Quick Notes Atomic Radius
- Atomic radius decreases across a period in the periodic table.
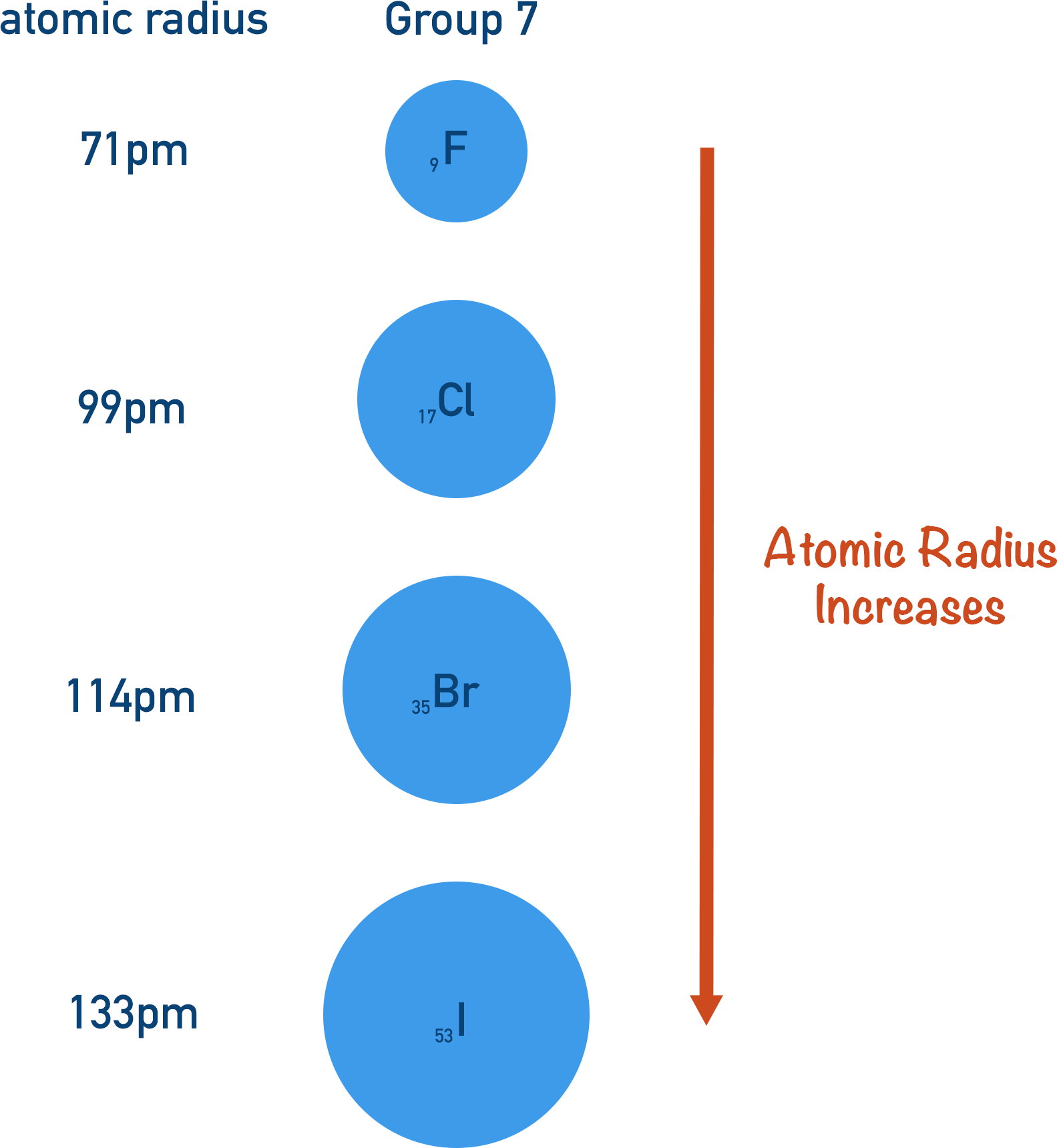
- Atomic radius increases down a group in the periodic table.
- Electrons in inner shells shield the positive charge of the nucleus from electrons in outer shells; this is called inner electron shielding.
- The greater the degree of electron shielding, the less tightly outer electrons are attracted to and ‘pulled towards’ the nucleus, so the larger the atomic radius.
- Across a period, the number of protons in the nucleus of an element increases, but the level of shielding stays the same. This means outer electrons get pulled into the nucleus more and the atomic radius decreases.
Full Notes Atomic Radius
Elements with a different number of outer electrons behave differently. This means as you move across a period in the periodic table, elements will exhibit different characteristics. These differences are repeated for each period; we describe these patterns as ‘periodic trends’.
As you go across a period, atomic radius decreases.
