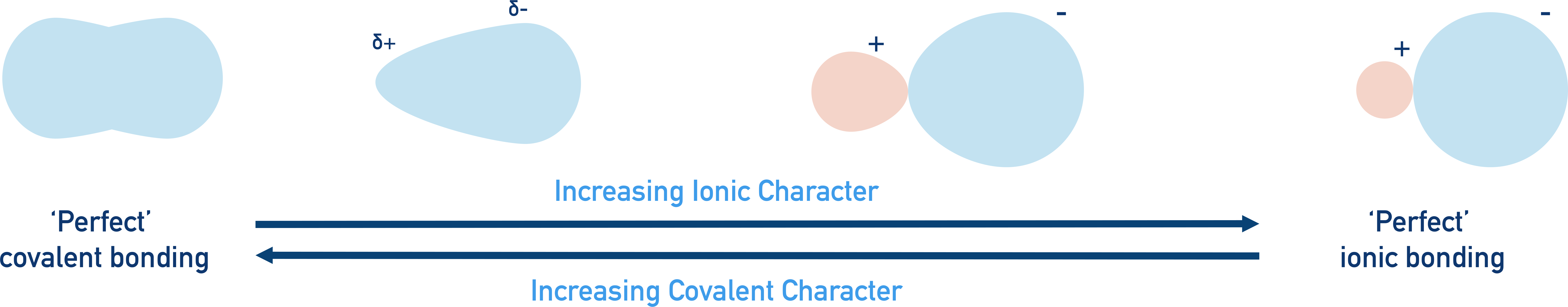
Quick Notes Ionic-Covalent Character
- The covalent bonding model shows electrons perfectly shared between atoms.
- In reality, electrons can be shared unequally, and the atoms in the bond can have partial charges.
- The partial charges attract, and the covalent bond now starts to behave a little like an ionic bond – it has ‘ionic character’.
- The ionic bonding model shows positively and negatively charged ions as spheres attracted to each other.
