Video Tutorial Catalysts
Quick Notes Transition Metals as Catalysts
- Transition metals often make good catalysts.
- Transition metals have variable oxidation states, enabling them to form ions with different charges.
- These ions can lose or gain electrons to form another stable ion, which can then gain or lose electrons to reform the original ion.
- In a reaction, this means a transition metal can provide an alternative route, allowing a species to be oxidised and another species to be reduced.
Full Notes Transition Metals as Catalysts
Transition metals have variable oxidation states, allowing them to form ions with different charges. The ability to lose or gain an electron to change oxidation state and then re-form the original oxidation state, allows transition metal ions to ‘help’ in a reaction but remain unchanged overall, making them very good catalysts.
Note, catalysts lower the activation energy of a reaction, by providing an alternative pathway for the reaction to occur (see Rates of Reaction, Catalysts ).
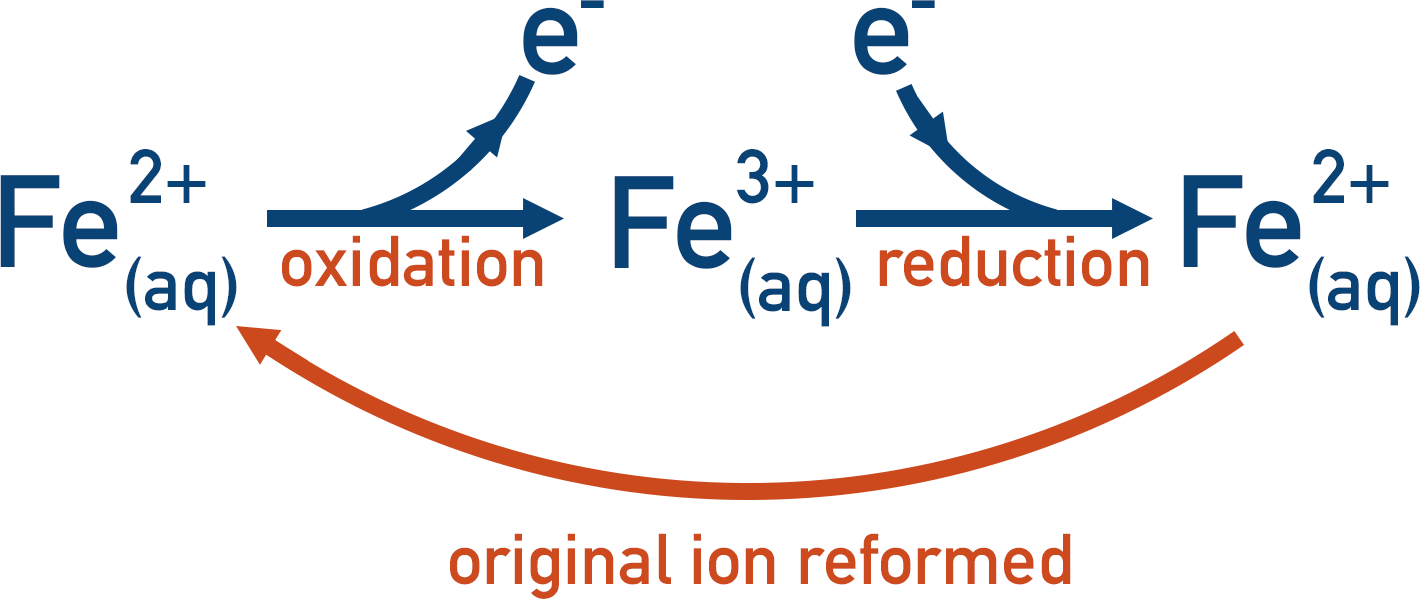
The partially filled d-orbitals of transition metal ions enable this to happen. It’s a little bit like a bus driving around with at least one back seat free: it can easily ‘pick up’ another passenger and ‘drop off’ that passenger later on, or it can drop off a passenger and pick up another one later on.