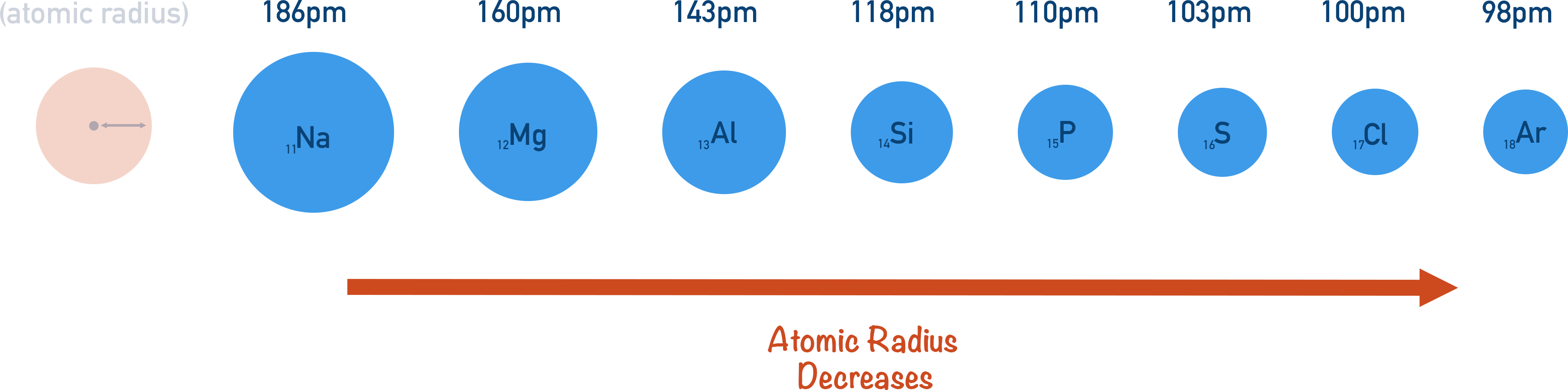
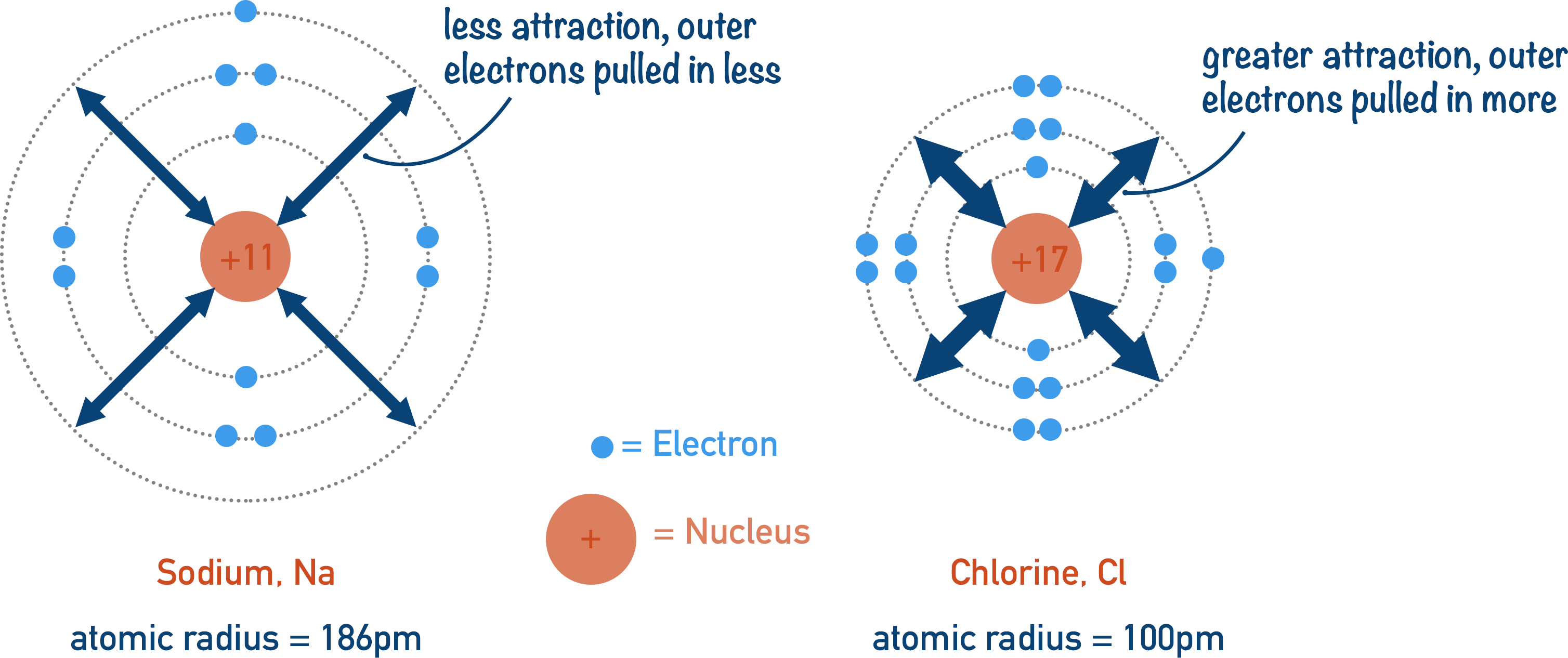
Each time you move across a period by one element, the atomic number increases by one; one extra proton and one extra electron are added to an atom of the element. The positive charge of the nucleus increases by one, and the added electrons go into the same outer shell as all the other elements in that period. This increases the electrostatic attraction between the nucleus and the outermost electrons. As the attraction is greater, the electrons are ‘pulled’ in tighter to the nucleus, decreasing the atomic radius.
As you go down a group, atomic radius increases.
Moving down the group, the number of electron shells increases. So, the outermost electrons have more inner shells between them and the nucleus. These extra inner shells effectively weaken the positive charge from the nucleus that reaches the outermost electrons; this is called inner electron shielding. As the inner electrons experience a greater positive charge than the outer electrons, the outer electrons are not ‘pulled’ in as tightly to the nucleus, increasing the atomic radius.