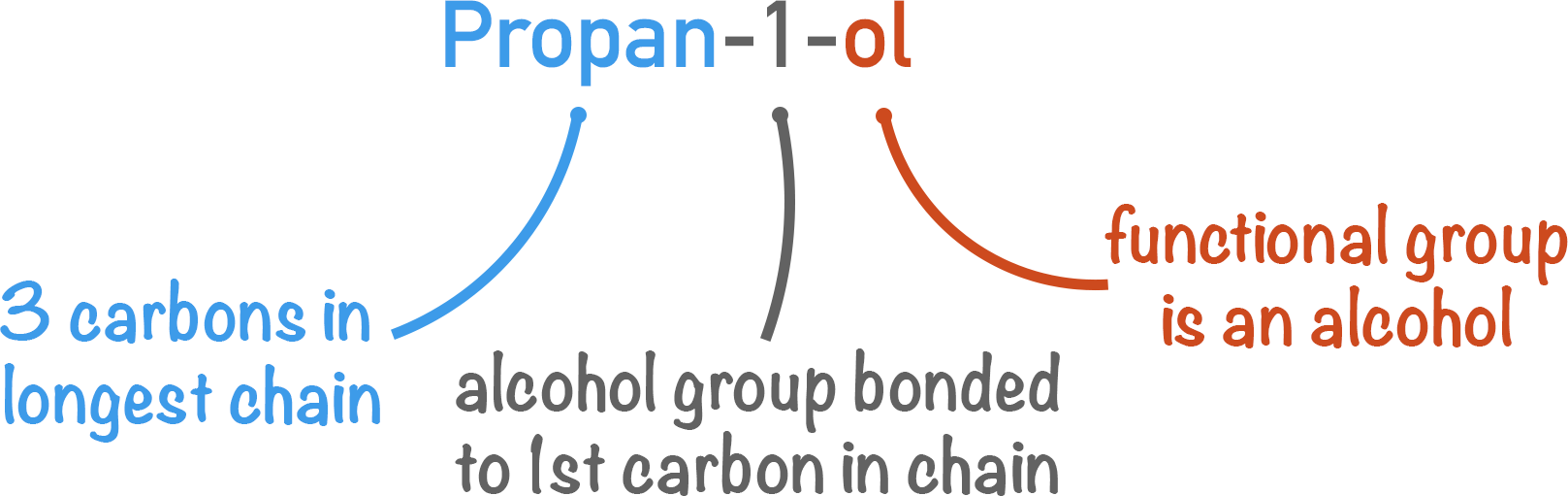
Quick Notes Nomenclature
- Nomenclature is the process of naming compounds in organic chemistry.
- To name a compound, four basic rules are followed:
- Step 1 Identify the of longest carbon chain and choose the prefix (meth, eth...).
- Step 2 Identify the functional groups and choose the suffix (-ol, al…).
- Step 3 Identify the position of functional groups on the carbon chain (-1-ol, -3-ene.).
- Step 4 Place functional groups and alkyl chains in alphabetical order – if more than one.
Full Notes Nomenclature
Organic molecules can be incredibly complex, so to ensure anyone can deduce the structure of a compound from its name a set of naming rules must be followed. An internationally accepted method for naming compounds is used – called the International Union of Pure and Applied Chemistry (IUPAC) system. The process of naming organic compounds in chemistry is called nomenclature.
For A-level purposes, below are four steps to check, which should help when naming a compound.
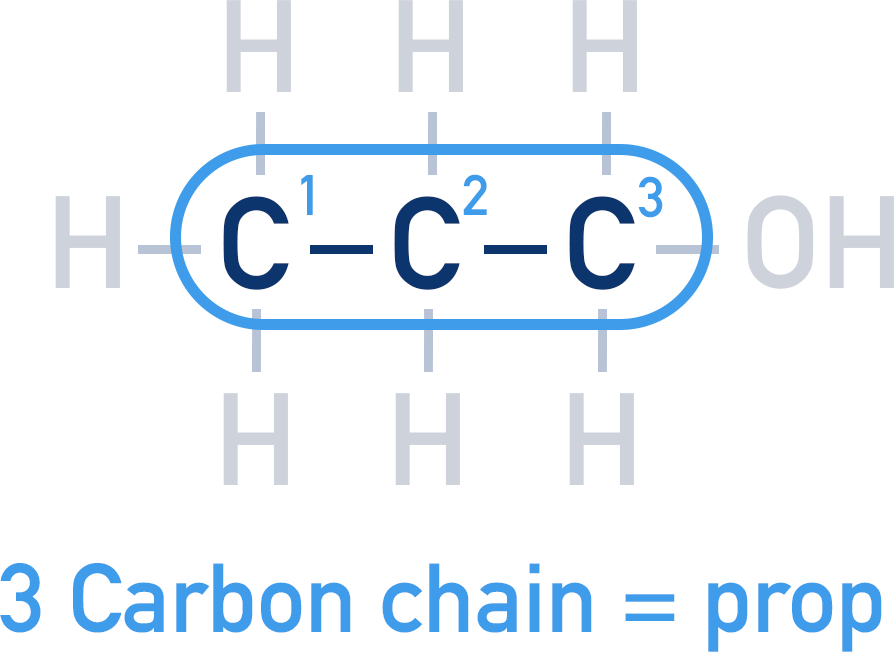
1. When naming a compound, the length of the longest carbon chain must be determined. Use the prefix as for an alkane (meth, eth, prop…etc.).
