Full Notes Functional Groups
Alkanes are, generally speaking, not very reactive. Reactions that arise in organic chemistry almost always occur because of chemical ‘groups’ that are bonded onto alkane chains. These ‘groups’ are called functional groups; they can react with other organic molecules and reagents to produce new products. The functional group of a molecule dictates its chemistry, and its presence enables us to predict how the molecule is likely to react.
When molecules have the same functional group, but differ by the length of their carbon chains, we say they belong to the same homologous series. Molecules within the same homologous series will react in a similar way to each other because they share the same functional group.
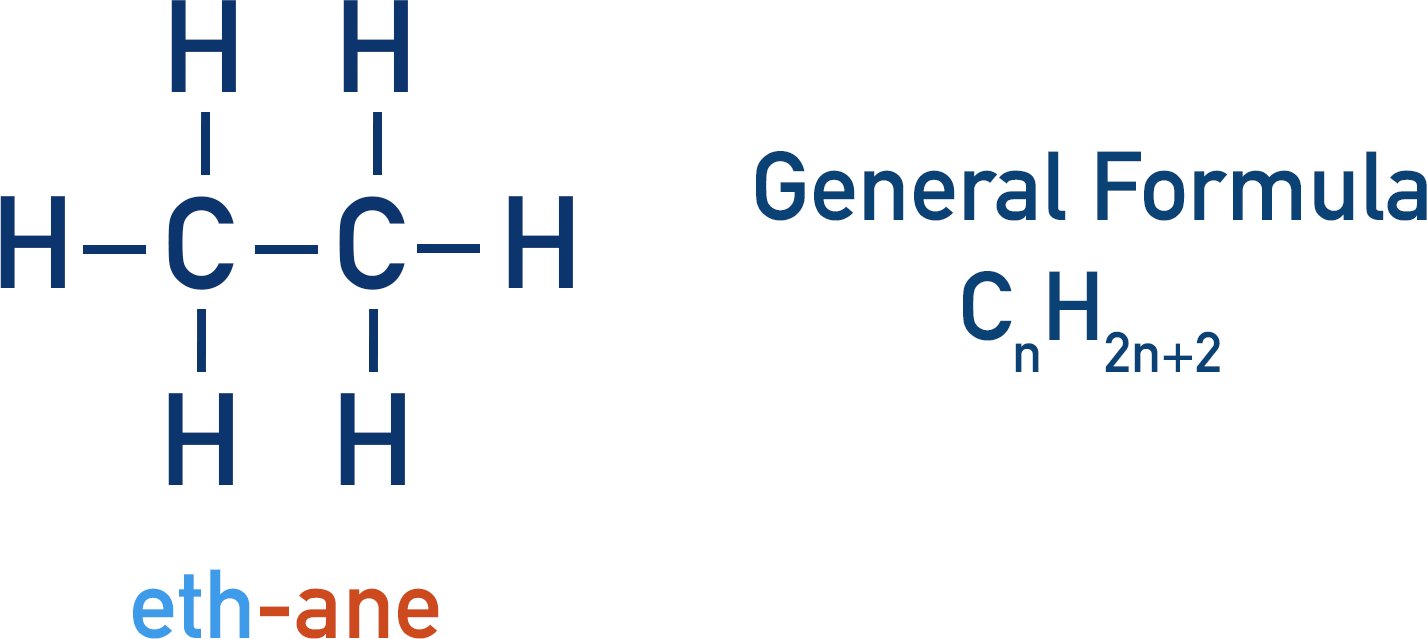
General formulas show how many atoms of an element are present in a molecule for a particular homologous series, based on the number of carbon atoms it has.
There are many different types of functional groups within organic chemistry. For AS-level (first year), you must be familiar with and recognise the following:
Alkane
Alkene

Halogenoalkane

Alcohol

Aldehyde

Ketone

Carboxylic Acid

