IMFs and Ion–Dipole Forces
i. The molecules are of the same chemical species.
ii. The molecules are of two different chemical species.
Quick Notes
- Intermolecular forces (IMFs) are forces between molecules, not within them.
- Types of IMFs (in increasing strength for similar sized molecules):
- London dispersion forces (LDFs) – between all molecules
- Dipole–dipole forces – between polar molecules
- Hydrogen bonding – special strong dipole–dipole interaction (H bonded to N, O, or F)
- Ion–dipole forces – between ions and polar molecules (strongest type of IMF)
- Larger molecules form stronger LDFs due to greater polarizability
- Hydrogen bonding and ion–dipole interactions are key in biological molecules
- Orientation and charge differences affect IMF strength and behavior
Full Notes
Intermolecular forces (IMFs) are the forces that exist between molecules. There are several different types.
1. London Dispersion Forces (LDFs)
London Dispersions Forces (LDFs) are attractions between temporary dipoles in molecules that occur due to random movement of electrons.

LDFs are present in all molecular substances, even noble gases and nonpolar molecules and are often the strongest IMFs in large, nonpolar molecules.
Factors affecting LDF strength:
- Number of electrons – more electrons means stronger dispersion forces
- Molecular size and shape – more surface contact between molecules means stronger interactions
- Polarizability – larger electron clouds distort more easily giving stronger forces
- Pi bonding also increases polarizability
Important: LDFs are a type of van der Waals force, but the terms are not interchangeable. Van der Waals forces include both dispersion and dipole interactions.