- In reality, electrons can be pulled away from the negatively charged ion towards to positively charged ion.
- Electrons are now slightly ‘shared’ between the two ions – the ionic bond has covalent character.
- The extent of ionic or covalent character is linked to the difference in electronegativities of the bonding elements.
Full Notes Ionic-Covalent Character
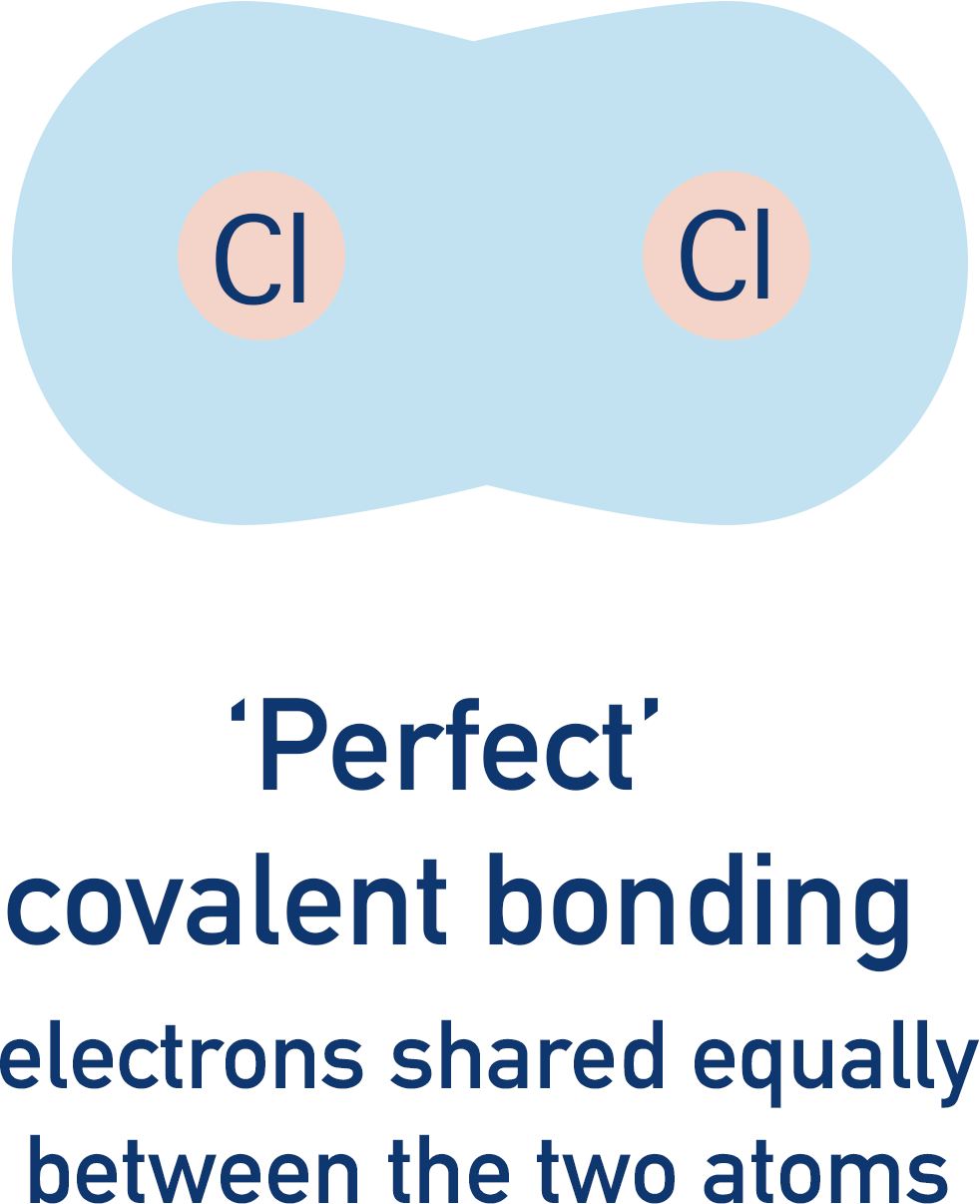
