Quick Notes Condensation Polymerisation
- Condensation polymers are formed when monomers bond together and one water molecule is released per bond.
- To break apart a condensation polymer (into its monomers), a water molecule is needed and a hydrolysis (breaking of bond with water) reaction happens.
- Polyamides are formed from the reaction of a di-carboxylic acid with a di-amine. The links between each monomer are called peptide links.
- Polyesters are formed from the reaction of a di-carboxylic acid with a di-ol in the presence of an acid.
Full Notes Condensation Polymerisation
Polymers are large molecules that are made up of repeating units. In a polymer, repeating units are joined together many times to produce long chains. The repeating units themselves are made from simple molecules called monomers.
Condensation Polymerisation
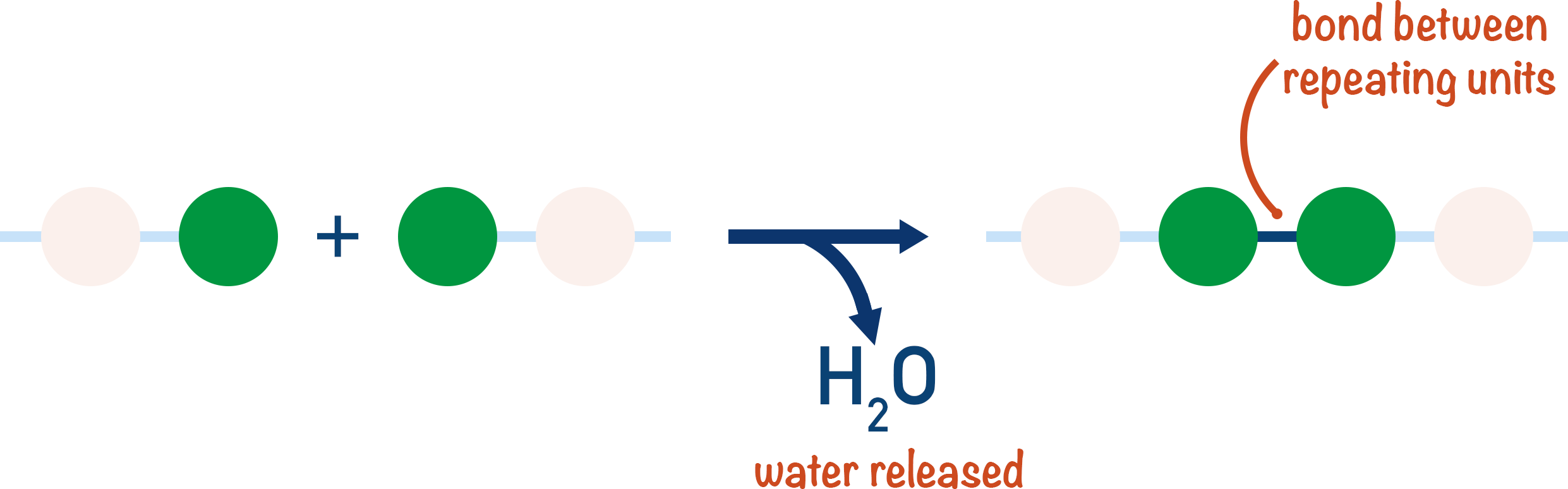
Condensation polymerisation involves the release of (usually) a water molecule when two monomers react together to form a new bond. There are many examples of condensation polymerisation and it is very common in nature.
