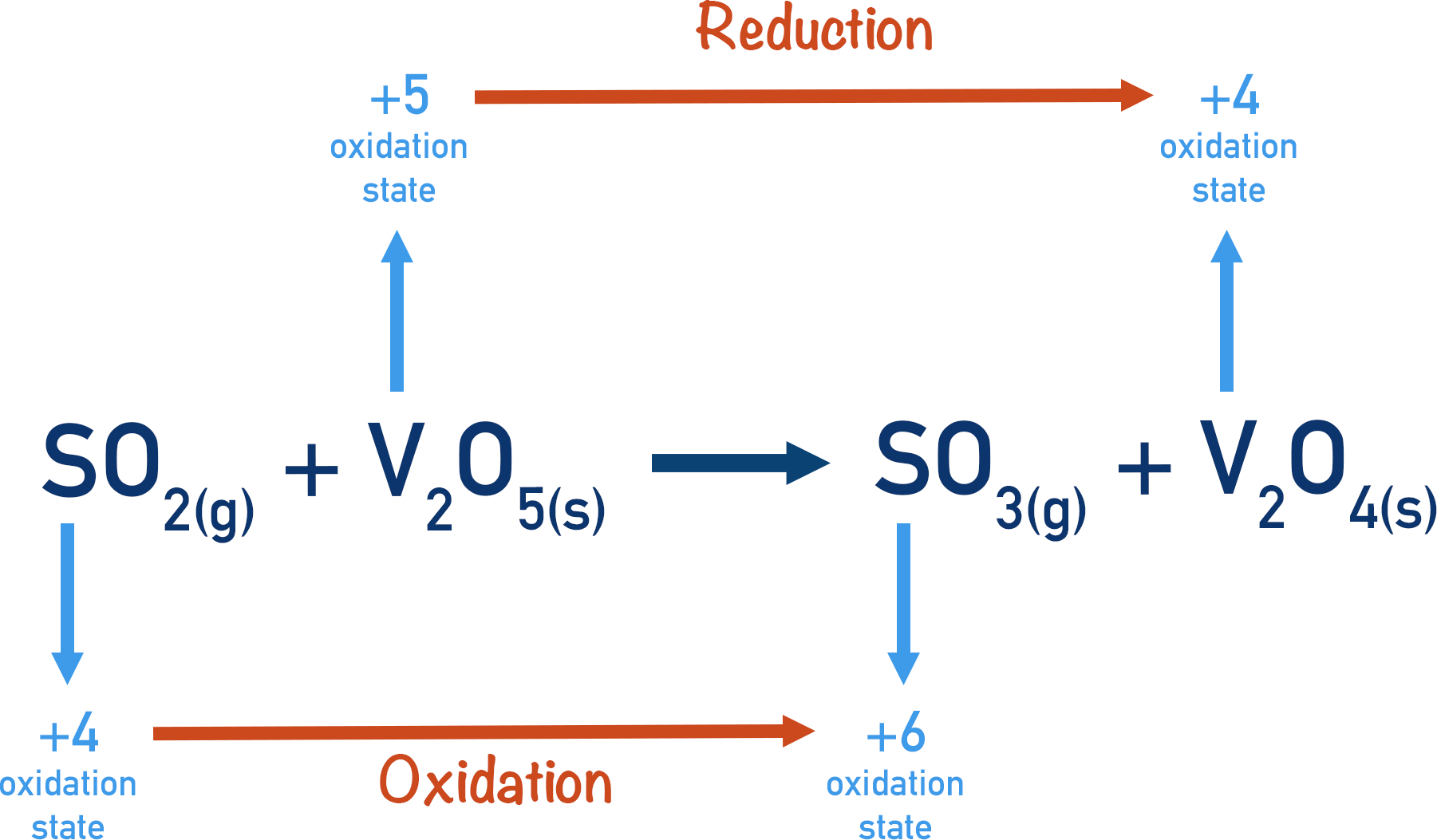
The vanadium (V) oxide oxidises the sulfur dioxide, going from vanadium (V) oxide to vanadium (IV) oxide.
The vanadium (IV) oxide then gets oxidised itself by the oxygen from the sulfur trioxide. The final product is vanadium (V) oxide – the same as at the start of the reaction.