In a perfect covalent bond, one pair of electrons is shared equally between two atoms. Each atom has an ‘equal’ share of the electrons, and there is equal distribution of electrons across the bond.
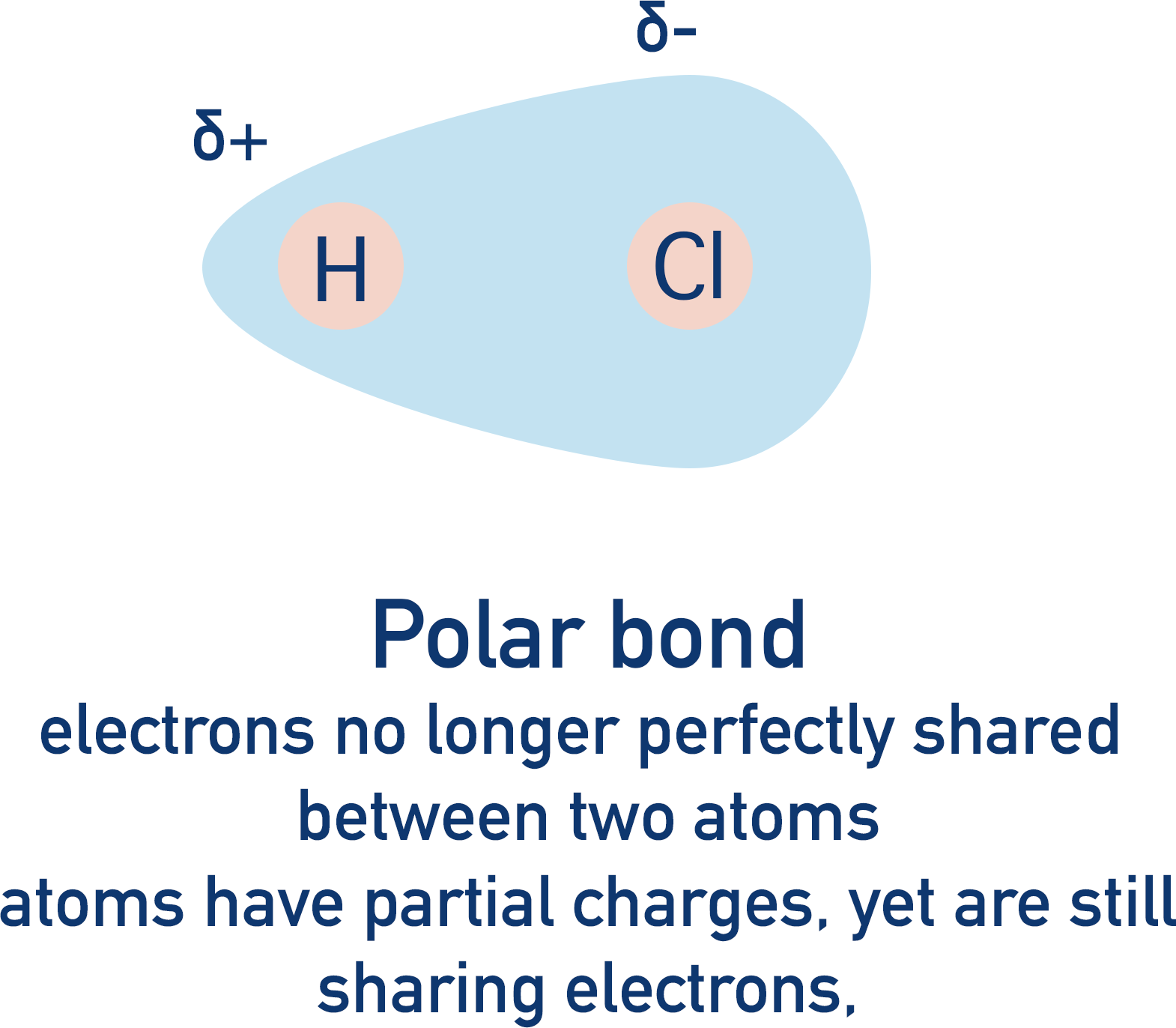
Very few covalent bonds are ‘perfect’ – electrons are very rarely found equally shared between two atoms. Electronegativity values show us that certain atoms pull electrons towards themselves in a covalent bond more than others. This creates unequal electron distribution in the bond and makes it