2. Alkyl and functional groups must be identified. Use the appropriate suffix (or prefix for halogenoalkanes) (-ol, -al, -oic acid, etc.).
3. If a positional isomer is possible, the carbons in the chain must be numbered. The chain is always numbered, so a functional group or alkyl group is bonded to the lowest carbon number possible.
4. When molecules contain two or more functional or alkyl groups, they are placed in alphabetical order.
For example, to name the following:
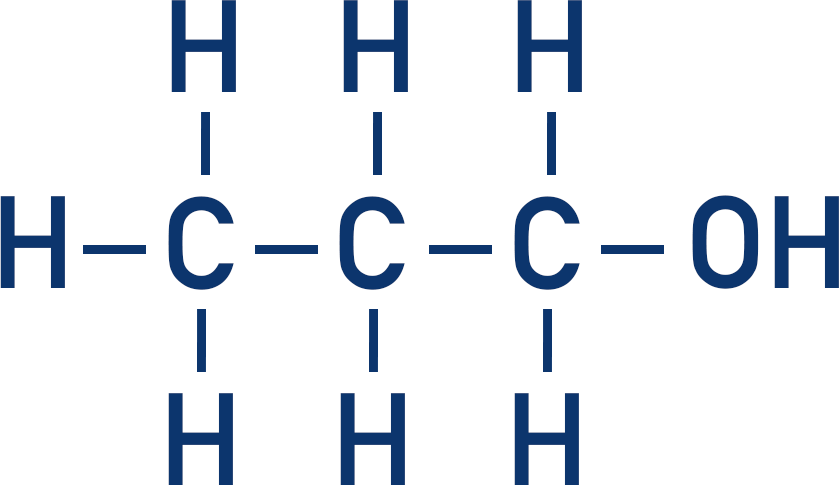
Determine the length of the longest carbon chain the molecule.
Identify any function groups.
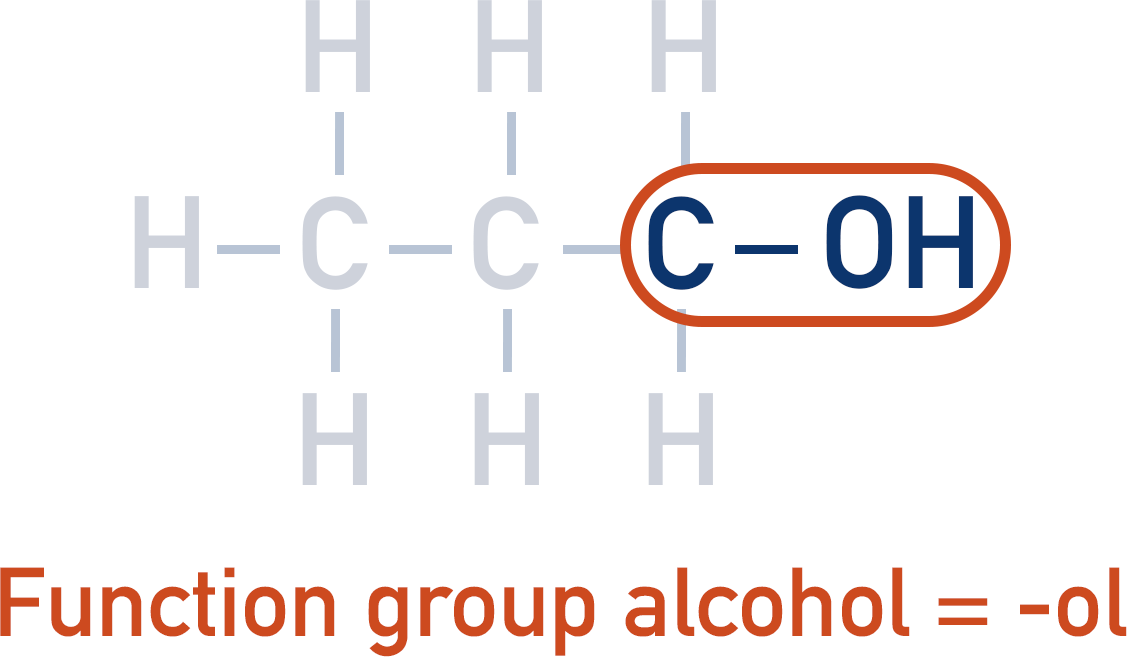
Could the functional group be placed anywhere else in the chain? If so, number the carbon atom the functional group is bonded to.