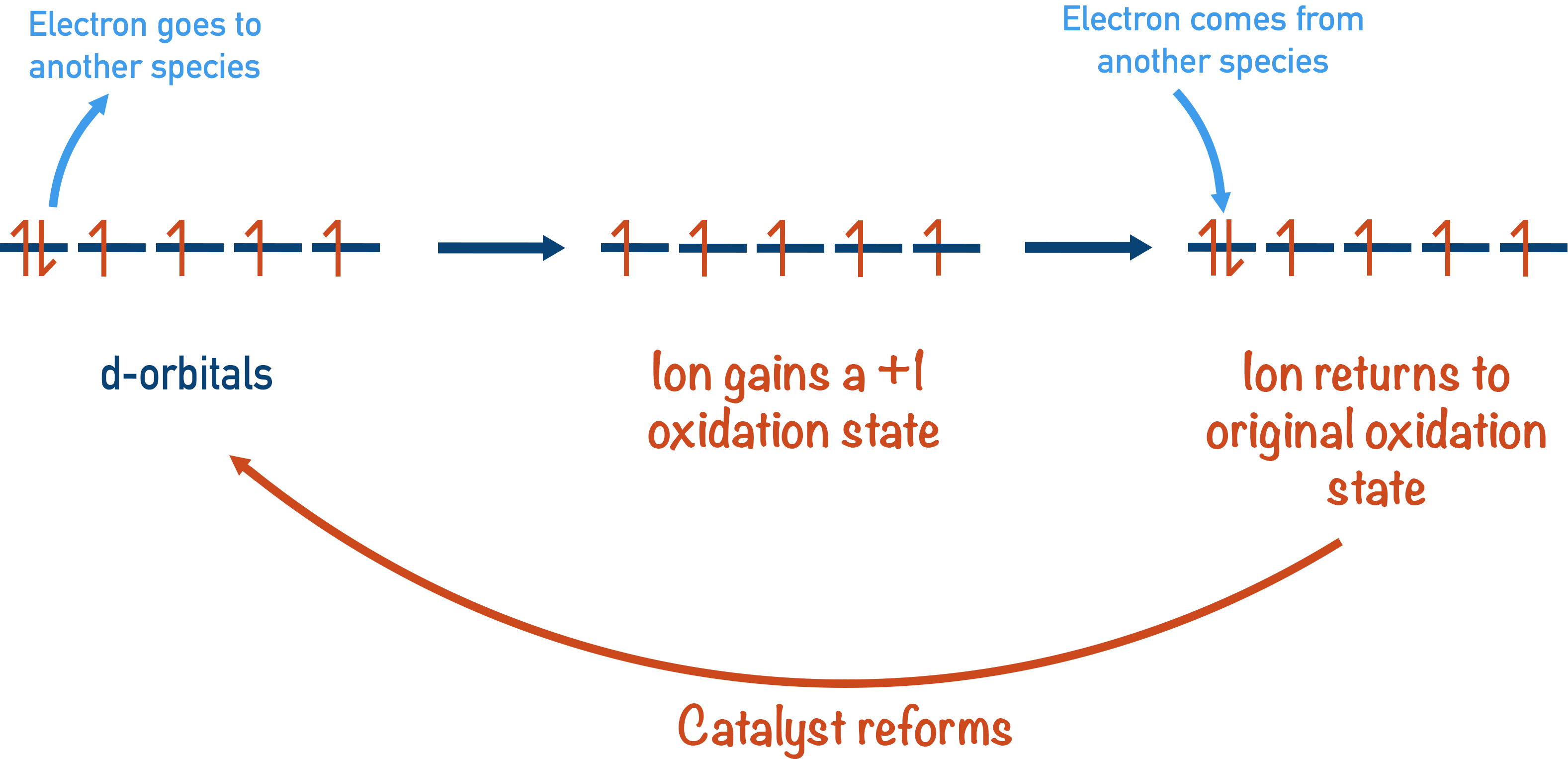
The same is true for the transition metal ions: an electron can ‘hop into’ an empty space in an orbital, enabling another species to be oxidised, and later on in the reaction it can ‘hop out’ of the orbital, allowing another species to be reduced.
Equally, an electron already in a d-orbital can ‘hop out’ and reduce another species, then later in the reaction another electron can ‘hop back in’ to oxidise another species. In both cases, although the transition metals oxidation state changes to help the reaction happen, the oxidation state of the metal is the same at the end as at the start, meaning they have acted as a catalyst.
We’ve launched our new site! 🎉
Course-specific notes with built-in search!
AP • A-Level (AQA • CIE • Edexcel • OCR) • IB • NCERT 11 + 12
over 750+ new pages and 3,500 images.
Visit the new homepage