polar. If the bonds become very polar, the bonding atoms start to behave like charged ions; the ‘covalent’ bonding starts to resemble ionic bonding. The covalent bond is said to have ‘ionic character’.
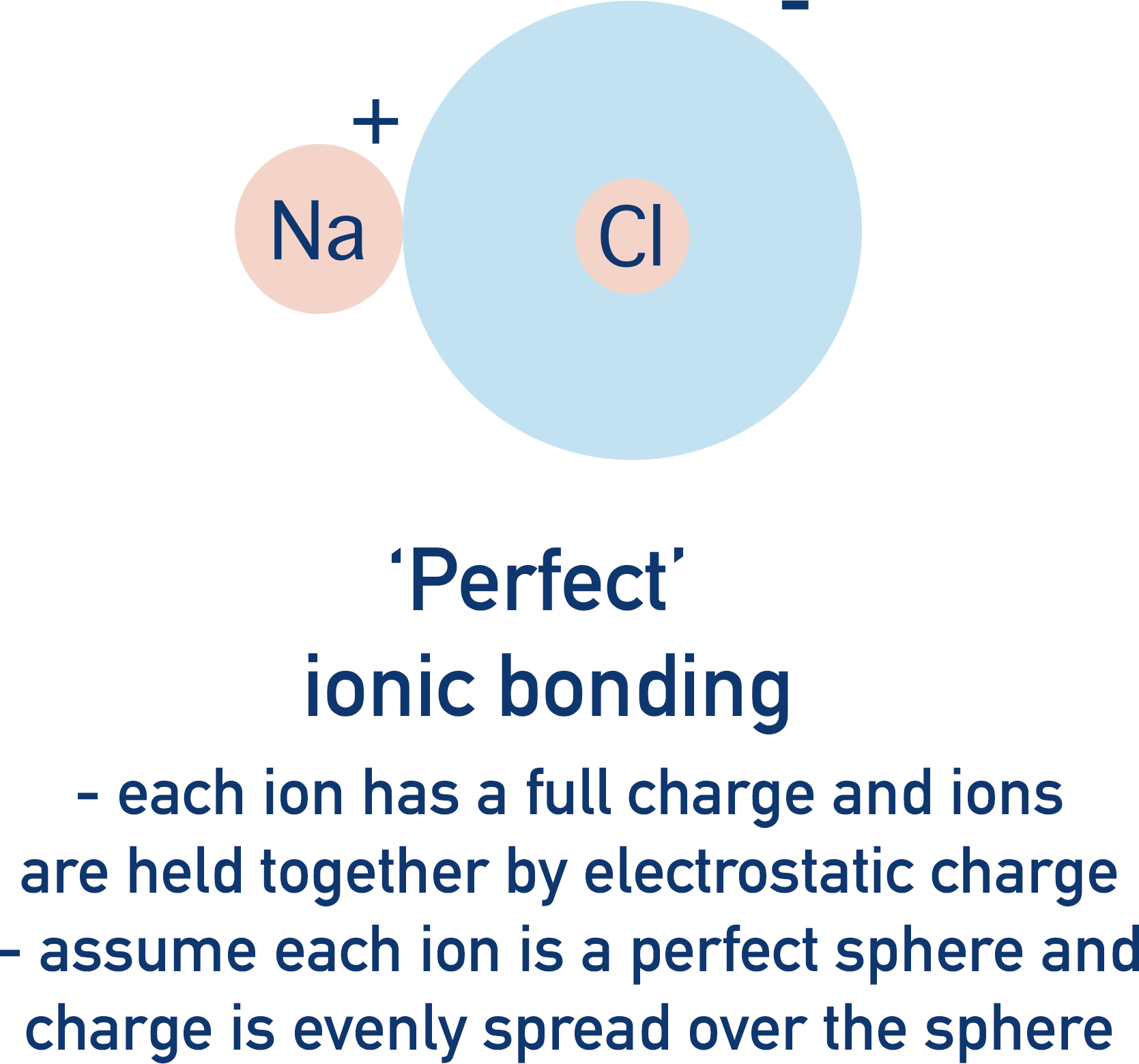
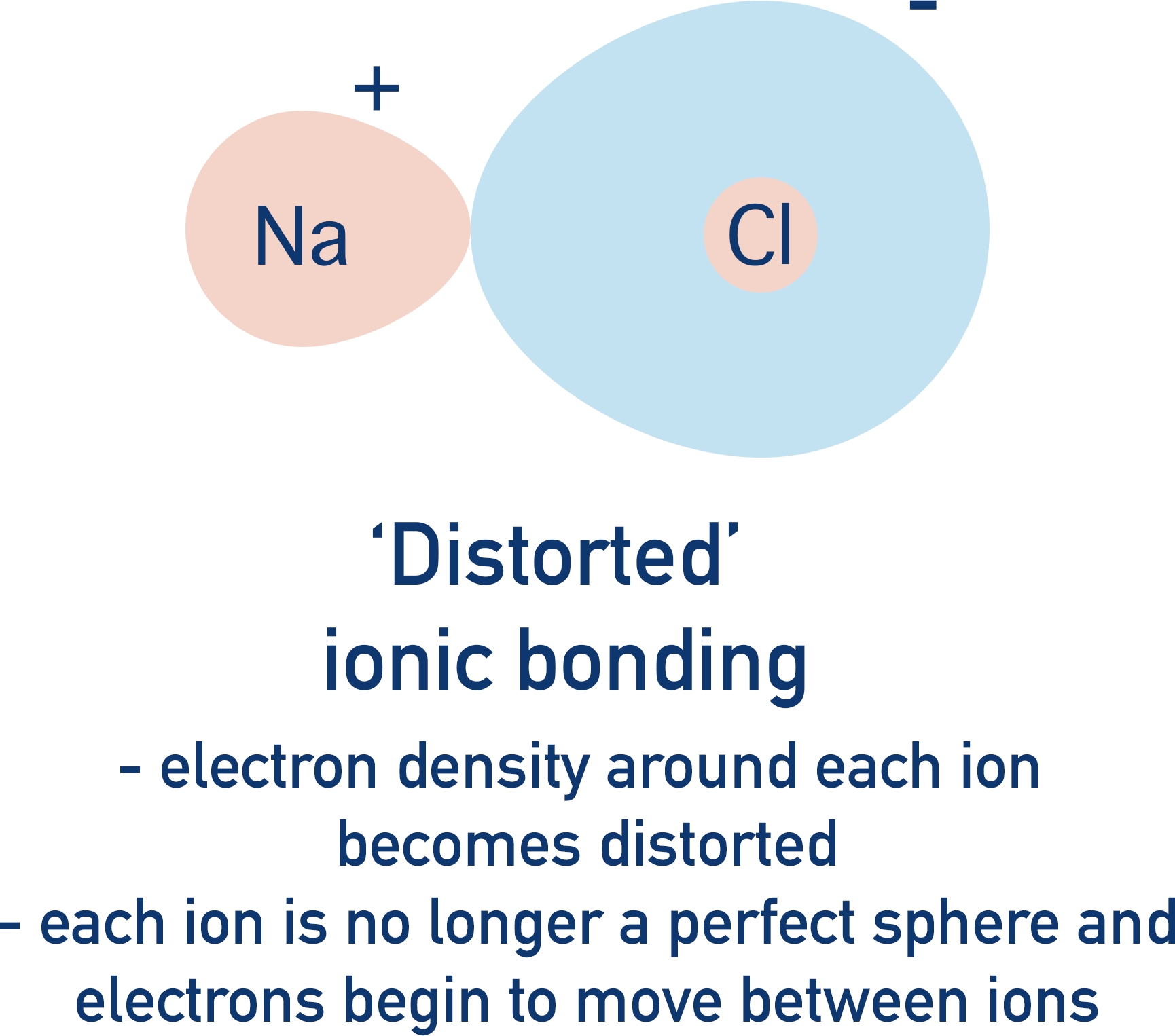
In perfect ionic bonding, oppositely charged ions are attracted to each other and can be considered as charged ‘spheres’, with full positive and negative charges evenly spread around the ions.
