Quick Notes The Halogens and Boiling Points
- The melting and boiling points of the halogens increase going down group 7.
- Halogens exist in elemental form as simple, diatomic molecules that are held together by weak intermolecular forces (temporary induced dipole-dipole).
- The larger the molecules, the larger the intermolecular forces that can arise between them, requiring more energy to overcome – giving the substance a higher melting point.
- The sizes of the halogen molecules increase down the group, meaning their melting and boiling points also increase.
Full Notes The Halogens and Boiling Points
The melting and boiling points of the halogens increase as you go down group 7.
To melt a substance, the forces holding the particles of the substance together have to be broken. The stronger these forces are, the greater the thermal energy required to break them. This means substances held together by strong forces have higher melting and boiling points.
The opposite is true for substances with weak forces between particles – little energy is required to overcome the forces, so a low temperature is needed, giving the substance a low melting point.
The halogens all exist di-atomically (two atoms covalently bonded together) as simple molecules. They have only very weak temporary induced dipole-dipole intermolecular forces between molecules.
Temporary induced dipole-dipole interactions arise when electrons are unequally distributed in a molecule momentarily. The more electrons present in the molecule, the greater and stronger the dipole-dipole interactions. Stronger interactions require more energy to overcome, so the molecules have higher melting points (see Intermolecular Forces).
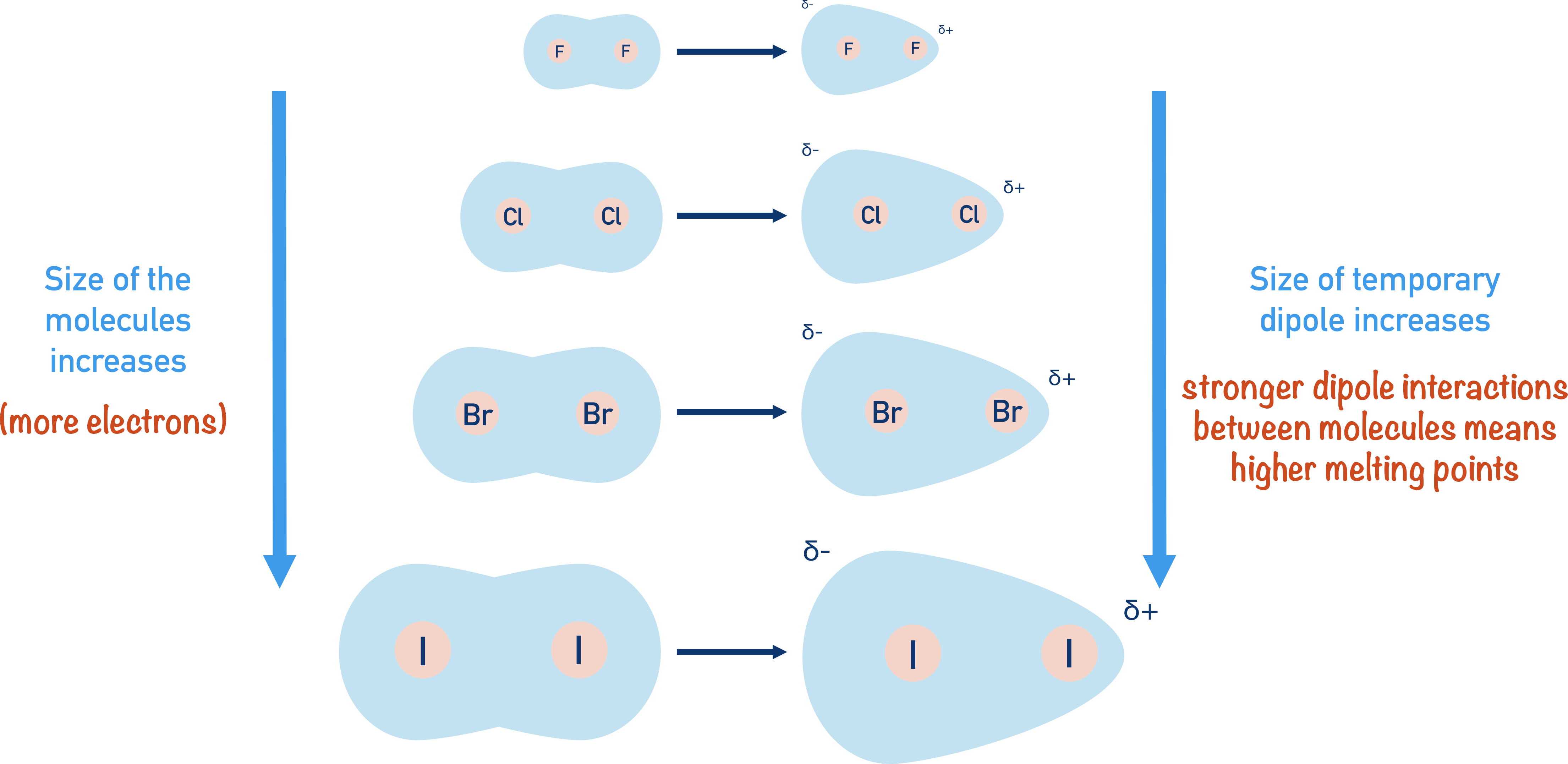
The size and the number of electrons in the halogen molecules increase as you go down the group, resulting in higher melting and boiling points.