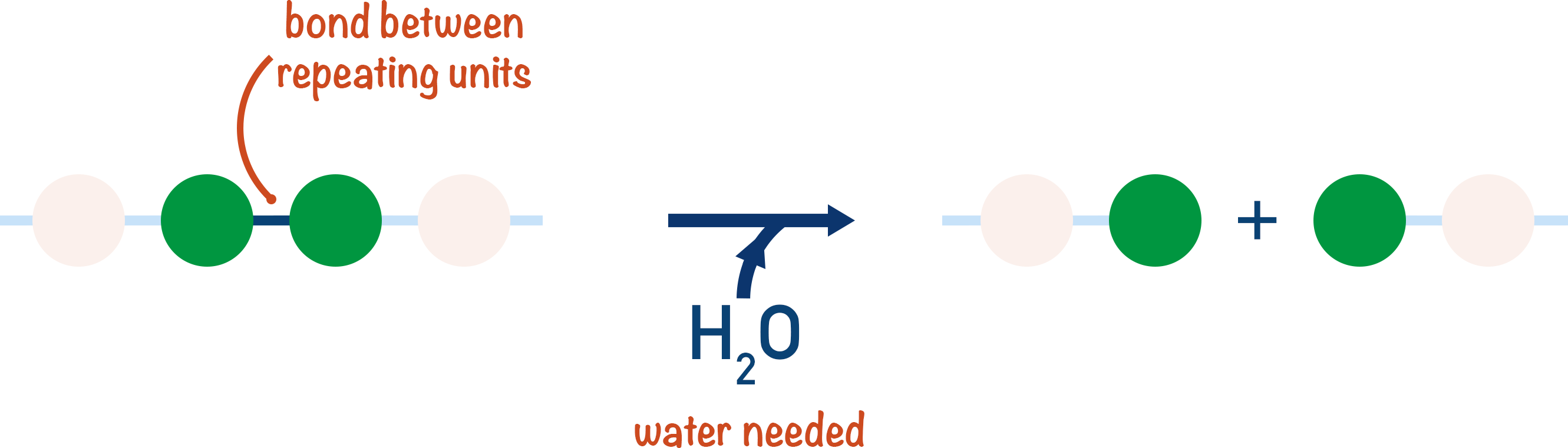
The addition of water can cause the bonds between repeating units to break and the polymer is broken down. This is the hydrolysis of a polymer.
At first glance, condensation polymerisation reactions can look very confusing, but the types of polymer formed are named after the functional groups present in the polymer.
Polyamides
Polyamides contain an amide group that links repeating units together.
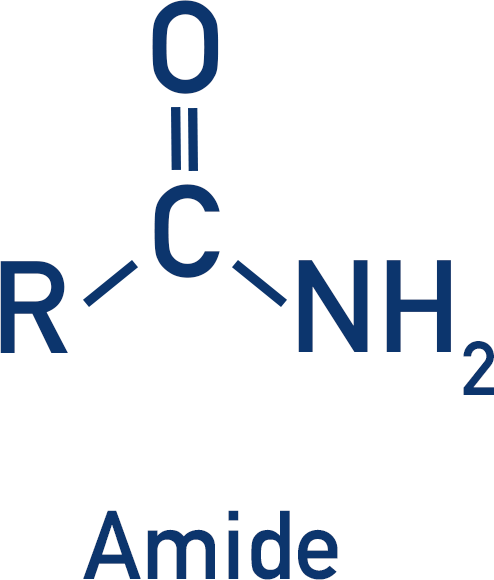
To produce an amide, a carboxylic acid and amine are reacted together (in organic reactions, acyl chlorides are usually used).

This forms an amide link, but not a polymer as the product is not able to form more amide links.
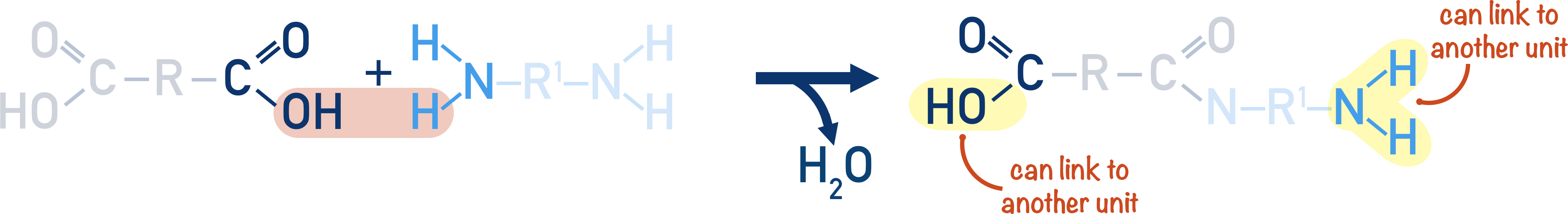
By reacting a dicarboxylic acid with a diamine, the repeating unit formed has both a carboxylic acid group and amine group.