2. Dipole–Dipole Interactions
Dipole-dipole interactions occur between polar molecules with permanent dipoles. Molecules align so that positive ends attract negative ends of nearby dipoles. They are stronger than LDFs (if comparing molecules of similar size).
Example: HCl molecules attract each other via permanent dipole–dipole forces
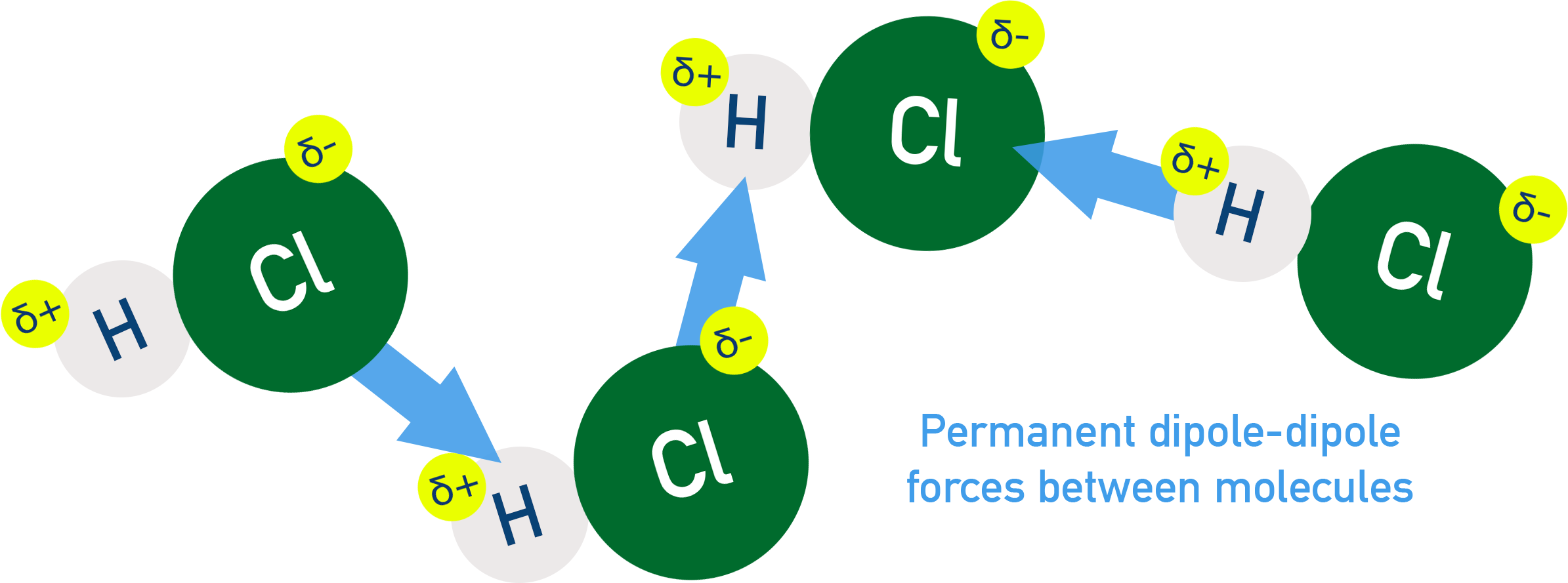
- Strength depends on:
- Magnitude of dipole moment
- Distance and orientation between dipoles
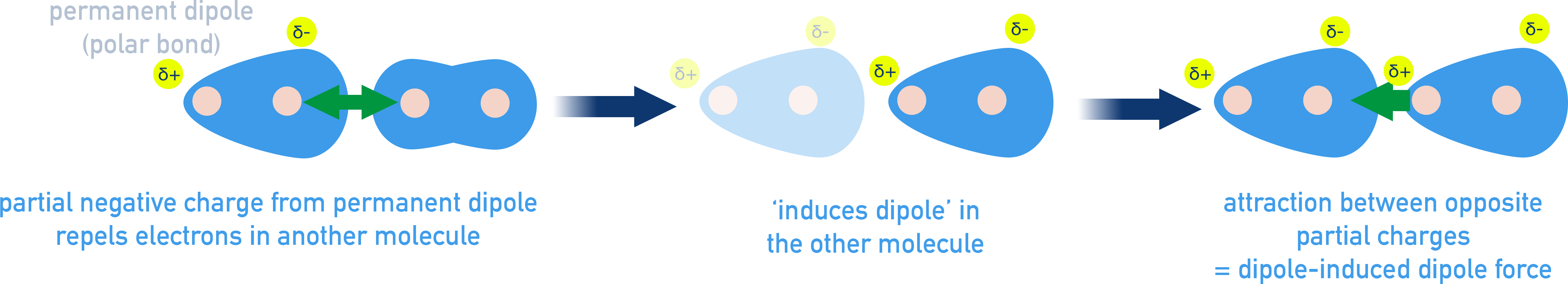
3. Dipole–Induced Dipole Forces
Dipole-Induced Forces occur when a polar molecule induces a temporary dipole in a nonpolar molecule. This causes an attraction between oppositely charged dipoles.
For ExampleH2O molecules have a permanent dipole that can induce a dipole in a non-polar molecule, such O2.
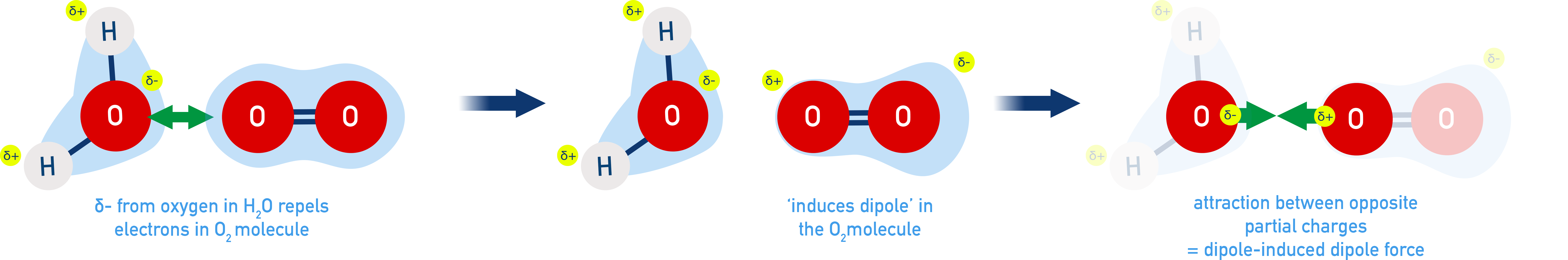
- Strength increases with:
- Larger dipole on the polar molecule
- Greater polarizability of the nonpolar molecule
4. Ion–Dipole Forces
Ion-Dipole Forces are attractions between a charged ion and oppositely charged end of a polar molecule.
They are stronger than dipole–dipole forces and key in dissolving ionic compounds in water (e.g., Li⁺ attracted to O end of H₂O)
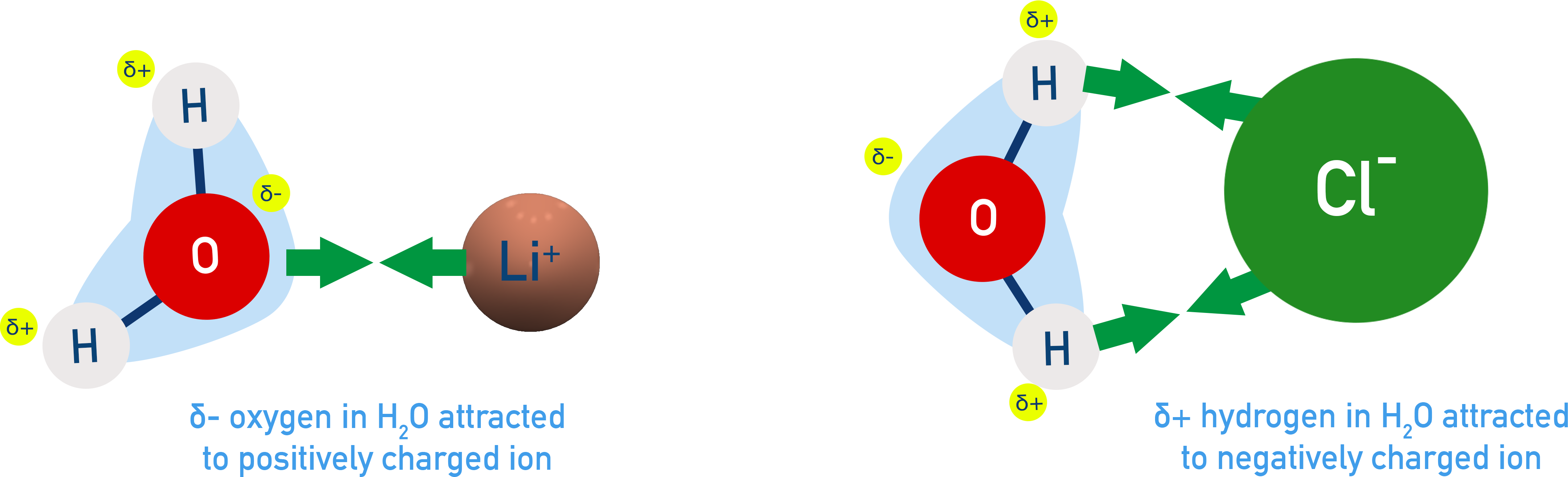
Strength depends on:
- Charge and size of the ion
- Magnitude and direction of the dipole
- Orientation matters – the negative end of a dipole is attracted to cations, the positive end to anions
5. Hydrogen Bonding
Hydrogen Bonding is a special type of strong dipole–dipole interaction that occurs when H is bonded to N, O, or F and is attracted to a lone pair of electrons on a N, O, or F atom in another molecule (or part of the same molecule).