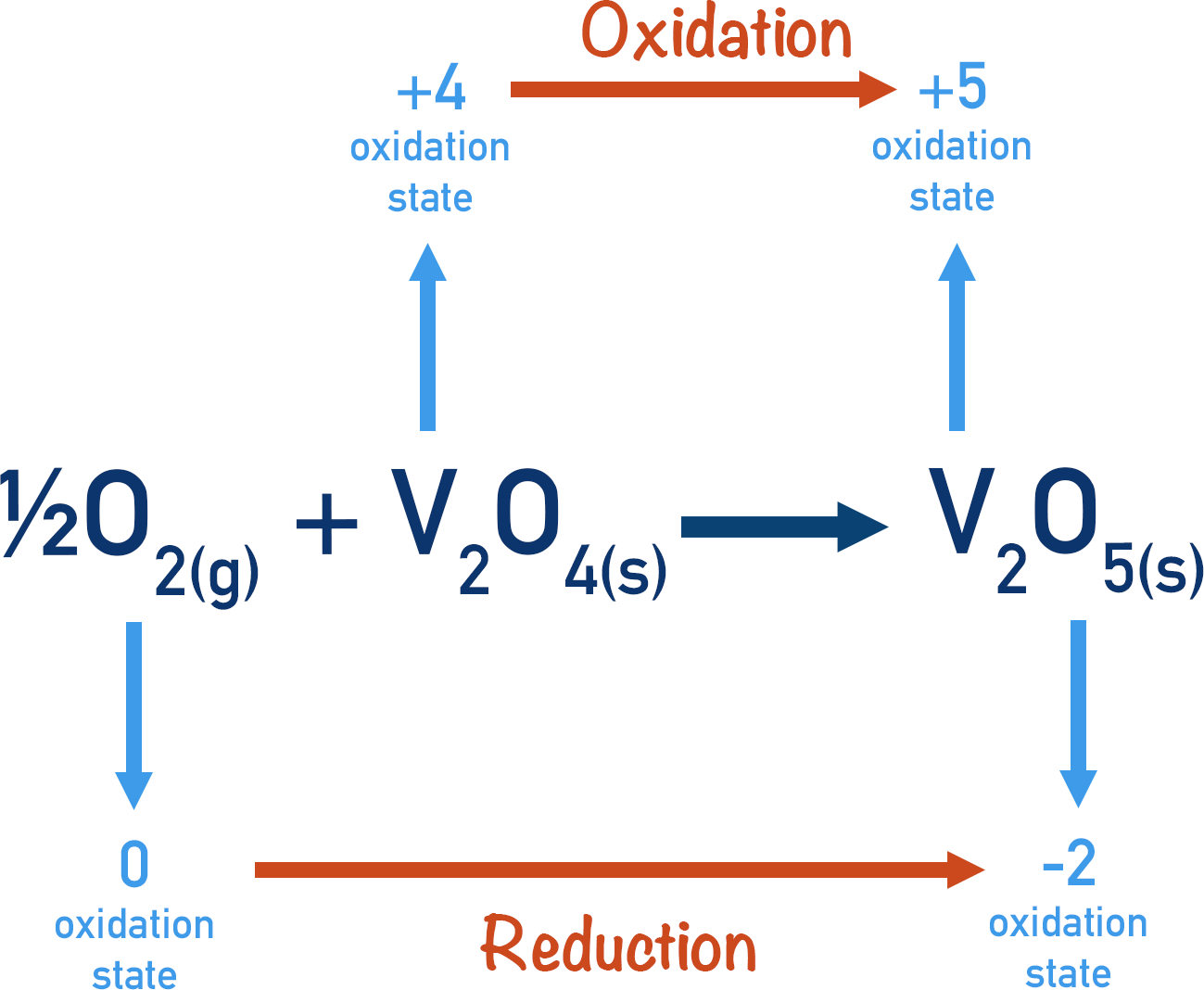
The key to the whole process is the fact that the vanadium in the catalyst can be reduced (to form the sulfur trioxide), then oxidised (to reform the catalyst). The variable oxidation state of vanadium allows this to happen.
We’ve launched our new site! 🎉
Course-specific notes with built-in search!
AP • A-Level (AQA • CIE • Edexcel • OCR) • IB • NCERT 11 + 12
over 750+ new pages and 3,500 images.
Visit the new homepage