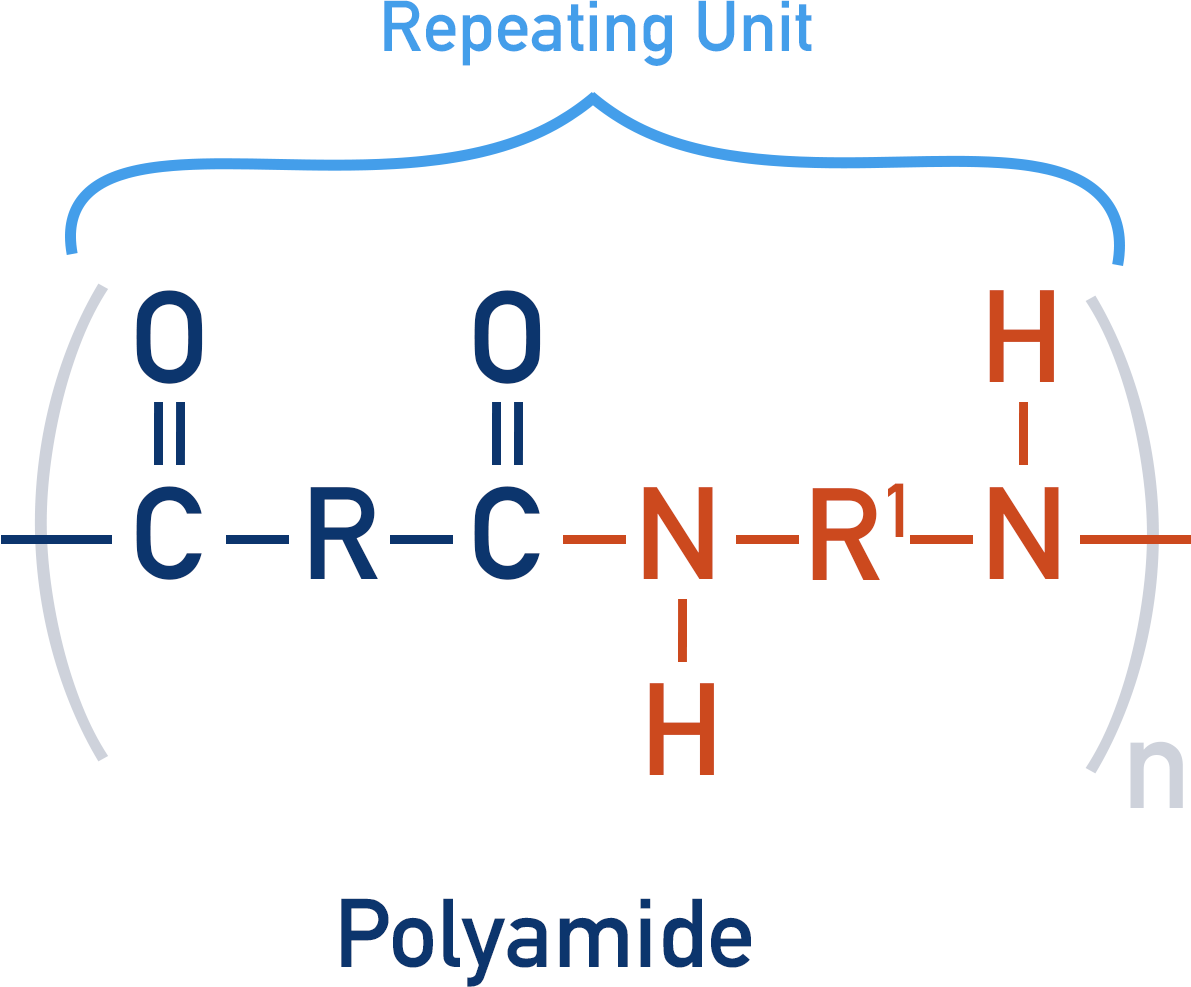
The repeating units can go on to react with another repeating unit, producing another amide link, creating a polyamide.