Again, the reality is a little messier. A positively charged ion will pull electrons away from a negatively charged ion towards itself. If the electrons are pulled away from the negatively charged ion enough, the electrons are held ‘between’ the ions; the ionic bonding starts to exhibit covalent properties – electrons being shared between two atoms. The ionic bonding is said to have ‘covalent character’.
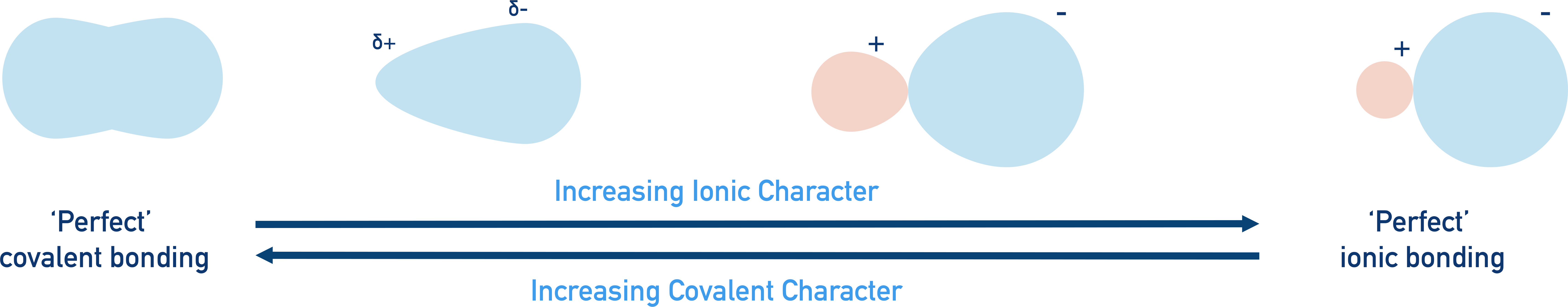
The greater the difference in electronegativity between two bonding elements, the greater the ionic character of a bond.
The smaller the difference in electronegativity between two bonding elements, the greater the covalent character of a bond.
In reality, most bonds are somewhere between perfect ionic and perfect covalent.
Effect on Melting and Boiling Points
It is important to recognise that ionic and covalent bonding are models used to describe bonding and, as with most models, they are simplifications. Simplified explanations to complicated topics are rarely a good idea, but students often find the two examples below contradict each other. As a result, these have tried to be explained in a simplified manner.
When ionic substances exhibit covalent character, it can cause higher than expected melting and boiling points. It can also, however, cause lower than expected melting and boiling points. Students can often be confused by the apparent contradiction.
The actual melting and boiling points that arise are due to the arrangement of ions and atoms in a structure.
Bonding in silver chloride, AgCl
The ionic bonding in AgCl exhibits covalent character and, due to the arrangement of ions in the solid structure, a new ‘sub-substructure’ arises. This is a bit like a secondary bond, meaning the structure now has (weakened) ionic bonding and a covalent-type bonding system. The two types of bonding make the structure stronger, increasing its melting and boiling points.
Bonding in aluminium oxide, Al2O3
The ionic bonding in Al2O3 also exhibits covalent character. However, unlike in silver chloride, the arrangement of ions means a sub-structure is unable to form. This means the ionic bonding is weakened by the polarising of the charged aluminium and oxide ions, and no covalent-type bonding system can form. This makes the structure slightly weaker, decreasing its melting and boiling points.
