Quick Notes Oxidising Power of the Halogens
- Halogen atoms can gain one electron to have a full outer shell – they are reduced.
- For a halogen atom to gain an electron, another species must lose an electron (be oxidised).
- Halogens can act as oxidising agents as they can force other species to be oxidised.
- Oxidising agents are themselves reduced.
- The ability of halogens to act as oxidising agents decreases down the group.
- Fluorine is the strongest oxidising agent of the halogens.
- Halogens can oxidise less reactive halide ions in displacement reactions.
Full Notes Oxidising Power of the Halogens
The halogens have seven electrons in their outer shell, so they easily gain one electron to have a full outer shell (eight electrons).
X + e- → X-
By gaining an electron they are reduced, but they need to get this electron from somewhere! In order for the halogen to gain an electron, another species must lose an electron. The species the halogen gets the electron from is oxidised. Because of this, we describe the halogens as oxidising agents, as they cause another species to be oxidised.
Oxidising agents are reduced!
As you go down group 7, the strength of the halogens as oxidising agents decreases. The number of electron shells increases, meaning that the outermost electrons have more inner shells between them and the nucleus.
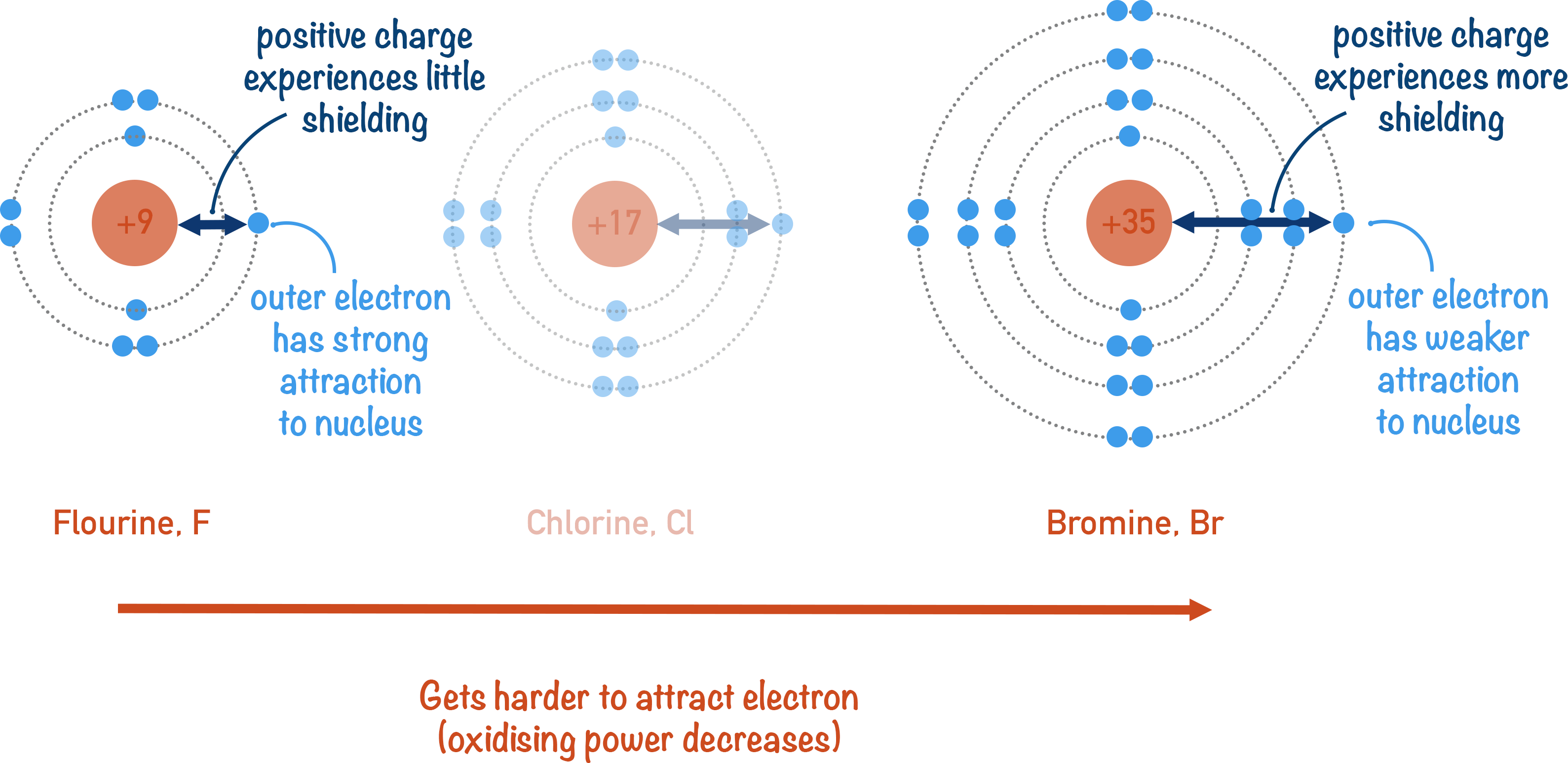
These extra inner shells effectively shield the positive charge from the nucleus that reaches the outermost electrons (inner electron shielding). This results in the positive charge being weaker at the surface of the atom. A weaker positive charge at the surface means the atom is less able to attract an electron from another species. Larger halogen atoms cannot act as an oxidising agent as well as smaller halogen atoms.
Fluorine is the strongest oxidising agent as it is the smallest halogen atom.
Displacement Reactions
As some of the halogens are stronger oxidising agents than others, they can actually oxidise the halide ions of halogens less reactive than themselves.
In solution (aq), chlorine is able to oxidise both bromide ions and iodide ions as it ‘wants’ an extra electron more than the bromine and iodine. The chlorine atoms basically steal electrons from the ions.
Cl2(aq) + 2Br-(aq) → 2Cl-(aq) + Br2(aq)
Cl2(aq) + 2I-(aq) → 2Cl-(aq) + I2(s)
Bromine is able to oxidise iodide ions, but not chloride or fluoride ions.
Br2(aq) + 2I-(aq) → 2Br-(aq) + I2(s)
The products made in such reactions are very easy to identify, as the halogen molecules have different colours and have different states at room temperatue (note bromine is slightly soluble in water, giving Br2(aq)).
Cl2 = pale green gas
Br2 = orange liquid
I2 = brown / purple solid