Polyesters
Polyesters contain an ester group that links repeating units together.
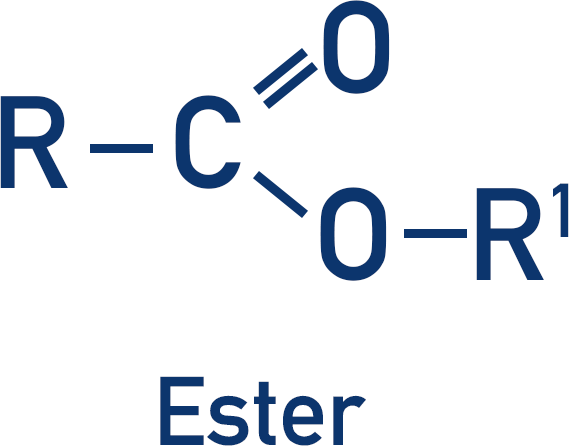
To produce an ester, a carboxylic acid and alcohol are reacted together (in the presence of an acid).
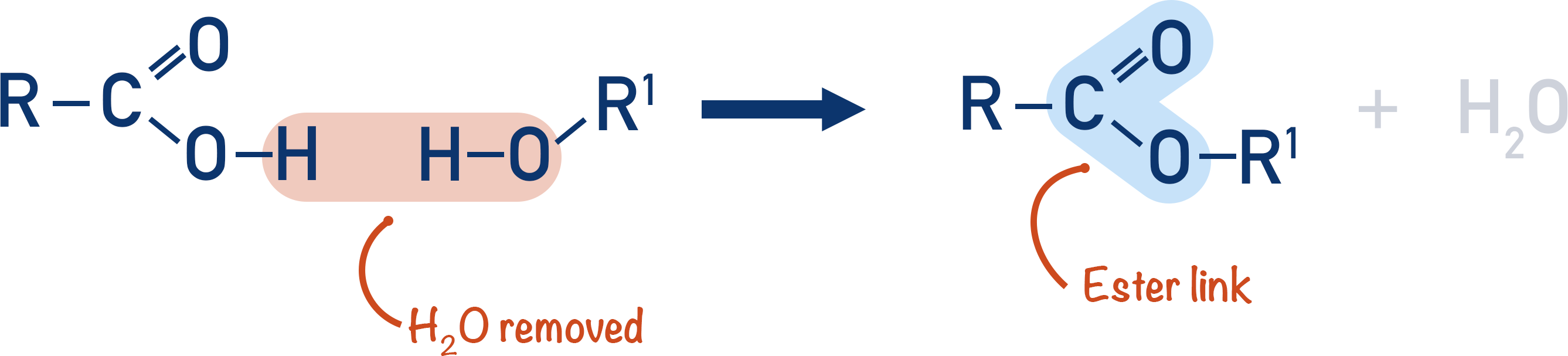
This forms an ester link, but not a polymer as the product is not able to form more ester links.
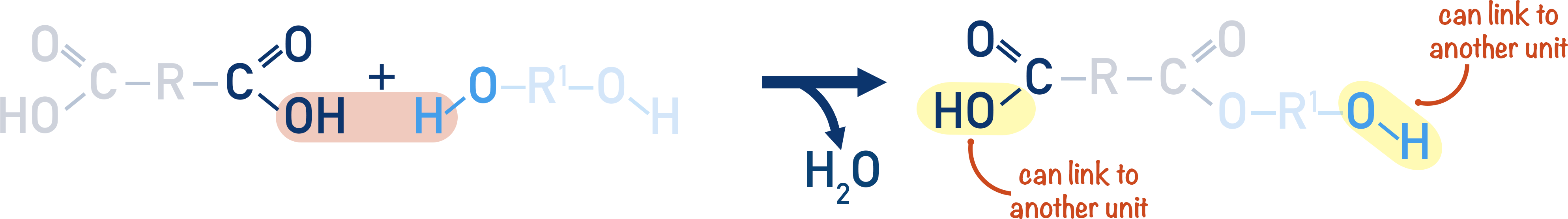
By reacting a dicarboxylic acid with a diol, the repeating unit formed has both a carboxylic acid group and alcohol group.