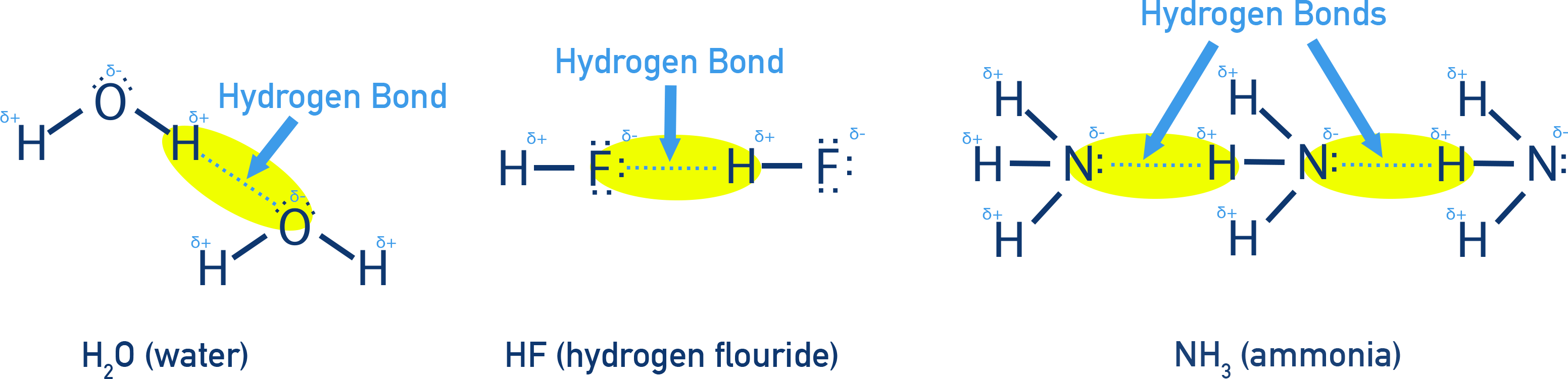
- Highly directional and relatively strong
- Critical in biological structures (e.g., DNA base pairing, protein folding)
6. Noncovalent Interactions in Biomolecules
In large biological molecules, IMFs are responsible for:
- Maintaining structure (e.g., alpha helices, beta sheets in proteins)
- Interactions between molecules (e.g., enzyme–substrate binding)
- Hydrogen bonding
- Dipole interactions
- Ion–dipole attractions
- Dispersion forces
Such interactions include:
See proteins and DNA for more information.
Question: Rank the IMFs present in the following interactions from weakest to strongest:
- Interaction between two Cl₂ molecules
- Interaction between two HCl molecules
- Interaction between Na⁺ and H₂O
- Interaction between HF and HF
Answer: 1 (Cl₂–Cl₂, LDF) < 2 (HCl–HCl, dipole–dipole) < 4 (HF–HF, hydrogen bonding) < 3 (Na⁺–H₂O, ion–dipole)

If H is bonded to N, O, or F and interacting with another N, O, or F — it's hydrogen bonding! Remember you must have those conditions for H-bonding to occur.
Summary
Intermolecular forces vary in strength and arise from electrostatic attractions between molecules, ions, or parts of molecules.
Key IMF types (in increasing strength for similar sized molecules):
- London dispersion: in all molecules; stronger in larger ones
- Dipole–induced dipole: between polar and nonpolar molecules
- Dipole–dipole: between polar molecules
- Hydrogen bonding: strong dipole–dipole when H is bonded to N, O, or F
- Ion–dipole: between ions and polar molecules; strongest IMF type