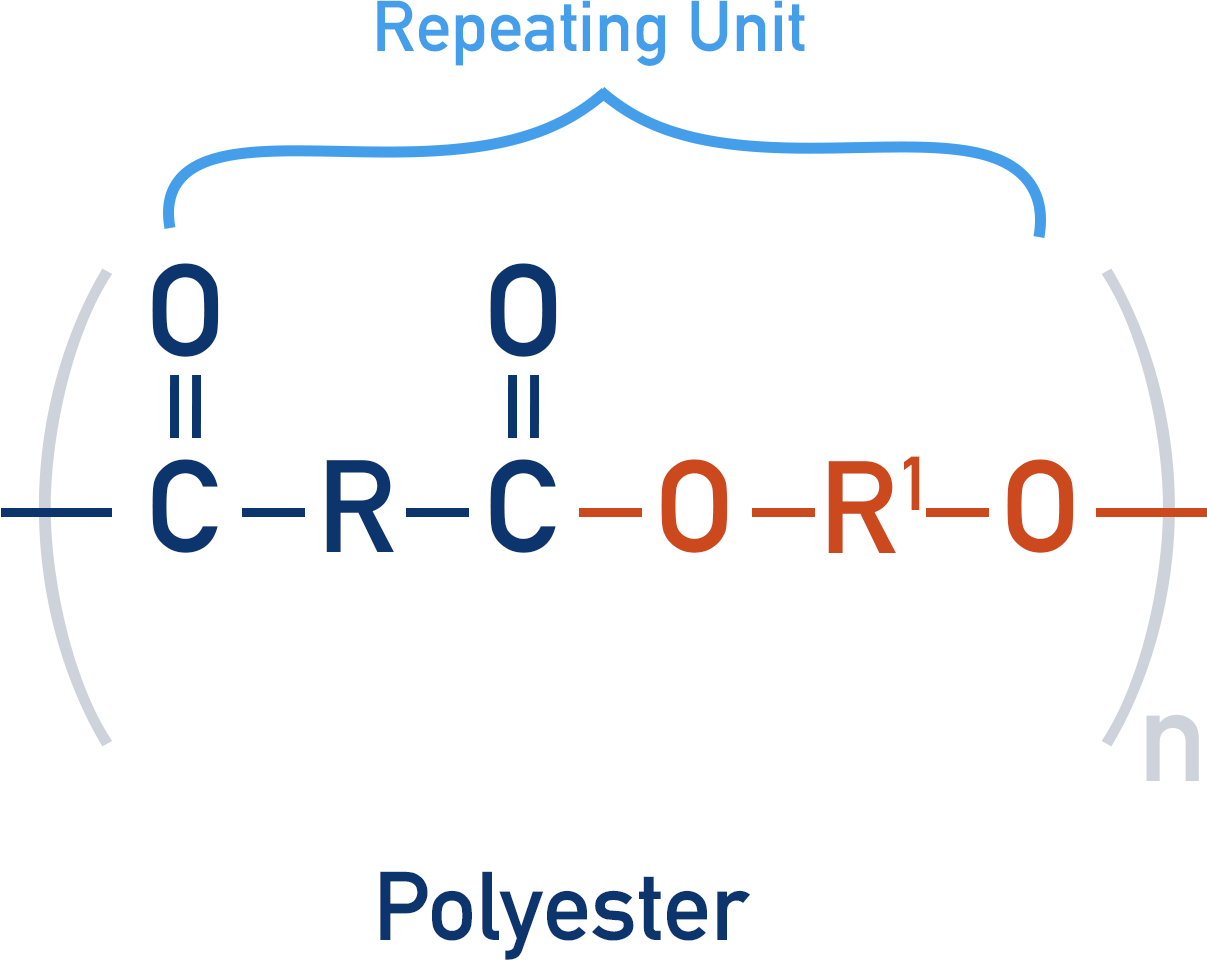
The repeating unit can go on to react with another repeating unit, producing another ester link, creating a polyester.
